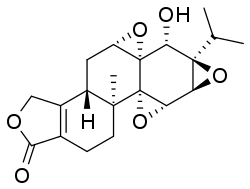
Triptolide
 | |
| Names | |
|---|---|
| Preferred IUPAC name
(3bS,4aS,5aS,6R,6aR,7aS,7bS,8aS,9bS)-6-Hydroxy-8b-methyl-6a-(propan-2-yl)-3b,4,4a,5a,6,6a,7a,7b,8a,8b,9,10-dodecahydrotris(oxireno)[2′,3′:4b,5;2′′,3′′:6,7;2′′′,3′′′:8a,9]phenanthro[1,2-c]furan-1(3H)-one | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.208.723 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
SMILES
| |
| Properties | |
| C20H24O6 | |
| Molar mass | 360.406 g·mol−1 |
| 0.017 mg/mL[1] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Triptolide is a
Triptolide is a component of
Mechanism of action
Several putative target proteins of triptolide have been reported, including
Water-soluble prodrugs
Minnelide is a more water-soluble synthetic prodrug of triptolide which is converted to triptolide in vivo.[3][14] In a preclinical mouse model of pancreatic cancer, it was "even more effective than gemcitabine". Its Phase II clinical trials are expected to conclude in February 2019.[15]

Glutriptolide, a glucose conjugate of triptolide with better solubility and lower toxicity, did not inhibit XPB activity in vitro, but exhibited tumor control in vivo, which is likely due to sustained stepwise release of active triptolide within cancer cells.[16] A second generation glutriptolide has been recently reported for targeting hypoxic cancer cells with increased glucose transporter expression.[17]
References
- PMID 26596892.
- PMID 17360534.
- ^ PMID 23076356.
- PMID 21255694.
- ^ "Study of Minnelide in Patients With Advanced GI Tumors". Retrieved 6 October 2016.
- ^ S. J. Leuenroth, D. Okuhara, J. D. Shotwell, G. S. Markowitz, Z. Yu, S. Somlo, C. M. Crews, Triptolide is a traditional Chinese medicine-derived inhibitor of polycystic kidney disease. Proc Natl Acad Sci U S A 2007, 104, 4389-4394;
- ^ R. Soundararajan, R. Sayat, G. S. Robertson, P. A. Marignani,Triptolide: An inhibitor of a disintegrin and metalloproteinase 10 (ADAM10) in cancer cells. Cancer Biol Ther 2009, 8, 2054-2062;
- ^ T. W. Corson, H. Cavga, N. Aberle, C. M. Crews, Triptolide directly inhibits dCTP pyrophosphatase. ChemBioChem 2011, 12, 1767-1773;
- ^ Y. Lu, Y. Zhang, L. Li, X. Feng, S. Ding, W.Zheng, J. Li, P. Shen,TAB1: A Target of Triptolide in Macrophages. Chem. Biol. 2014, 21, 246 – 256.
- ^ a b Q. L. He, D. V. Titov, J. Li, M. Tan, Z. Ye, Y. Zhao, D. Romo, and J. O. Liu. Covalent Modification of a Cysteine Residue in the XPB Subunit of the General Transcription Factor TFIIH Through Single Epoxide Cleavage of the Transcription Inhibitor Triptolide. Angew. Chem. Int. Ed. 2015, 54, 1859 –1863
- ^ D. V. Titov, B. Gilman, Q. L.He, S. Bhat,W. K. Low, Y. Dang,M.Smeaton, A. L. Demain, P. S. Miller, J. F. Kugel, J. A. Goodrich,J. O. Liu, XPB, a subunit of TFIIH, is a target of the natural product triptolide. Nat. Chem. Biol. 2011, 7, 182 – 188.
- ^ a b Y. Smurnyy, M. Cai, H. Wu, E. McWhinnie, J. A. Tallarico, Y.Yang, Y. Feng, DNA sequencing and CRISPR-Cas9 gene editing for target validation in mammalian cells. Nat. Chem. Biol. 2014, 10, 623 – 625
- PMID 33168807.
- ^ Thunder God Vine Drug Zaps Pancreatic Cancer. GenEng. 2012
- ^ "A Phase II, International Open Label Trial of Minnelide in Patients With Refractory Pancreatic Cancer". ClinicalTrials.gov. Retrieved 13 March 2018.
- .
- PMID 33083765.
You can help expand this article with text translated from the corresponding article in German. (October 2012) Click [show] for important translation instructions.
|
