Alkane: Difference between revisions
HotCat |
→Further reading: Adding a new external link that talks about alkanes as an interesting chemical Tag: Reverted |
||
| Line 586: | Line 586: | ||
* [https://www2.chemistry.msu.edu/faculty/reusch/VirtTxtJml/intro1.htm Virtual Textbook of Organic Chemistry] |
* [https://www2.chemistry.msu.edu/faculty/reusch/VirtTxtJml/intro1.htm Virtual Textbook of Organic Chemistry] |
||
* [https://log-web.de/chemie/Start.htm?name=ethaneCryst&lang=en A visualization of the crystal structures of alkanes up to nonan] |
* [https://log-web.de/chemie/Start.htm?name=ethaneCryst&lang=en A visualization of the crystal structures of alkanes up to nonan] |
||
* [https://www.wolframscience.com/nks/notes-12-11--interesting-chemicals/ Interesting Chemicals - Alkanes (paraffins)] |
|||
{{Hydrocarbons}} |
{{Hydrocarbons}} |
||
Revision as of 20:25, 9 March 2021

In
The International Union of Pure and Applied Chemistry (IUPAC) defines alkanes as "acyclic branched or unbranched hydrocarbons having the general formula Template:CarbonnTemplate:Hydrogen2n+2, and therefore consisting entirely of hydrogen atoms and saturated carbon atoms". However, some sources use the term to denote any saturated hydrocarbon, including those that are either monocyclic (i.e. the cycloalkanes) or polycyclic,[2] despite their having a distinct general formula (i.e. cycloalkanes are CnH2n).
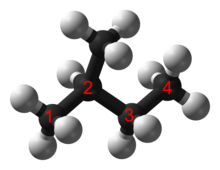
In an alkane, each carbon atom is sp3-hybridized with 4 sigma bonds (either C–C or C–H), and each hydrogen atom is joined to one of the carbon atoms (in a C–H bond). The longest series of linked carbon atoms in a molecule is known as its carbon skeleton or carbon backbone. The number of carbon atoms may be considered as the size of the alkane.
One group of the
Methane is produced by methanogenic bacteria and some long-chain alkanes function as pheromones in certain animal species or as protective waxes in plants and fungi. Nevertheless, most alkanes do not have much biological activity. They can be viewed as molecular trees upon which can be hung the more active/reactive functional groups of biological molecules.
The alkanes have two main commercial sources: petroleum (crude oil)[3] and natural gas.
An
Structure and classification
Saturated hydrocarbons are hydrocarbons having only single covalent bonds between their carbons. They can be:
- linear (general formula C
nH
2n+2) wherein the carbon atoms are joined in a snake-like structure; - branched (general formula C
nH
2n+2, n > 3) wherein the carbon backbone splits off in one or more directions; the simplest branched alkane is 2-methyl-propane, or more commonly known as isobutane, which has three carbon atoms as backbone, and another carbon (a methyl group) linked to the second atom of the backbone, so as to form a "T" shaped molecule. The 'iso-' prefix comes from the fact that a hydrogen atom misses from the central carbon atom of the isopropyl radical (most known for its isopropyl alcohol compound, or rubbing alcohol); - cyclic (general formula C
nH
2n, n > 3) wherein the carbon backbone is linked so as to form a loop.
According to the definition by
Isomerism
Bicyclo[1.1.0]butane is the only C4H6 alkane and has no alkane isomer; tetrahedrane

Alkanes with more than three carbon atoms can be arranged in various ways, forming structural isomers. The simplest isomer of an alkane is the one in which the carbon atoms are arranged in a single chain with no branches. This isomer is sometimes called the n-isomer (n for "normal", although it is not necessarily the most common). However the chain of carbon atoms may also be branched at one or more points. The number of possible isomers increases rapidly with the number of carbon atoms. For example, for acyclic alkanes:[4]
- C1: methane only
- C2: ethane only
- C3: propane only
- C4: 2 isomers: butane and isobutane
- C5: 3 isomers: pentane, isopentane, and neopentane
- C6: 5 isomers: hexane, 2-methylpentane, 3-methylpentane, 2,2-dimethylbutane, and 2,3-dimethylbutane
- C7: 9 isomers: 2,2,3-trimethylbutane
- C12: 355 isomers
- C32: 27,711,253,769 isomers
- C60: 22,158,734,535,770,411,074,184 isomers, many of which are not stable.
