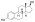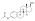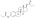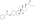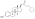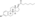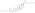Estradiol valerate
subcutaneous injection | |
| Drug class | Estrogen; Estrogen ester |
|---|---|
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Oral: 3–5%[4][5] IM injection: 100%[6][4] |
| Protein binding | Estradiol: ~98% (to albumin and SHBG)[7][8] |
| Metabolism | Cleavage via esterases in the liver, blood, and tissues[4] |
| Metabolites | Estradiol, valeric acid, and metabolites of estradiol[4] |
| Elimination half-life | Oral: 12–20 hours (as E2)[4][7] IM injection: 3.5 (1.2–7.2) days[9] |
| Duration of action | IM injection: • 5 mg: 7–8 days[10] • 10 mg: 10–14 days[11][12] • 40 mg: 2–3 weeks[11] • 100 mg: 3–4 weeks[11] |
| Excretion | Urine (80%)[4] |
| Identifiers | |
| |
JSmol) | |
| Melting point | 144 to 145 °C (291 to 293 °F) |
| |
| |
Estradiol valerate (EV), sold for use
Estradiol valerate was first described in 1940 and was introduced for medical use in 1954.[19][20][21] Along with estradiol cypionate, it is one of the most widely used esters of estradiol.[22] Estradiol valerate is used in the United States, Canada, Europe, and throughout much of the rest of the world.[23][24] It is available as a generic medication.[25]
Medical uses
The
In the
Estradiol valerate by intramuscular injection is usually used at a dosage of 10 to 20 mg every 4 weeks in the treatment of menopausal symptoms and hypoestrogenism due to hypogonadism, castration, or primary ovarian failure in women.: 60
| Route/form | Estrogen | Low | Standard | High | |||
|---|---|---|---|---|---|---|---|
| Oral | Estradiol | 0.5–1 mg/day | 1–2 mg/day | 2–4 mg/day | |||
| Estradiol valerate | 0.5–1 mg/day | 1–2 mg/day | 2–4 mg/day | ||||
| Estradiol acetate | 0.45–0.9 mg/day | 0.9–1.8 mg/day | 1.8–3.6 mg/day | ||||
| Conjugated estrogens | 0.3–0.45 mg/day | 0.625 mg/day | 0.9–1.25 mg/day | ||||
| Esterified estrogens | 0.3–0.45 mg/day | 0.625 mg/day | 0.9–1.25 mg/day | ||||
| Estropipate | 0.75 mg/day | 1.5 mg/day | 3 mg/day | ||||
| Estriol | 1–2 mg/day | 2–4 mg/day | 4–8 mg/day | ||||
| Ethinylestradiola | 2.5–10 μg/day | 5–20 μg/day | – | ||||
| Nasal spray | Estradiol | 150 μg/day | 300 μg/day | 600 μg/day | |||
| Transdermal patch | Estradiol | 25 μg/dayb | 50 μg/dayb | 100 μg/dayb | |||
Transdermal gel |
Estradiol | 0.5 mg/day | 1–1.5 mg/day | 2–3 mg/day | |||
Vaginal |
Estradiol | 25 μg/day | – | – | |||
| Estriol | 30 μg/day | 0.5 mg 2x/week | 0.5 mg/day | ||||
SC injection |
Estradiol valerate | – | – | 4 mg 1x/4 weeks | |||
| Estradiol cypionate | 1 mg 1x/3–4 weeks | 3 mg 1x/3–4 weeks | 5 mg 1x/3–4 weeks | ||||
| Estradiol benzoate | 0.5 mg 1x/week | 1 mg 1x/week | 1.5 mg 1x/week | ||||
| SC implant | Estradiol | 25 mg 1x/6 months | 50 mg 1x/6 months | 100 mg 1x/6 months | |||
| Footnotes: a = No longer used or recommended, due to health concerns. b = As a single patch applied once or twice per week (worn for 3–4 days or 7 days), depending on the formulation. Note: Dosages are not necessarily equivalent. Sources: See template. | |||||||
Available forms
Estradiol valerate is and has been available in the form of
In addition to single-drug formulations, oral estradiol valerate is available in combination with the
| Route | Ingredient | Form | Dose[b] | Brand names[c] |
|---|---|---|---|---|
| Oral | Estradiol | Tablet | 0.1, 0.2, 0.5, 1, 2, 4 mg | Estrace, Ovocyclin |
| Estradiol valerate | Tablet | 0.5, 1, 2, 4 mg | Progynova | |
Transdermal
|
Estradiol | Patch | 14, 25, 37.5, 50, 60, 75, 100 µg/d | Climara, Vivelle |
| Gel pump | 0.06% (0.52, 0.75 mg/pump) | Elestrin, EstroGel | ||
| Gel packet | 0.1% (0.25, 0.5, 1.0 mg/pk.) | DiviGel, Sandrena | ||
| Emulsion | 0.25% (25 µg/pouch) | Estrasorb | ||
| Spray | 1.53 mg/spray | Evamist, Lenzetto | ||
Vaginal
|
Estradiol | Tablet | 10, 25 µg | Vagifem |
| Cream | 0.01% (0.1 mg/gram) | Estrace | ||
