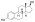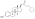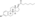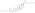Estradiol dipropionate
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Agofollin, Di-Ovocylin, Progynon DP, others |
| Other names | EDP; Estradiol dipropionate; Estradiol 3,17β-dipropionate; Estra-1,3,5(10)-triene-3,17β-diol 3,17β-dipropanoate |
| Routes of administration | Intramuscular injection |
| Drug class | Estrogen; Estrogen ester |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | IM: High[1] |
| Protein binding | Estradiol: ~98% (to albumin and SHBG)[2][3] |
| Metabolism | Cleavage via esterases in the liver, blood, and tissues[4][5] |
| Metabolites | Estradiol, benzoic acid, and metabolites of estradiol[4][5] |
| Elimination half-life | Unknown |
| Duration of action | IM (5 mg): 5–8 days[6][7] |
| Excretion | Urine |
| Identifiers | |
| |
JSmol) | |
| |
| |
Estradiol dipropionate (EDP), sold under the brand names Agofollin, Di-Ovocylin, and Progynon DP among others, is an
Estradiol dipropionate was patented in 1937[18] and was introduced for medical use by 1940.[19][20] It was one of the earliest estradiol esters to be used.[8] Along with estradiol benzoate, estradiol dipropionate was among the most widely used esters of estradiol for many years following its introduction.[15]
Medical uses
The
Estradiol dipropionate has typically been used at a dosage of 1 to 5 mg once or twice per week by
Available forms
Estradiol dipropionate was previously available by itself as an
Contraindications
Contraindications of estrogens include coagulation problems, cardiovascular diseases, liver disease, and certain hormone-sensitive cancers such as breast cancer and endometrial cancer, among others.[33][34][35][36]
Side effects
The
Overdose
Interactions
Inhibitors and inducers of cytochrome P450 may influence the metabolism of estradiol and by extension circulating estradiol levels.[37]
Pharmacology

Pharmacodynamics
Estradiol dipropionate is an
| Estrogen | Form | Dose (mg) | Duration by dose (mg) | ||
|---|---|---|---|---|---|
| EPD | CICD | ||||
| Estradiol | Aq. soln. | ? | – | <1 d | |
| Oil soln. | 40–60 | – | 1–2 ≈ 1–2 d | ||
| Aq. susp. | ? | 3.5 | 0.5–2 ≈ 2–7 d; 3.5 ≈ >5 d | ||
| Microsph. | ? | – | 1 ≈ 30 d | ||
| Estradiol benzoate | Oil soln. | 25–35 | – | 1.66 ≈ 2–3 d; 5 ≈ 3–6 d | |
| Aq. susp. | 20 | – | 10 ≈ 16–21 d | ||
| Emulsion | ? | – | 10 ≈ 14–21 d | ||
| Estradiol dipropionate | Oil soln. | 25–30 | – | 5 ≈ 5–8 d | |
| Estradiol valerate | Oil soln. | 20–30 | 5 | 5 ≈ 7–8 d; 10 ≈ 10–14 d; 40 ≈ 14–21 d; 100 ≈ 21–28 d | |
| Estradiol benz. butyrate | Oil soln. | ? | 10 | 10 ≈ 21 d | |
| Estradiol cypionate | Oil soln. | 20–30 | – | 5 ≈ 11–14 d | |
| Aq. susp. | ? | 5 | 5 ≈ 14–24 d | ||
Estradiol enanthate |
Oil soln. | ? | 5–10 | 10 ≈ 20–30 d | |
Estradiol dienanthate |
Oil soln. | ? | – | 7.5 ≈ >40 d | |
| Estradiol undecylate | Oil soln. | ? | – | 10–20 ≈ 40–60 d; 25–50 ≈ 60–120 d | |
| Polyestradiol phosphate | Aq. soln. | 40–60 | – | 40 ≈ 30 d; 80 ≈ 60 d; 160 ≈ 120 d | |
| Estrone | Oil soln. | ? | – | 1–2 ≈ 2–3 d | |
| Aq. susp. | ? | – | 0.1–2 ≈ 2–7 d | ||
| Estriol | Oil soln. | ? | – | 1–2 ≈ 1–4 d | |
| Polyestriol phosphate | Aq. soln. | ? | – | 50 ≈ 30 d; 80 ≈ 60 d | |
Notes and sources
Notes: All ovulation-inhibiting dose of estradiol undecylate is 20–30 mg/month. Sources: See template. | |||||
Pharmacokinetics
Compared to