Branched alkanes can be
Nomenclature
The IUPAC nomenclature (systematic way of naming compounds) for alkanes is based on identifying hydrocarbon chains. Unbranched, saturated hydrocarbon chains are named systematically with a Greek numerical prefix denoting the number of carbons and the suffix "-ane".[5]
In 1866,
It is difficult or impossible to find compounds with more than one IUPAC name. This is because shorter chains attached to longer chains are prefixes and the convention includes brackets. Numbers in the name, referring to which carbon a group is attached to, should be as low as possible so that 1- is implied and usually omitted from names of organic compounds with only one side-group. Symmetric compounds will have two ways of arriving at the same name.[citation needed]
Linear alkanes
Straight-chain alkanes are sometimes indicated by the prefix "n-" or "n-"(for "normal") where a non-linear isomer exists. Although this is not strictly necessary, the usage is still common in cases where there is an important difference in properties between the straight-chain and branched-chain isomers, e.g., "n-butane" rather than simply "butane" to distinguish it from isobutane. Alternative names for this group are: linear paraffins or n-paraffins.[citation needed]
The first six members of the series (in terms of number of carbon atoms) are named as follows:
- methane
- CH4 – one carbon and 4 hydrogen
- ethane
- C2H6 – two carbon and 6 hydrogen
- propane
- C3H8 – three carbon and 8 hydrogen
- butane
- C4H10 – four carbon and 10 hydrogen
- pentane
- C5H12 – five carbon and 12 hydrogen
- hexane
- C6H14 – six carbon and 14 hydrogen
The first four names were
Branched alkanes
Simple branched alkanes often have a common name using a prefix to distinguish them from linear alkanes, for example n-pentane, isopentane, and neopentane.
IUPAC naming conventions can be used to produce a systematic name.
The key steps in the naming of more complicated branched alkanes are as follows:[10]
- Identify the longest continuous chain of carbon atoms
- Name this longest root chain using standard naming rules
- Name each side chain by changing the suffix of the name of the alkane from "-ane" to "-yl"
- Number the longest continuous chain in order to give the lowest possible numbers for the side-chains[11]
- Number and name the side chains before the name of the root chain
- If there are multiple side chains of the same type, use prefixes such as "di-" and "tri-" to indicate it as such, and number each one.
- Add side chain names in alphabetical (disregarding "di-" etc. prefixes) order in front of the name of the root chain
| Common name | n-pentane | isopentane | neopentane |
|---|---|---|---|
| IUPAC name | pentane | 2-methylbutane | 2,2-dimethylpropane |
| Structure |  |
|
Saturated cyclic hydrocarbons
Though technically distinct from the alkanes, this class of hydrocarbons is referred to by some as the "cyclic alkanes." As their description implies, they contain one or more rings.[citation needed]
Simple cycloalkanes have a prefix "cyclo-" to distinguish them from alkanes. Cycloalkanes are named as per their acyclic counterparts with respect to the number of carbon atoms in their backbones, e.g., cyclopentane (C5H10) is a cycloalkane with 5 carbon atoms just like pentane (C5H12), but they are joined up in a five-membered ring. In a similar manner, propane and cyclopropane, butane and cyclobutane, etc.[citation needed]
Substituted cycloalkanes are named similarly to substituted alkanes – the cycloalkane ring is stated, and the substituents are according to their position on the ring, with the numbering decided by the Cahn–Ingold–Prelog priority rules.[9]
Trivial/common names
The trivial (non-
It is almost certain that the term 'paraffin' stems from the petrochemical industry. Branched-chain alkanes are called isoparaffins. The use of the term "paraffin" is a general term and often does not distinguish between pure compounds and mixtures of isomers, i.e., compounds of the same chemical formula, e.g., pentane and isopentane.[citation needed]
- In IUPAC
The following trivial names are retained in the IUPAC system:
- isobutane for 2-methylpropane
- isopentane for 2-methylbutane
- neopentane for 2,2-dimethylpropane.