| Insert | 4, 10 µg | Imvexxy | ||
| Ring | 2 mg/ring (7.5 µg/d, 3 mon.) | Estring | ||
| Estradiol acetate | Ring | 50, 100 µg/d, 3 months | Femring | |
| Injection[d] | Estradiol | Microspheres | 1 mg/mL | Juvenum E |
| Estradiol benzoate | Oil solution | 0.167, 0.2, 0.333, 1, 1.67, 2, 5, 10, 20, 25 mg/mL | Progynon-B | |
| Estradiol cypionate | Oil solution | 1, 3, 5 mg/mL | Depo-Estradiol | |
| Estradiol valerate | Oil solution | 5, 10, 20, 40 mg/mL | Progynon Depot | |
Implant
|
Estradiol | Pellet | 20, 25, 50, 100 mg, 6 mon. | Estradiol Implants |
Contraindications
Contraindications of estrogens include coagulation problems, cardiovascular diseases, liver disease, and certain hormone-sensitive cancers such as breast cancer and endometrial cancer, among others.[58][59][60][61]
Side effects
The
Overdose
Estradiol valerate has been used at very high doses of 40 to 100 mg once per week in women and men, without overt signs of acute
Interactions
Inhibitors and inducers of cytochrome P450 may influence the metabolism of estradiol and by extension circulating estradiol levels.[74]
Pharmacology

Pharmacodynamics
Estradiol valerate is an
| Estrogen | Other names | RBA (%)a
|
REP (%)b | |||
|---|---|---|---|---|---|---|
| ER | ERα
|
ERβ
| ||||
| Estradiol | E2 | 100 | 100 | 100 | ||
Estradiol 3-sulfate
|
E2S; E2-3S | ? | 0.02 | 0.04 | ||
| Estradiol 3-glucuronide | E2-3G | ? | 0.02 | 0.09 | ||
Estradiol 17β-glucuronide
|
E2-17G | ? | 0.002 | 0.0002 | ||
| Estradiol benzoate | EB; Estradiol 3-benzoate | 10 | 1.1 | 0.52 | ||
| Estradiol 17β-acetate | E2-17A | 31–45 | 24 | ? | ||
| Estradiol diacetate | EDA; Estradiol 3,17β-diacetate | ? | 0.79 | ? | ||
Estradiol propionate
|
EP; Estradiol 17β-propionate | 19–26 | 2.6 | ? | ||
| Estradiol valerate | EV; Estradiol 17β-valerate | 2–11 | 0.04–21 | ? | ||
| Estradiol cypionate | EC; Estradiol 17β-cypionate | ?c | 4.0 | ? | ||
| Estradiol palmitate | Estradiol 17β-palmitate | 0 | ? | ? | ||
| Estradiol stearate | Estradiol 17β-stearate | 0 | ? | ? | ||
| Estrone | E1; 17-Ketoestradiol | 11 | 5.3–38 | 14 | ||
| Estrone sulfate | E1S; Estrone 3-sulfate | 2 | 0.004 | 0.002 | ||
| Estrone glucuronide | E1G; Estrone 3-glucuronide | ? | <0.001 | 0.0006 | ||
| Ethinylestradiol | EE; 17α-Ethynylestradiol | 100 | 17–150 | 129 | ||
| Mestranol | EE 3-methyl ether | 1 | 1.3–8.2 | 0.16 | ||
| Quinestrol | EE 3-cyclopentyl ether | ? | 0.37 | ? | ||
| Footnotes: a = ERβ. Both mammalian cells and yeast have the capacity to hydrolyze estrogen esters. c = The affinities of estradiol cypionate for the ERs are similar to those of estradiol valerate and estradiol benzoate (figure ). Sources: See template page.
| ||||||
| Compound | Dosage for specific uses (mg usually)[a] | ||||||
|---|---|---|---|---|---|---|---|
| ETD[b] | EPD[b] | MSD[b] | MSD[c] | OID[c] | TSD[c] | ||
| Estradiol (non-micronized) | 30 | ≥120–300 | 120 | 6 | - | - | |
| Estradiol (micronized) | 6–12 | 60–80 | 14–42 | 1–2 | >5 | >8 | |
| Estradiol valerate | 6–12 | 60–80 | 14–42 | 1–2 | - | >8 | |
| Estradiol benzoate | - | 60–140 | - | - | - | - | |
| Estriol | ≥20 | 120–150[d] | 28–126 | 1–6 | >5 | - | |
| Estriol succinate | - | 140–150[d] | 28–126 | 2–6 | - | - | |
| Estrone sulfate | 12 | 60 | 42 | 2 | - | - | |
| Conjugated estrogens | 5–12 | 60–80 | 8.4–25 | 0.625–1.25 | >3.75 | 7.5 | |
| Ethinylestradiol | 200 μg | 1–2 | 280 μg | 20–40 μg | 100 μg | 100 μg | |
| Mestranol | 300 μg | 1.5–3.0 | 300–600 μg | 25–30 μg | >80 μg | - | |
| Quinestrol | 300 μg | 2–4 | 500 μg | 25–50 μg | - | - | |
| Methylestradiol | - | 2 | - | - | - | - | |
| Diethylstilbestrol | 2.5 | 20–30 | 11 | 0.5–2.0 | >5 | 3 | |
| DES dipropionate | - | 15–30 | - | - | - | - | |
| Dienestrol | 5 | 30–40 | 42 | 0.5–4.0 | - | - | |
| Dienestrol diacetate | 3–5 | 30–60 | - | - | - | - | |
| Hexestrol | - | 70–110 | - | - | - | - | |
| Chlorotrianisene | - | >100 | - | - | >48 | - | |
| Methallenestril | - | 400 | - | - | - | - | |
| Estrogen | HF |
VE | UCa | FSH | LH | HDL-C | SHBG | CBG |
AGT |
Liver |
