Oncovirus

An oncovirus or oncogenic virus is a virus that can cause cancer.[4] This term originated from studies of acutely transforming retroviruses in the 1950–60s,[5] when the term "oncornaviruses" was used to denote their RNA virus origin.[6] With the letters "RNA" removed, it now refers to any virus with a DNA or RNA genome causing cancer and is synonymous with "tumor virus" or "cancer virus". The vast majority of human and animal viruses do not cause cancer, probably because of longstanding co-evolution between the virus and its host. Oncoviruses have been important not only in epidemiology, but also in investigations of cell cycle control mechanisms such as the retinoblastoma protein.
The World Health Organization's International Agency for Research on Cancer estimated that in 2002, infection caused 17.8% of human cancers, with 11.9% caused by one of seven viruses.[7] A 2020 study of 2,658 samples from 38 different types of cancer found that 16% were associated with a virus.[8] These cancers might be easily prevented through vaccination (e.g., papillomavirus vaccines), diagnosed with simple blood tests, and treated with less-toxic antiviral compounds.
Causality
Generally, tumor viruses cause little or no disease after infection in their hosts, or cause non-
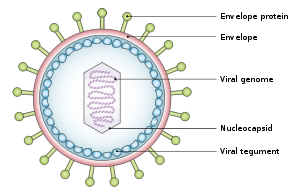
Tumor viruses come in a variety of forms: Viruses with a
Oncogenic viral mechanism

A direct oncogenic viral mechanism
Indirect viral oncogenicity involves chronic nonspecific inflammation occurring over decades of infection, as is the case for HCV-induced liver cancer. These two mechanisms differ in their biology and epidemiology: direct tumor viruses must have at least one virus copy in every tumor cell expressing at least one protein or RNA that is causing the cell to become cancerous. Because foreign virus antigens are expressed in these tumors, persons who are immunosuppressed such as AIDS or transplant patients are at higher risk for these types of cancers.[citation needed]
Chronic indirect tumor viruses, on the other hand, can be lost (at least theoretically) from a mature tumor that has accumulated sufficient mutations and growth conditions (hyperplasia) from the chronic inflammation of viral infection. In this latter case, it is controversial but at least theoretically possible that an indirect tumor virus could undergo "hit-and-run" and so the virus would be lost from the clinically diagnosed tumor. In practical terms, this is an uncommon occurrence if it does occur.[citation needed]
DNA oncoviruses

DNA oncoviruses typically impair two families of tumor suppressor proteins: tumor proteins p53 and the retinoblastoma proteins (Rb). It is evolutionarily advantageous for viruses to inactivate p53 because p53 can trigger cell cycle arrest or apoptosis in infected cells when the virus attempts to replicate its DNA.[13] Similarly, Rb proteins regulate many essential cell functions, including but not limited to a crucial cell cycle checkpoint, making them a target for viruses attempting to interrupt regular cell function.[14]
While several DNA oncoviruses have been discovered, three have been studied extensively.
Integration of viral DNA
DNA oncoviruses transform infected cells by integrating their DNA into the host cell's genome.[18] The DNA is believed to be inserted during transcription or replication, when the two annealed strands are separated.[18] This event is relatively rare and generally unpredictable; there seems to be no deterministic predictor of the site of integration.[18] After integration, the host's cell cycle loses regulation from Rb and p53, and the cell begins cloning to form a tumor.[19]
G1/S Checkpoint
Rb and p53 regulate the transition between
Inactivation of p53
Viruses employ various methods of inactivating p53. The
Inactivation of Rb
Rb is inactivated (thereby allowing the G1/S transition to progress unimpeded) by different but analogous viral oncoproteins. The adenovirus early region 1A (E1A) is an oncoprotein which binds to Rb and can stimulate transcription and transform cells.[13] SV40 uses the same protein for inactivating Rb, LT, to inactivate p53.[21] HPV contains a protein, E7, which can bind to Rb in much the same way.[24] Rb can be inactivated by phosphorylation, or by being bound to a viral oncoprotein, or by mutations—mutations which prevent oncoprotein binding are also associated with cancer.[22]
Variations
DNA oncoviruses typically cause cancer by inactivating p53 and Rb, thereby allowing unregulated cell division and creating tumors. There may be many different mechanisms which have evolved separately; in addition to those described above, for example, the
SV40 has been well studied and does not cause cancer in humans, but a recently discovered analogue called
RNA oncoviruses
In the 1960s, the replication process of RNA virus was believed to be similar to other single-stranded RNA. Single-stranded RNA replication involves RNA-dependent RNA synthesis which meant that virus-coding enzymes would make partial double-stranded RNA. This belief was shown to be incorrect because there were no double-stranded RNA found in the retrovirus cell. In 1964, Howard Temin proposed a provirus hypothesis, but shortly after reverse transcription in the retrovirus genome was discovered.
Description of virus
All retroviruses have three major coding domains; gag, pol and env. In the gag region of the virus, the synthesis of the internal virion proteins are maintained which make up the matrix, capsid and nucleocapsid proteins. In pol, the information for the reverse transcription and integration enzymes are stored. In env, it is derived from the surface and transmembrane for the viral envelope protein. There is a fourth coding domain which is smaller, but exists in all retroviruses. Pol is the domain that encodes the virion protease.
Retrovirus enters host cell
The retrovirus begins the journey into a host cell by attaching a surface glycoprotein to the cell's plasma membrane receptor. Once inside the cell, the retrovirus goes through reverse transcription in the cytoplasm and generates a double-stranded DNA copy of the RNA genome. Reverse transcription also produces identical structures known as long terminal repeats (LTRs). Long terminal repeats are at the ends of the DNA strands and regulates viral gene expression. The viral DNA is then translocated into the nucleus where one strand of the retroviral genome is put into the chromosomal DNA by the help of the virion integrase. At this point the retrovirus is referred to as provirus. Once in the chromosomal DNA, the provirus is transcribed by the cellular RNA polymerase II. The transcription leads to the splicing and full-length mRNAs and full-length progeny virion RNA. The virion protein and progeny RNA assemble in the cytoplasm and leave the cell, whereas the other copies send translated viral messages in the cytoplasm.
Classification