- Non-IUPAC
Some non-IUPAC trivial names are occasionally used:
- cetane, for hexadecane
- cerane, for hexacosane[12]
Physical properties
All alkanes are colorless.[13][14] Alkanes with the lowest molecular weights are gasses, those of intermediate molecular weight are liquids, and the heaviest are waxy solids.[citation needed]
Table of alkanes
| Alkane | Formula | Boiling point [°C] | Melting point [°C] | Density [kg/m3] (at 20 °C) | Isomers |
| Methane | CH4 | -162 | −182 | 0.656 (gas) | 1 |
| Ethane | C2H6 | −89 | −183 | 1.26 (gas) | 1 |
| Propane | C3H8 | −42 | −188 | 2.01 (gas) | 1 |
| Butane | C4H10 | 0 | −138 | 2.48 (gas) | 2 |
| Pentane | C5H12 | 36 | −130 | 626 (liquid) | 3 |
| Hexane | C6H14 | 69 | −95 | 659 (liquid) | 5 |
| Heptane | C7H16 | 98 | −91 | 684 (liquid) | 9 |
| Octane | C8H18 | 126 | −57 | 703 (liquid) | 18 |
| Nonane | C9H20 | 151 | −54 | 718 (liquid) | 35 |
| Decane | C10H22 | 174 | −30 | 730 (liquid) | 75 |
| Undecane | C11H24 | 196 | −26 | 740 (liquid) | 159 |
| Dodecane | C12H26 | 216 | −10 | 749 (liquid) | 355 |
| Tridecane | C13H28 | 235 | -5.4 | 756 (liquid) | 802 |
| Tetradecane | C14H30 | 253 | 5.9 | 763 (liquid) | 1858 |
| Pentadecane | C15H32 | 270 | 10 | 769 (liquid) | |
| Hexadecane | C16H34 | 287 | 18 | 773 (liquid) | |
| Heptadecane | C17H36 | 303 | 22 | 777 (solid) | |
| Octadecane | C18H38 | 317 | 28 | 781 (solid) | |
| Nonadecane | C19H40 | 330 | 32 | 785 (solid) | |
| Icosane | C20H42 | 343 | 37 | 789 (solid) | |
Triacontane
|
C30H62 | 450 | 66 | 810 (solid) | |
Tetracontane
|
C40H82 | 525 | 82 | 817 (solid) | |
Pentacontane
|
C50H102 | 575 | 91 | 824 (solid) | |
Hexacontane
|
C60H122 | 625 | 100 | 829 (solid) | |
Heptacontane
|
C70H142 | 653 | 109 | 869 (solid) |
Boiling point

Alkanes experience intermolecular van der Waals forces. Stronger intermolecular van der Waals forces give rise to greater boiling points of alkanes.[15]
There are two determinants for the strength of the van der Waals forces:
- the number of electrons surrounding the molecule, which increases with the alkane's molecular weight
- the surface area of the molecule
Under
A straight-chain alkane will have a boiling point higher than a branched-chain alkane due to the greater surface area in contact, thus the greater van der Waals forces, between adjacent molecules. For example, compare
On the other hand, cycloalkanes tend to have higher boiling points than their linear counterparts due to the locked conformations of the molecules, which give a plane of intermolecular contact.[16]
Melting points
The
The melting points of branched-chain alkanes can be either higher or lower than those of the corresponding straight-chain alkanes, again depending on the ability of the alkane in question to pack well in the solid phase: This is particularly true for isoalkanes (2-methyl isomers), which often have melting points higher than those of the linear analogues.[citation needed]
Conductivity and solubility
Alkanes do not conduct electricity in any way, nor are they substantially
Their solubility in nonpolar solvents is relatively high, a property that is called lipophilicity. Alkanes are, for example, miscible in all proportions among themselves.[citation needed]
The density of the alkanes usually increases with the number of carbon atoms but remains less than that of water. Hence, alkanes form the upper layer in an alkane–water mixture.[citation needed]
Molecular geometry
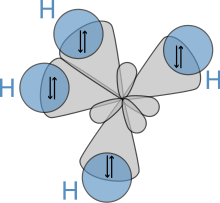
The molecular structure of the alkanes directly affects their physical and chemical characteristics. It is derived from the electron configuration of carbon, which has four valence electrons. The carbon atoms in alkanes are always sp3-hybridized, that is to say that the valence electrons are said to be in four equivalent orbitals derived from the combination of the 2s orbital and the three 2p orbitals. These orbitals, which have identical energies, are arranged spatially in the form of a tetrahedron, the angle of cos−1(−1/3) ≈ 109.47° between them.[citation needed]