|---|---|---|---|---|---|---|---|---|---|---|
| Estradiol | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Estrone | ? | ? | ? | 0.3 | 0.3 | ? | ? | ? | ? | ? |
| Estriol | 0.3 | 0.3 | 0.1 | 0.3 | 0.3 | 0.2 | ? | ? | ? | 0.67 |
| Estrone sulfate | ? | 0.9 | 0.9 | 0.8–0.9 | 0.9 | 0.5 | 0.9 | 0.5–0.7 | 1.4–1.5 | 0.56–1.7 |
| Conjugated estrogens | 1.2 | 1.5 | 2.0 | 1.1–1.3 | 1.0 | 1.5 | 3.0–3.2 | 1.3–1.5 | 5.0 | 1.3–4.5 |
Equilin sulfate |
? | ? | 1.0 | ? | ? | 6.0 | 7.5 | 6.0 | 7.5 | ? |
| Ethinylestradiol | 120 | 150 | 400 | 60–150 | 100 | 400 | 500–600 | 500–600 | 350 | 2.9–5.0 |
| Diethylstilbestrol | ? | ? | ? | 2.9–3.4 | ? | ? | 26–28 | 25–37 | 20 | 5.7–7.5 |
Sources and footnotes
Notes: Values are ratios, with estradiol as standard (i.e., 1.0). Abbreviations: HF = Clinical relief of liver proteins. Liver = Ratio of liver estrogenic effects to general/systemic estrogenic effects (hot flashes/gonadotropins ). Sources: See template. | ||||||||||
| Estrogen | Form | Dose (mg) | Duration by dose (mg) | ||
|---|---|---|---|---|---|
| EPD | CICD | ||||
| Estradiol | Aq. soln. | ? | – | <1 d | |
| Oil soln. | 40–60 | – | 1–2 ≈ 1–2 d | ||
| Aq. susp. | ? | 3.5 | 0.5–2 ≈ 2–7 d; 3.5 ≈ >5 d | ||
| Microsph. | ? | – | 1 ≈ 30 d | ||
| Estradiol benzoate | Oil soln. | 25–35 | – | 1.66 ≈ 2–3 d; 5 ≈ 3–6 d | |
| Aq. susp. | 20 | – | 10 ≈ 16–21 d | ||
| Emulsion | ? | – | 10 ≈ 14–21 d | ||
| Estradiol dipropionate | Oil soln. | 25–30 | – | 5 ≈ 5–8 d | |
| Estradiol valerate | Oil soln. | 20–30 | 5 | 5 ≈ 7–8 d; 10 ≈ 10–14 d; 40 ≈ 14–21 d; 100 ≈ 21–28 d | |
| Estradiol benz. butyrate | Oil soln. | ? | 10 | 10 ≈ 21 d | |
| Estradiol cypionate | Oil soln. | 20–30 | – | 5 ≈ 11–14 d | |
| Aq. susp. | ? | 5 | 5 ≈ 14–24 d | ||
Estradiol enanthate |
Oil soln. | ? | 5–10 | 10 ≈ 20–30 d | |
Estradiol dienanthate |
Oil soln. | ? | – | 7.5 ≈ >40 d | |
| Estradiol undecylate | Oil soln. | ? | – | 10–20 ≈ 40–60 d; 25–50 ≈ 60–120 d | |
| Polyestradiol phosphate | Aq. soln. | 40–60 | – | 40 ≈ 30 d; 80 ≈ 60 d; 160 ≈ 120 d | |
| Estrone | Oil soln. | ? | – | 1–2 ≈ 2–3 d | |
| Aq. susp. | ? | – | 0.1–2 ≈ 2–7 d | ||
| Estriol | Oil soln. | ? | – | 1–2 ≈ 1–4 d | |
| Polyestriol phosphate | Aq. soln. | ? | – | 50 ≈ 30 d; 80 ≈ 60 d | |
Notes and sources
Notes: All ovulation-inhibiting dose of estradiol undecylate is 20–30 mg/month. Sources: See template. | |||||
Effects on liver protein synthesis
The influence of 2 mg/day oral estradiol valerate on
Pharmacokinetics
Regardless of the
Oral administration
A dosage of 1 mg/day oral estradiol valerate has been found to produce approximate circulating concentrations of 50 pg/mL estradiol and 160 pg/mL estrone, while a dosage of 2 mg/day results in circulating levels of 60 pg/mL estradiol and 300 pg/mL estrone.[108] These concentrations of estradiol and estrone are comparable to those observed with 1 and 2 mg/day oral estradiol.[108] A review of selected studies reported a range of mean peak estradiol levels of 24 to 140 pg/mL occurring 1 to 12 hours after administration of 2 mg oral estradiol valerate.[4] A study found that, in accordance with their differences in molecular weights, oral estradiol produced higher levels of estradiol than oral estradiol valerate.[109] Likewise, other studies found that levels of estradiol and estrone are very similar after oral administration of roughly equimolar doses of estradiol (1.5 mg) and estradiol valerate (2 mg).[110][111][112] A study of high-dose oral estradiol valerate found levels of estradiol of about 250 pg/mL after a single 10-mg dose in three women.[107]
Sublingual administration

Estradiol valerate has been studied by
The administration of 2 mg oral micronized estradiol valerate tablets (Progynova, Schering) sublingually 3 or 4 times per day has been found to result in circulating estradiol levels of about 290 pg/mL to 460 pg/mL in premenopausal women (time of measurements not given).
Intramuscular injection
In contrast to oral administration, the bioavailability of estradiol valerate is complete (i.e., 100%) via intramuscular injection.