DNA viruses
- penis cancer, anal cancer, and HPV-positive oropharyngeal cancers.[7][27][28][29][30][31][32] There are nearly 200 distinct human papillomaviruses (HPVs),[30] and many HPV types are carcinogenic.[7][27]
- Hepatocarcinoma[33]
- Epstein–Barr virus (EBV or HHV-4) is associated with four types of cancers
- Human cytomegalovirus (CMV or HHV-5) is associated with mucoepidermoid carcinoma and possibly other malignancies.[34]
- Kaposi's sarcoma-associated herpesvirus (KSHV or HHV-8) is associated with Kaposi's sarcoma, a type of skin cancer.[35]
- Merkel cell carcinoma[25]
RNA viruses
Not all oncoviruses are
Overview table
| Virus | Percent of cancers[7] | Associated cancer types |
|---|---|---|
| Hepatitis B virus (HBV) | Hepatocarcinoma[33]
| |
| Hepatitis C virus (HCV) | HCV is a known carcinogen, causing hepatocarcinoma[36]
| |
Human T-lymphotropic virus (HTLV) |
0.03 | Adult T-cell leukemia[37]
|
Human papillomaviruses (HPV) |
5.2 | HPV types 16 and 18 are associated with cancers of cervix,[7][27][28][30][31] anus,[7][29][30] penis,[7][29][30] vulva,[7][29][30] vagina,[7][29][30] and HPV-positive oropharyngeal cancers.[7][29][32] According to statistics in the United States, females are more impacted by HPV-associated cancers (83%) than males (74%).[38] |
| Kaposi's sarcoma-associated herpesvirus (HHV-8) | 0.9 | Castleman's disease and primary effusion lymphoma
|
| Merkel cell polyomavirus (MCV) | NA | Merkel cell carcinoma
|
| Epstein–Barr virus (EBV) | NA | post-transplant lymphoproliferative disease, nasopharyngeal carcinoma[39] and a subtype of stomach cancer.[40]
|
Estimated percent of new cancers attributable to the virus worldwide in 2002.[7] NA indicates not available. The association of other viruses with human cancer is continually under research.
Main viruses associated with human cancer

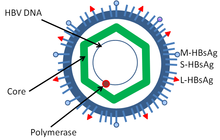
The main viruses associated with human cancers are the
Hepatitis viruses, including hepatitis B and hepatitis C, can induce a chronic viral infection that leads to liver cancer in 0.47% of hepatitis B patients per year (especially in Asia, less so in North America), and in 1.4% of hepatitis C carriers per year. Liver cirrhosis, whether from chronic viral hepatitis infection or alcoholism, is associated with the development of liver cancer, and the combination of cirrhosis and viral hepatitis presents the highest risk of liver cancer development. Worldwide, liver cancer is one of the most common, and most deadly, cancers due to a huge burden of viral hepatitis transmission and disease.[citation needed]
Through advances in cancer research, vaccines designed to prevent cancer have been created. The hepatitis B vaccine is the first vaccine that has been established to prevent cancer (
History
The history of cancer virus discovery is intertwined with the
History of non-human oncoviruses