A single intramuscular injection of 50 μg/kg estradiol dipropionate in oil in 15 pubertal girls (about 1 mg for a 50-kg (110-lb) girl) was found to produce peak estradiol levels of about 215 pg/mL after 1.5 days.[40] Estradiol levels declined to about 90 pg/mL after 4 days.[40]
Chemistry
Estradiol dipropionate, also known as estradiol 3,17β-dipropionate, is a
The experimental
| Estrogen | Structure | Ester(s) | Relative mol. weight |
Relative E2 contentb |
log Pc | ||||
|---|---|---|---|---|---|---|---|---|---|
| Position(s) | Moiet(ies) | Type | Lengtha | ||||||
| Estradiol | – | – | – | – | 1.00 | 1.00 | 4.0 | ||
| Estradiol acetate | C3 | Ethanoic acid |
Straight-chain fatty acid | 2 | 1.15 | 0.87 | 4.2 | ||
| Estradiol benzoate | C3 | Benzoic acid | Aromatic fatty acid | – (~4–5) | 1.38 | 0.72 | 4.7 | ||
| Estradiol dipropionate | C3, C17β | Propanoic acid (×2) |
Straight-chain fatty acid | 3 (×2) | 1.41 | 0.71 | 4.9 | ||
| Estradiol valerate | C17β | Pentanoic acid |
Straight-chain fatty acid | 5 | 1.31 | 0.76 | 5.6–6.3 | ||
| Estradiol benzoate butyrate | C3, C17β | Benzoic acid, butyric acid | Mixed fatty acid | – (~6, 2) | 1.64 | 0.61 | 6.3 | ||
| Estradiol cypionate | C17β | Cyclopentylpropanoic acid |
Cyclic fatty acid | – (~6) | 1.46 | 0.69 | 6.9 | ||
Estradiol enanthate |
C17β | Heptanoic acid |
Straight-chain fatty acid | 7 | 1.41 | 0.71 | 6.7–7.3 | ||
Estradiol dienanthate |
C3, C17β | Heptanoic acid (×2) |
Straight-chain fatty acid | 7 (×2) | 1.82 | 0.55 | 8.1–10.4 | ||
| Estradiol undecylate | C17β | Undecanoic acid |
Straight-chain fatty acid | 11 | 1.62 | 0.62 | 9.2–9.8 | ||
| Estradiol stearate | C17β | Octadecanoic acid |
Straight-chain fatty acid | 18 | 1.98 | 0.51 | 12.2–12.4 | ||
| Estradiol distearate | C3, C17β | Octadecanoic acid (×2) |
Straight-chain fatty acid | 18 (×2) | 2.96 | 0.34 | 20.2 | ||
| Estradiol sulfate | C3 | Sulfuric acid | Water-soluble conjugate | – | 1.29 | 0.77 | 0.3–3.8 | ||
| Estradiol glucuronide | C17β | Glucuronic acid | Water-soluble conjugate | – | 1.65 | 0.61 | 2.1–2.7 | ||
| Estramustine phosphated | C3, C17β | Normustine, phosphoric acid | Water-soluble conjugate | – | 1.91 | 0.52 | 2.9–5.0 | ||
| Polyestradiol phosphatee | C3–C17β | Phosphoric acid | Water-soluble conjugate | – | 1.23f | 0.81f | 2.9g | ||
| Footnotes: a = Length of hydrophobicity). Retrieved from PubChem, ChemSpider, and DrugBank. d = Also known as estradiol normustine phosphate. e = Polymer of estradiol phosphate (~13 repeat units ). f = Relative molecular weight or estradiol content per repeat unit. g = log P of repeat unit (i.e., estradiol phosphate). Sources: See individual articles.
| |||||||||
History
Estradiol dipropionate was first
Society and culture
Generic names
Estradiol dipropionate is the
Brand names
Estradiol dipropionate has been marketed under a variety of brand names, including Agofollin, Akrofolline, Dihidrofolina "Kével", Dimenformon, Dimenformon Dipropionate, Diovocylin, Di-Ovocylin, Diprostron, Diprovex, Endofollicolina D.P., EP Hormone Depot (in combination with
Availability
Estradiol dipropionate has been discontinued in most countries, but remains available in Japan and Macedonia alone under the brand names Ovahormon and Oestradiol Galenika and/or in combination with hydroxyprogesterone caproate under the brand name EP Hormone Depot.[11][13] It is also marketed for use in veterinary medicine in combination with hydroxyprogesterone caproate and nandrolone decanoate under the brand name Reepair in Australia.[13]
See also
References
- PMID 7169965.
- PMID 23375353.
- ISBN 978-0-323-03309-1.