A single intramuscular injection of 4 mg estradiol valerate has been found to result in maximal circulating levels of estradiol of about 390 pg/mL within 3 days of administration, with levels declining to 100 pg/mL (baseline, in the study) by 12 to 13 days.[43] Studies in general have found that a single intramuscular injection of 4 mg estradiol valerate results in peak levels of estradiol of 240 to 540 pg/mL after 1 to 5 days following administration.[120] A study found that a single intramuscular injection of 5 mg estradiol valerate resulted in peak circulating levels of 667 pg/mL estradiol and 324 pg/mL estrone within approximately 2 and 3 days, respectively.[10] The duration of estradiol valerate at this dose and in this study was considered to be 7 to 8 days.[10] Other studies have found that larger doses of intramuscular estradiol valerate exceeding 20 mg have a duration of more than 15 days.[10] A third study, in contrast to the preceding study, found that a single 10 mg intramuscular injection of estradiol valerate resulted in maximal estradiol levels of 506 to 544 pg/mL and maximal estrone levels of 205 to 219 pg/mL in postmenopausal women.[9]
With intramuscular injections of estradiol valerate, it has been reported that a dose of 5 mg has a duration of 7 to 8 days, 10 mg a duration of 10 to 14 days, 40 mg a duration of 2 to 3 weeks (14 to 21 days), and 100 mg a duration of 3 to 4 weeks (21 to 28 days).[11][12][10]
A study of
| Estrogen | Dose | Cmax | Tmax |
Duration |
|---|---|---|---|---|
| Estradiol benzoate | 5 mg | E2: 940 pg/mL E1: 343 pg/mL |
E2: 1.8 days E1: 2.4 days |
4–5 days |
| Estradiol valerate | 5 mg | E2: 667 pg/mL E1: 324 pg/mL |
E2: 2.2 days E1: 2.7 days |
7–8 days |
| Estradiol cypionate | 5 mg | E2: 338 pg/mL E1: 145 pg/mL |
E2: 3.9 days E1: 5.1 days |
11 days |
| Notes: All via chromatographic separation . Sources: See template.
| ||||
-
Hormone levels after a single intramuscular injection of 5 mg estradiol valerate in oil in 17 postmenopausal women.[121] Assays were performed using EIA.[121] Estrone levels were likely overestimated, possibly due to cross reactivity of the assay with estrogen conjugates.[9] Source was Göretzlehner et al. (2002).[121]
-
Estradiol levels after a single intramuscular injection of 10 mg estradiol valerate or 100 mg estradiol undecylate in oil both in 4 individuals each.[123] Subject characteristics and assay method were not described.[123] Source was Vermeulen (1975).[123]
-
Estradiol and DHEA levels after a single intramuscular injection of Gynodian Depot (4 mg estradiol valerate, 200 mg prasterone enanthate in oil) or Primogyn Depot (10 mg estradiol valerate in oil) in women.[124][119][125] Assays were performed using RIA.[119][125] Sources were Düsterberg & Wendt (1983) and Rauramo et al. (1980).[124][119][125]
Subcutaneous injection
Estradiol esters like estradiol valerate and
Intravenous injection
The administration of estradiol valerate by intravenous injection has been studied.
Chemistry

Estradiol valerate is a
The experimental log
| Estrogen | Structure | Ester(s) | Relative mol. weight |
Relative E2 contentb |
log Pc | ||||
|---|---|---|---|---|---|---|---|---|---|
| Position(s) | Moiet(ies) | Type | Lengtha | ||||||
| Estradiol | – | – | – | – | 1.00 | 1.00 | 4.0 | ||
| Estradiol acetate | C3 | Ethanoic acid |
Straight-chain fatty acid | 2 | 1.15 | 0.87 | 4.2 | ||
| Estradiol benzoate | C3 | Benzoic acid | Aromatic fatty acid | – (~4–5) | 1.38 | 0.72 | 4.7 | ||
| Estradiol dipropionate | C3, C17β | Propanoic acid (×2) |
Straight-chain fatty acid | 3 (×2) | 1.41 | 0.71 | 4.9 | ||
| Estradiol valerate | C17β | Pentanoic acid |
Straight-chain fatty acid | 5 | 1.31 | 0.76 | 5.6–6.3 | ||
| Estradiol benzoate butyrate | C3, C17β | Benzoic acid, butyric acid | Mixed fatty acid | – (~6, 2) | 1.64 | 0.61 | 6.3 | ||
| Estradiol cypionate | C17β | Cyclopentylpropanoic acid |
Cyclic fatty acid | – (~6) | 1.46 | 0.69 | 6.9 | ||
Estradiol enanthate |
C17β | Heptanoic acid |
Straight-chain fatty acid | 7 | 1.41 | 0.71 | 6.7–7.3 | ||
Estradiol dienanthate |
C3, C17β | Heptanoic acid (×2) |
Straight-chain fatty acid | 7 (×2) | 1.82 | 0.55 | 8.1–10.4 | ||
| Estradiol undecylate | C17β | Undecanoic acid |
Straight-chain fatty acid | 11 | 1.62 | 0.62 | 9.2–9.8 | ||
| Estradiol stearate | C17β | Octadecanoic acid |
Straight-chain fatty acid | 18 | 1.98 | 0.51 | 12.2–12.4 | ||
| Estradiol distearate | C3, C17β | Octadecanoic acid (×2) |
Straight-chain fatty acid | 18 (×2) | 2.96 | 0.34 | 20.2 | ||
| Estradiol sulfate | C3 | Sulfuric acid | Water-soluble conjugate | – | 1.29 | 0.77 | 0.3–3.8 | ||
| Estradiol glucuronide | C17β | Glucuronic acid | Water-soluble conjugate | – | 1.65 | 0.61 | 2.1–2.7 | ||
| Estramustine phosphated | C3, C17β | Normustine, phosphoric acid | Water-soluble conjugate | – | 1.91 | 0.52 | 2.9–5.0 | ||
| Polyestradiol phosphatee | C3–C17β | Phosphoric acid | Water-soluble conjugate | – | 1.23f | 0.81f | 2.9g | ||
| Footnotes: a = Length of hydrophobicity). Retrieved from PubChem, ChemSpider, and DrugBank. d = Also known as estradiol normustine phosphate. e = Polymer of estradiol phosphate (~13 repeat units ). f = Relative molecular weight or estradiol content per repeat unit. g = log P of repeat unit (i.e., estradiol phosphate). Sources: See individual articles.