The theory that cancer could be caused by a virus began with the experiments of
By the early 1950s, it was known that viruses could remove and incorporate genes and genetic material in cells. It was suggested that such types of viruses could cause cancer by introducing new genes into the genome. Genetic analysis of mice infected with
In 1961 Eddy discovered the simian vacuolating virus 40 (
History of human oncoviruses
In 1964
In 1980 the first human retrovirus, Human T-lymphotropic virus 1 (HTLV-I), was discovered by Bernard Poiesz and Robert Gallo at NIH,[61][62] and independently by Mitsuaki Yoshida and coworkers in Japan.[63] But it was not certain whether HTLV-I promoted leukemia. In 1981 Yorio Hinuma and his colleagues at Kyoto University reported visualization of retroviral particles produced by a leukemia cell line derived from patients with Adult T-cell leukemia/lymphoma. This virus turned out to be HTLV-1 and the research established the causal role of the HTLV-1 virus to ATL.[42]
Between 1984 and 1986
In 1994 Patrick S. Moore and Yuan Chang at Columbia University), working together with Ethel Cesarman,[66][67] isolated Kaposi's sarcoma-associated herpesvirus (KSHV or HHV8) using representational difference analysis. This search was prompted by work from Valerie Beral and colleagues who inferred from the epidemic of Kaposi's sarcoma among patients with AIDS that this cancer must be caused by another infectious agent besides HIV, and that this was likely to be a second virus.[68] Subsequent studies revealed that KSHV is the "KS agent" and is responsible for the epidemiologic patterns of KS and related cancers.[69]
In 2008 Yuan Chang and Patrick S. Moore developed a new method to identify cancer viruses based on computer subtraction of human sequences from a tumor transcriptome, called digital transcriptome subtraction (DTS).[70] DTS was used to isolate DNA fragments of Merkel cell polyomavirus from a Merkel cell carcinoma and it is now believed that this virus causes 70–80% of these cancers.[25]
See also
- Infectious causes of cancer
- Carcinogen
- Oncogenic
- Oncogene
- Adult T-cell leukemia/lymphoma
- Cancer bacteria
- Oncolytic virus, a virus that infects and kills cancer cells
- Gag-onc fusion protein
- List of infectious diseases
References
- PMID 25848462.
- PMID 23871966.
- ^ ACS Medical and Editorial Content Team (30 July 2020). "Viruses that can lead to cancer". www.cancer.org. American Cancer Society. Retrieved 27 February 2021.
- PMID 28893931.
- PMID 13779782.
- ^
ISBN 9781468410129. Retrieved 20 October 2016.
Viruses known as oncogenic RNA viruses or oncornaviruses have been described in a wide variety of vertebrate species [...] there have been very few, if any, completely convincing examples of isolation of human oncornaviruses.
- ^ S2CID 10042384.
- PMID 32025001.
- PMID 8665474.
- PMID 14283879.
- ISBN 978-0-19-510401-1.[page needed]
- PMID 26545119.
- ^ PMID 11753670.
- PMID 16936748.
- ISBN 978-1-904455-99-8.
- ^ PMID 16963733.
- PMID 11896579.
- ^ PMID 6253158.
- PMID 9039259.
- ^ ISBN 978-1-904455-99-8.
- ^ PMID 19081592.
- ^ PMID 19150725.
- S2CID 27437768.
- PMID 2537532.
- ^ PMID 18202256.
- S2CID 7268482.
- ^ S2CID 1451343.
- ^ S2CID 20196938.
- ^ S2CID 13768854.
- ^ S2CID 24219797.
- ^ a b "Human papillomavirus (HPV) and cervical cancer". WHO. June 2016. Archived from the original on 5 August 2016.
- ^ )
- ^ PMID 24876747.
- S2CID 41446671.
- S2CID 29977325.
- ^ NTP (National Toxicology Program) (2016). Report on Carcinogens (Fourteenth ed.). Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service. Retrieved 8 January 2017.
- PMID 17726237.
- ^ How Many Cancers Are Linked with HPV Each Year? CDC.gov
- PMID 17322915.
- PMID 24914366.
- PMID 1659743.
- ^ PMID 18829521.
- ^ Ellermann V.; Bang O. (1908). "Experimentelle Leukämie bei Hühnern". Zentralbl. Bakteriol. Parasitenkd. Infectionskr. Hyg. Abt. Orig. 46: 595–609.
- ^ Ellerman C, Bang O (1908). "Experimentelle Leukämie bei Hühnern". Zentralbl. Bakteriol. Parasitenkd. Infectionskr. Hyg. Abt. Orig. 46: 595–609.
- PMID 19867354.
- ^ PMID 19867421.
- PMID 15756727.
- ^ PMID 13416470.
- S2CID 10527142.
- PMID 2842714.
- S2CID 34223353.
- PMID 13549981.
- PMID 13819251.
- ISBN 978-1118704639.
- ^ Tontonoz M (13 March 2019). "From Controversy to Consensus: How Charlotte Friend Changed Cancer Biology". Memorial Sloan Kettering Cancer Center. Retrieved 18 April 2019.
- PMID 11720463.
- S2CID 20679358.
- PMID 14107961.
- ^ "Baruch S. Blumberg – Autobiography". Nobelprize.org. Retrieved 17 March 2010.
- S2CID 32264986.
- .
- PMID 16155599.
- PMID 6294664.
- ^ "Harald zur Hausen – Autobiography". Nobelprize.org. Retrieved 17 March 2010.
- PMID 2523562.
- PMID 18398088.
- ^ "Ethel Cesarman, M.D., Ph.D. | Weill Cornell Medical College". Med.cornell.edu. Retrieved 17 March 2010.
- S2CID 35639169.
- PMID 10749966.
- PMID 17686852.
External links
- Oncoviruses at the U.S. National Library of Medicine Medical Subject Headings (MeSH)