- ^ ISBN 978-3-642-60107-1.
Natural estrogens considered here include: [...] Esters of 17β-estradiol, such as estradiol valerate, estradiol benzoate and estradiol cypionate. Esterification aims at either better absorption after oral administration or a sustained release from the depot after intramuscular administration. During absorption, the esters are cleaved by endogenous esterases and the pharmacologically active 17β-estradiol is released; therefore, the esters are considered as natural estrogens.
- ^ S2CID 24616324.
- ^ ISBN 978-3-662-00526-2.
- ^ ISBN 978-3-662-00942-0.
- ^ ISSN 0095-9561.
- ^ PMID 13638626.
- ^ ISBN 978-1-4757-2085-3.
- ^ ISBN 978-3-88763-075-1.
- ^ ISBN 978-94-011-4439-1.
- ^ a b c d e f g h "Estradiol: Uses, Dosage & Side Effects". Drugs.com.
- ^ S2CID 73253356.
- ^ PMID 14388061.
- ^ ISBN 978-3-642-68385-5.
- ^ ISBN 978-0-19-975569-1.
- ^ a b US patent 2233025, Miescher K, Scholz C, "Estradiol-17-monoesters", published 1941-02-25, assigned to Ciba Pharmaceutical Products, Inc.
- ^ ISSN 0013-7227.
The estradiol dipropionate used in this case was furnished by the Ciba Co. Their trade name for this product is Di-Ovocylin.
- ^ S2CID 75787837.
Grateful acknowledgment is made to Dr. Erwin Schwenk of the Schering Corporation for the estradiol benzoate (Progynon B), estradiol dipropionate (Progynon DP), progesterone (Proluton), and pregneninolone (Pranone) used in these experiments;
- ISBN 978-0-88416-175-2.
Intramuscular: For replacement therapy, (Estradiol, Estradiol Benzoate) 0.5 to 1.5 mg two or three times weekly; (Estradiol Cypionate) 1 to 5 mg weekly for two or three weeks; (Estradiol Dipropionate) 1 to 5 mg every one to two weeks; (Estradiol Valerate) 10 to 40 mg every one to four weeks.
- ^ ISSN 0095-9561.
- ^ Indian Pharmaceutical Guide. Pamposh Publications. 1968.
- ISBN 978-3-662-40303-7.
- ^ Hospital Formulary and Compendium of Useful Information. University of California Press. 1952. pp. 49–. GGKEY:2UAAZRZ5LN0.
- PMID 8271528.
- S2CID 22732235.
- PMID 2137212.
- S2CID 23267652.
- PMID 7513682.
- PMID 8360101.
- PMID 1752778.
- ^ PMID 2215269.
- ISBN 978-0-203-48612-2.
- ISBN 978-3-901299-34-6.
- ISBN 978-94-011-6167-1.
- PMID 11741520.
- ^ ISSN 0013-7227.
- ^ ISBN 978-1-4612-5525-3.
- ^ ISSN 0003-388X.
- ^ PMID 14388061.
- ^ "Estradiol Dipropionate | C24H32O4". ChemSpider.
- ^ ISSN 0002-9378.
- PMID 18746057.
- ^ ISSN 0002-9955.
- PMID 29646930.
- S2CID 72668930.
- ISSN 0025-7125.
- PMID 7389356.
- ISBN 978-0-89573-552-2.
- ISBN 978-92-832-1221-8.
- ISBN 978-0-470-18024-2.
- ISBN 978-1-4684-2007-4.

![Estradiol levels after a single intramuscular injection of 50 μg/kg estradiol dipropionate in oil in pubertal girls.[40] This dose would be 1 mg in a 50-kg (110-lb) girl.[40] Source was Presl et al. (1976).[40]](http://upload.wikimedia.org/wikipedia/commons/thumb/d/db/Estradiol_levels_after_a_single_intramuscular_injection_of_50_%CE%BCg_per_kg_estradiol_dipropionate_in_pubertal_girls.png/323px-Estradiol_levels_after_a_single_intramuscular_injection_of_50_%CE%BCg_per_kg_estradiol_dipropionate_in_pubertal_girls.png)
![Vaginal cornification with a single intramuscular injection of different estradiol esters in oil solution in women.[41] Source was Schwartz & Soule (1955).[41]](http://upload.wikimedia.org/wikipedia/commons/thumb/2/23/Vaginal_cornification_with_a_single_intramuscular_injection_of_different_estradiol_esters_in_women.png/395px-Vaginal_cornification_with_a_single_intramuscular_injection_of_different_estradiol_esters_in_women.png)