| |||||||||
History
Estradiol valerate was
Society and culture
Generic names
Estradiol valerate is the
Brand names
Estradiol valerate has been marketed under the brand names Altadiol, Androtardyl-Oestradiol, Ardefem, Climaval, Cyclabil, Cyclocur, Deladiol, Delahormone Unimatic, Delestrogen, Delestrogen 4X, Depogen, Diol-20, Dioval, Ditate, Dura-Estate, Dura-Estradiol, Duratrad, Duragen, Estate, Estra-L, Estradiol Depot, Estraval, Estraval Depot, Estraval PA, Estravel, Femogen, Femogex, Gynogen L.A., Gynokadin, Lastrogen, Menaval, Merimono, Neofollin, Nuvelle, Oestrogynal, Ostrin Depo, Pelanin, Pharlon, Postoval, Primogyna, Primogyn, Primogyn Depot, Progynon, Progynon Depot, Progynova, Repestrogen, Repo-Estra, Reposo-E, Retestrin, Ronfase, Span-Est, Testaval, and Valergen, among others.
Availability
Oral estradiol valerate is used primarily in
Research
See also
- Estradiol valerate/hydroxyprogesterone caproate
- Estradiol valerate/norethisterone enantate
- Estradiol valerate/prasterone enanthate
- Estradiol valerate/testosterone enanthate
References
- ^ a b c "Estradiol and dienogest Advanced Patient Information". Drugs.com.
- ISBN 978-3-11-085727-6. Retrieved 20 May 2012.
- FDA. Retrieved 22 October 2023.
- ^ PMID 7169965.
- ^ S2CID 24616324.
- ^ PMID 8013217.
- ^ PMID 23375353.
- ISBN 978-0-323-03309-1.
- ^ PMID 22257576.
- ^ PMID 7389356.
- ^ OCLC 35483492.
- ^ ISBN 978-3-642-96158-8.
- ^ a b c d e f g h "DELESTROGEN (estradiol valerate injection, USP)" (PDF). Par Pharmaceutical, Inc. U.S. Food and Drug Administration. November 2017.
- ^ a b c d e "PROGYNOVA Product Information" (PDF). Bayer group, Germany. 20 October 2016. Archived from the original (PDF) on 16 September 2018.
- ^ ISBN 978-0-19-975569-1.
- ^ ISBN 978-3-642-60107-1.
Natural estrogens considered here include: [...] Esters of 17β-estradiol, such as estradiol valerate, estradiol benzoate and estradiol cypionate. Esterification aims at either better absorption after oral administration or a sustained release from the depot after intramuscular administration. During absorption, the esters are cleaved by endogenous esterases and the pharmacologically active 17β-estradiol is released; therefore, the esters are considered as natural estrogens.
- ^ PMID 17627398.
- ^ ISBN 978-93-5090-575-3.
- ^ ISBN 978-3-13-179525-0.
- ^ ISBN 978-0-8155-1856-3.
- ^ a b Duetsch LL (1969). Research and development, market power, and patent policy in ethical drugs. University of Wisconsin--Madison. p. 95.
1954. Delestrogen. Estradiol valerate.
- ^ ISBN 978-0-7216-3206-3. Retrieved 20 May 2012.
- ^ ISBN 978-1-4757-2085-3.
- ^ ISBN 978-3-88763-075-1. Retrieved 20 May 2012.
- ^ "Generic Delestrogen Availability". Drugs.com.
- ^ PMID 21151673.
- ^ PMID 12290848.
- ^ S2CID 3415547.
- ^ PMID 28159148.
- ^ S2CID 26979177.
- ^ ISBN 978-1-317-51460-2.
- ISBN 978-1-56639-852-7.
- PMID 27889048.
- S2CID 3363705.
- S2CID 74365268.
- ISBN 978-0-88416-175-2.
Intramuscular: For replacement therapy, (Estradiol, Estradiol Benzoate) 0.5 to 1.5 mg two or three times weekly; (Estradiol Cypionate) 1 to 5 mg weekly for two or three weeks; (Estradiol Dipropionate) 1 to 5 mg every one to two weeks; (Estradiol Valerate) 10 to 40 mg every one to four weeks.
- ISBN 978-94-009-8195-9.
- ^ Piersol G (1975). The Cyclopedia of Medicine, Surgery, Specialties. F. A. Davis Company.
- ^ a b c d e f g "Drugs@FDA: FDA Approved Drug Products". United States Food and Drug Administration. Retrieved 16 November 2016.
- ISBN 978-3-7692-2114-5.
- ISBN 978-0-7817-1750-2.
- ^ PMID 10098476.
Oestradiol valerate and oestradiol in a micronized form are the most widely used oestrogen per os for steroid substitution therapy. Our regimen, as of most other groups [...] is oestradiol valerate (Progynova; Schering, Berlin, Germany) given in various concentrations throughout the cycle [...]. According to Norfolk's protocol, 2 mg of micronized oestradiol valerate are given on cycle days 1–5. [...] In tablet form, micronized oestradiol valerate is also efficiently absorbed [...]
- ^ ISBN 978-94-009-4145-8.
- ^ "Drugs@FDA: FDA Approved Drug Products". United States Food and Drug Administration. Retrieved 26 July 2018.
- ISBN 978-0-08-055309-2.
- ISBN 978-3-319-52210-4.
- ISBN 978-0-7817-1750-2.
- ISBN 978-3-13-179525-0.
- ISBN 978-3-7692-2114-5.
- ISBN 978-81-250-0910-8.
- ISSN 0095-9561.
- ^ "AERODIOL (Oestradiol hemihydrate 150 micrograms/actuation)" (PDF). Servier Laboratories (Aust) Pty Ltd.
- ^ "Estradiol". Drugs.com.
- PMID 18382901.
300 μg 17β-estradiol (Aerodiol®; Servier, Chambrayles-Tours, France) was administered via the nasal route by a gynecologist. This product is unavailable after March 31, 2007 because its manufacturing and marketing are being discontinued.
- ISBN 978-1-4612-4330-4.
- ^ Hospital Formulary and Compendium of Useful Information. University of California Press. 1952. pp. 49–. GGKEY:2UAAZRZ5LN0.
- ISBN 978-3-662-08110-5.
- ^ PMID 2215269.
- ISBN 978-0-203-48612-2.
- ISBN 978-3-901299-34-6.
- ISBN 978-94-011-6167-1.
- ^ S2CID 37001563.
- ISBN 978-3-11-020864-1.
Dosierungsbeispiele bei Mammahypoplasie und Infantilismus [...] Parenteral 1. 40 mg Estradiolvalerat (Estradiol-Depot 10 mg JENAPHARM) und 250 mg Hydroxyprogesteroncaproat (Progesteron-Depot JENAPHARM, Proluton Depot) i. m. einmal wöchentlich über 15–20 Wochen lang. 2. 20–40 mg Estradiolvalerat (Estradiol-Depot 10 mg JENAPHARM) i. m. in der ersten und zweiten Woche. 40 mg Estradiolvalerat (Estradiol-Depot 10 mg JENAPHARM) und 250 mg Hydroxyprogesteroncaproat (Progesteron-Depot JENAPHARM, Proluton Depot) i. m. in der dritten und vierten Woche. Therapiedauer 4–5 Monate. Evtl. Abstand zwischen 2 Injektionen auf 2 Wochen erweitern (Abb. 6.2).
- ^ S2CID 260169203.
- PMID 6014870.
- ^ S2CID 70908169.
- ISSN 0039-2073.
- ^ PMID 1202923.
- PMID 968923.
- PMID 4142204. Archived from the originalon 23 November 2018. Retrieved 11 June 2019.
- S2CID 39028149.
- ^ Benjamin H, Lal GB, Green R, Masters RE (1966). The Transsexual Phenomenon. Ace Publishing Company. p. 107.
In my own practice, Squibb's Delestrogen for intramuscular injections was employed with much satisfaction and positive results. This is a slowly absorbing, well-tolerated, potent preparation (chemically, Estradiol Valerate), and was applied in doses of 20 to 60 mg. (½ to 1 ½ cc.). Usually 30 to 60 mg. of Delalutin (Squibb) was added, an equally potent progesterone. This combination was given once a week or once in two to three weeks, according to the response as measured by the patient's emotional balance and physical feminization symptoms. Generally I found that dosage seems less important than length and regularity of administration.
- ISSN 0022-4499.
Estrogen treatment—as already indicated—helps greatly but does not cure. I have employed either Squibb's Delestrogen, a slowly absorbing, highly potent preparation which is, chemically, estradiol valerate (40 mg. to 1 cc); or the still more potent Delestrec, which is estradiol undecylate (100 mg. to 1 cc). This preparation, however, is not yet on the market in this country, though it is widely used in Europe. In the majority of cases, I used from 30 to 100 mg. weekly, or every two to three weeks, by intramuscular injection.
- PMID 11741520.
- PMID 559617.
- ISBN 978-3-642-75101-1.
- ISBN 978-3-11-024568-4.
- ISBN 978-3-662-00942-0.
- ISBN 978-94-009-8195-9.
- ISBN 978-3-11-150424-7.
- PMID 779393.
- ISSN 0001-6349.
- ISSN 0172-777X.
- PMID 29603164.
- PMID 29756046.
- PMID 15432047.
- PMID 14902290.
- ISSN 0001-6349.
There is no doubt that the conversion of the endometrium with injections of both synthetic and native estrogenic hormone preparations succeeds, but the opinion whether native, orally administered preparations can produce a proliferation mucosa changes with different authors. PEDERSEN-BJERGAARD (1939) was able to show that 90% of the folliculin taken up in the blood of the vena portae is inactivated in the liver. Neither KAUFMANN (1933, 1935), RAUSCHER (1939, 1942) nor HERRNBERGER (1941) succeeded in bringing a castration endometrium into proliferation using large doses of orally administered preparations of estrone or estradiol. Other results are reported by NEUSTAEDTER (1939), LAUTERWEIN (1940) and FERIN (1941); they succeeded in converting an atrophic castration endometrium into an unambiguous proliferation mucosa with 120–300 oestradiol or with 380 oestrone.
- ISBN 978-3-642-57636-2.
- ^ Martinez-Manautou J, Rudel HW (1966). "Antiovulatory Activity of Several Synthetic and Natural Estrogens". In Robert Benjamin Greenblatt (ed.). Ovulation: Stimulation, Suppression, and Detection. Lippincott. pp. 243–253.
- ISBN 978-3-642-49506-9.
- PMID 13370006.
- S2CID 31125733.
- PMID 19524378.
- S2CID 20631200.
- PMID 15288212.
- ^ PMID 22244780.
- PMID 2170823.
- PMID 3817605.
- PMID 3173937.
- S2CID 23760705.
- PMID 22468839.
- PMID 15994752.
- S2CID 21521025.
- S2CID 26054257.
- ^ "Progynova 1mg (SPC) | Drugs.com". Retrieved 6 September 2012.
- ^ ISBN 978-94-010-8339-3.
- ^ S2CID 10159196.
- ^ S2CID 260353858.
- PMID 20004267.
- S2CID 23550553.
- ^ S2CID 20513936.
- ISSN 1179-1527.
- ^ S2CID 21953983.
- ^ PMID 2207608.
- PMID 30671393.
When we prescribed estradiol, we preferred sublingual estradiol valerate instead of the oral form for feminizing HT since prior researchers have reported the effectiveness of sublingual administration in maintaining high blood estradiol concentration and low E1/E2 ratio [13].
- PMID 10585176.
- ISBN 978-81-317-0031-0. Retrieved 20 May 2012.
- ^ PMID 6220949.
- ^ PMID 2987096.
- ^ a b c Göretzlehner G, Ackermann W, Angelow K, Bergmann G, Bieck E, Golbs S, Kliem O (2002). "Pharmakokinetik von Estron, Estradiol, FSH, LH und Prolaktin nach intramuskulärer Applikation von 5 mg Estradiolvalerat" [Pharmacokinetics of estradiol valerate in postmenopausal women after intramuscular administration]. Journal für Menopause. 9 (2): 51–55.
- ^ a b c del Cisne Valle Alvarez D (11 May 2011). Efecto de una Dosis de 50 mg de Enantato de Noretisterona y 5 mg de Valerato de Estradiol en los Niveles de Testosterona Total en Hombres Mexicanos Sanos [Effect of a Dose of 50 mg of Norethisterone Enanthate and 5 mg of Estradiol Valerate on Total Testosterone Levels in Healthy Mexican Men] (MSc). National Polytechnic Institute of Mexico.
- ^ PMID 1231448.
- ^ ISBN 978-3137008019.
- ^ PMID 7402086.
- ^ OCLC 632312599.
- ^ PMID 1150068.
- ^ PMID 3826177.
- ^ PMID 8013219.
- PMID 28078219.
- PMID 22078184.
- ^ "Estradiol valerate | C23H32O3 | ChemSpider". www.chemspider.com.
- ^ "Esters of unsaturated polyhydroxy estrane".
- PMID 15193460.
- S2CID 20753905.
- S2CID 29364660.
Um die "Menarche" sollte eine verstärkte Substitutionstherapie (20 Tage lang tgl. 0,1 mg Follikelhormon per os oder einmalig Progynon-Depot (10 mg i.m.), [...]
- ^ ISBN 978-3-83742-043-2.
With Progynon Depot-10, an oily solution of 10 mg estradiol valerate, an injection preparation had been available since 1953 and since 1966 coated tablets with estradiol valerate for oral therapy. The first Schering preparation containing micronized estradiol was marketed in 1968 as Progynova 21 (2 mg) and Progynova 21 mite (1 mg).
- S2CID 20357182.
NEUE SPEZIALITATEN [...] Progynova. 1 Dragee enthält 2 mg Oestradiolvalerinat (Klimakterium). Hersteller: Schering AG, Berlin 65.
- PMID 5593020.
- PMID 5728263.
- PMID 5045321.
- ISSN 0301-2492.
- ^ "Drugs@FDA: FDA-Approved Drugs". www.accessdata.fda.gov.
- S2CID 25856898.
- S2CID 72668930.
- ISSN 0002-9955.
Progynon-B, Schering Corporation: This is crystalline hydroxyestrin benzoate obtained by hydrogenation of theelin and subsequent conversion to the benzoate. [...] Progynon-B is marketed in ampules containing 1 cc. of a sesame oil solution of hydroxyestrin benzoate of either 2,500, 5,000, 10,000 or 50,000 international units.
- ISBN 978-0-8155-1144-1. Retrieved 20 May 2012.
- ^ a b "Estradiol". Drugs.com.
- ISBN 978-0-470-18024-2.
- ISSN 1801-2434.
- ^ "NEOFOLLIN Injekční roztok (Estradioli valeras)" (PDF).
- ISBN 978-0-387-94739-6.
- S2CID 2586346.
- PMID 5088730.
Further reading
- Vermeulen A (1975). "Longacting steroid preparations". Acta Clinica Belgica. 30 (1): 48–55. PMID 1231448.
- Düsterberg B, Nishino Y (December 1982). "Pharmacokinetic and pharmacological features of oestradiol valerate". Maturitas. 4 (4): 315–324. PMID 7169965.
- Sang GW (April 1994). "Pharmacodynamic effects of once-a-month combined injectable contraceptives". Contraception. 49 (4): 361–385. PMID 8013220.
- O'Connell MB (September 1995). "Pharmacokinetic and pharmacologic variation between different estrogen products". Journal of Clinical Pharmacology. 35 (9S): 18S–24S. S2CID 10159196.
- Kuhl H (August 2005). "Pharmacology of estrogens and progestogens: influence of different routes of administration". Climacteric. 8 (Suppl 1): 3–63. S2CID 24616324.
- Stanczyk FZ, Archer DF, Bhavnani BR (June 2013). "Ethinyl estradiol and 17β-estradiol in combined oral contraceptives: pharmacokinetics, pharmacodynamics and risk assessment". Contraception. 87 (6): 706–727. PMID 23375353.


![Baseline-adjusted estradiol levels after a single oral dose of 1.5 mg micronized estradiol or 2.0 mg estradiol valerate in postmenopausal women.[113][112] Source was Timmer & Geurts (1999).[112]](http://upload.wikimedia.org/wikipedia/commons/thumb/0/0d/Estradiol_levels_with_oral_micronized_estradiol_and_oral_estradiol_valerate_in_postmenopausal_women.png/400px-Estradiol_levels_with_oral_micronized_estradiol_and_oral_estradiol_valerate_in_postmenopausal_women.png)
![Estradiol levels after a single oral dose of 2 mg micronized estradiol or 2 mg estradiol valerate and with continuous oral administration of 2 mg/day micronized estradiol or 2 mg/day estradiol valerate (at steady state) in postmenopausal women.[109] Source was Wiegratz et al. (2001).[109]](http://upload.wikimedia.org/wikipedia/commons/thumb/2/2c/Estradiol_levels_after_a_single_dose_and_with_continuous_administration_of_oral_estradiol_or_oral_estradiol_valerate_in_women.png/377px-Estradiol_levels_after_a_single_dose_and_with_continuous_administration_of_oral_estradiol_or_oral_estradiol_valerate_in_women.png)
![Estrogen levels after a single intramuscular injection of 10 mg estradiol valerate in oil in 24 postmenopausal women.[9] Determinations were made for both Progynon Depot 10 and Estradiol Depot 10, for a total of 48 measurements per point.[9] Assays were performed using GC/MS-NCI/SIM.[9] Source was Schug et al. (2012).[9]](http://upload.wikimedia.org/wikipedia/commons/thumb/d/d0/Estrogen_levels_after_a_single_intramuscular_injection_of_10_mg_estradiol_valerate_in_postmenopausal_women.png/300px-Estrogen_levels_after_a_single_intramuscular_injection_of_10_mg_estradiol_valerate_in_postmenopausal_women.png)
![Hormone levels after a single intramuscular injection of 5 mg estradiol valerate in oil in 17 postmenopausal women.[121] Assays were performed using EIA.[121] Estrone levels were likely overestimated, possibly due to cross reactivity of the assay with estrogen conjugates.[9] Source was Göretzlehner et al. (2002).[121]](http://upload.wikimedia.org/wikipedia/commons/thumb/0/0c/Hormone_levels_after_a_single_intramuscular_injection_of_5_mg_estradiol_valerate_in_postmenopausal_women.png/300px-Hormone_levels_after_a_single_intramuscular_injection_of_5_mg_estradiol_valerate_in_postmenopausal_women.png)
![Hormone levels after a single intramuscular injection of estradiol valerate/norethisterone enanthate (5 mg/50 mg) (Mesigyna) in healthy young men.[122] Testosterone decreased from ~503 ng/dL to ~30 ng/dL (–94%).[122] Source was Valle Alvarez (2011).[122]](http://upload.wikimedia.org/wikipedia/commons/thumb/4/4a/Hormone_levels_in_men_with_a_single_intramuscular_injection_of_5_mg_estradiol_valerate_and_50_mg_norethisterone_enanthate_in_oil.png/300px-Hormone_levels_in_men_with_a_single_intramuscular_injection_of_5_mg_estradiol_valerate_and_50_mg_norethisterone_enanthate_in_oil.png)
![Estradiol levels after single intramuscular injections of 5 mg of different estradiol esters in oil in about 10 premenopausal women each.[10] Assays were performed using RIA with CS.[10] Source was Oriowo et al. (1980).[10]](http://upload.wikimedia.org/wikipedia/commons/thumb/3/33/Estradiol_levels_after_a_single_5_mg_intramuscular_injection_of_estradiol_esters.png/300px-Estradiol_levels_after_a_single_5_mg_intramuscular_injection_of_estradiol_esters.png)
![Estradiol levels after a single intramuscular injection of 10 mg estradiol valerate or 100 mg estradiol undecylate in oil both in 4 individuals each.[123] Subject characteristics and assay method were not described.[123] Source was Vermeulen (1975).[123]](http://upload.wikimedia.org/wikipedia/commons/thumb/6/65/Estradiol_levels_after_a_single_intramuscular_injection_of_10_mg_estradiol_valerate_and_100_mg_estradiol_undecylate.png/300px-Estradiol_levels_after_a_single_intramuscular_injection_of_10_mg_estradiol_valerate_and_100_mg_estradiol_undecylate.png)
![Estradiol and DHEA levels after a single intramuscular injection of Gynodian Depot (4 mg estradiol valerate, 200 mg prasterone enanthate in oil) or Primogyn Depot (10 mg estradiol valerate in oil) in women.[124][119][125] Assays were performed using RIA.[119][125] Sources were Düsterberg & Wendt (1983) and Rauramo et al. (1980).[124][119][125]](http://upload.wikimedia.org/wikipedia/commons/thumb/d/dd/Estradiol_and_dehydroepiandrosterone_levels_after_a_single_intramuscular_injection_of_Gynodian_Depot_in_women.png/300px-Estradiol_and_dehydroepiandrosterone_levels_after_a_single_intramuscular_injection_of_Gynodian_Depot_in_women.png)
![Estradiol levels after a short intravenous infusion of 20 mg estradiol in aqueous solution or an intramuscular injection of equimolar doses of estradiol esters in oil solution in postmenopausal women.[126][127] Assays were performed using RIA with CS.[126][127] Source was Geppert (1975).[126][127]](http://upload.wikimedia.org/wikipedia/commons/thumb/b/b1/Estradiol_levels_after_injections_of_estradiol%2C_estradiol_benzoate%2C_estradiol_valerate%2C_and_estradiol_undecylate_in_women.png/300px-Estradiol_levels_after_injections_of_estradiol%2C_estradiol_benzoate%2C_estradiol_valerate%2C_and_estradiol_undecylate_in_women.png)
![Estradiol levels after an intramuscular injection of 10 mg estradiol valerate in oil, Climacteron (150 mg testosterone enanthate, 1 mg estradiol benzoate, 7.5 mg estradiol dienanthate in oil), and control group in 20, 11, and 11 ovariectomized women, respectively.[128] Assays were performed using RIA.[128] Source was Sherwin et al. (1987).[128]](http://upload.wikimedia.org/wikipedia/commons/thumb/e/e5/Estradiol_levels_after_an_intramuscular_injection_of_Climacteron_or_estradiol_valerate_in_ovariectomized_women.png/300px-Estradiol_levels_after_an_intramuscular_injection_of_Climacteron_or_estradiol_valerate_in_ovariectomized_women.png)
![Simplified curves of estradiol levels after injection of different estradiol esters in women.[129] Source was Garza-Flores (1994).[129]](http://upload.wikimedia.org/wikipedia/commons/thumb/8/8c/Idealized_curves_of_estradiol_levels_after_injection_of_different_estradiol_esters_in_women.png/293px-Idealized_curves_of_estradiol_levels_after_injection_of_different_estradiol_esters_in_women.png)