Bond lengths and bond angles
An alkane has only C–H and C–C single bonds. The former result from the overlap of an sp3 orbital of carbon with the 1s orbital of a hydrogen; the latter by the overlap of two sp3 orbitals on adjacent carbon atoms. The bond lengths amount to 1.09 × 10−10 m for a C–H bond and 1.54 × 10−10 m for a C–C bond.[citation needed]

The spatial arrangement of the bonds is similar to that of the four sp3 orbitals—they are tetrahedrally arranged, with an angle of 109.47° between them. Structural formulae that represent the bonds as being at right angles to one another, while both common and useful, do not correspond with the reality.[citation needed]
Conformation
The structural formula and the
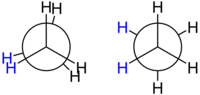
The two conformations differ in energy: the staggered conformation is 12.6 kJ/mol lower in energy (more stable) than the eclipsed conformation (the least stable).
This difference in energy between the two conformations, known as the
The case of
Spectroscopic properties
Virtually all organic compounds contain carbon–carbon, and carbon–hydrogen bonds, and so show some of the features of alkanes in their spectra. Alkanes are notable for having no other groups, and therefore for the absence of other characteristic spectroscopic features of a functional group like –OH, –CHO, –COOH etc.
Infrared spectroscopy
The carbon–hydrogen stretching mode gives a strong absorption between 2850 and 2960 cm−1, while the carbon–carbon stretching mode absorbs between 800 and 1300 cm−1. The carbon–hydrogen bending modes depend on the nature of the group: methyl groups show bands at 1450 cm−1 and 1375 cm−1, while methylene groups show bands at 1465 cm−1 and 1450 cm−1. Carbon chains with more than four carbon atoms show a weak absorption at around 725 cm−1.
NMR spectroscopy
The proton resonances of alkanes are usually found at
Mass spectrometry
Alkanes have a high
Chemical properties
Alkanes are only weakly reactive with most chemical compounds. The
In highly branched alkanes, the bond angle may differ significantly from the optimal value (109.5°) to accommodate bulky groups. Such distortions introduce a tension in the molecule, known as
However, in general and perhaps surprisingly, when branching is not extensive enough to make highly disfavorable 1,2- and 1,3-alkyl–alkyl steric interactions (worth ~3.1 kcal/mol and ~3.7 kcal/mol in the case of the eclipsing conformations of butane and pentane, respectively) unavoidable, the branched alkanes are actually more thermodynamically stable than their linear (or less branched) isomers. For example, the highly branched 2,2,3,3-tetramethylbutane is about 1.9 kcal/mol more stable than its linear isomer, n-octane.[19] Due to the subtlety of this effect, the exact reasons for this rule have been vigorously debated in the chemical literature and is yet unsettled. Several explanations, including stabilization of branched alkanes by electron correlation,[20] destabilization of linear alkanes by steric repulsion,[21] stabilization by neutral hyperconjugation,[22][19] and/or electrostatic effects[23] have been advanced as possibilities. The controversy is related to the question of whether the traditional explanation of hyperconjugation is the primary factor governing the stability of alkyl radicals.[24][21]
Reactions with oxygen (combustion reaction)
All alkanes react with oxygen in a combustion reaction, although they become increasingly difficult to ignite as the number of carbon atoms increases. The general equation for complete combustion is:
- CnH2n+2 + (3/2n + 1/2) O2 → (n + 1) H2O + n CO2
- or CnH2n+2 + (3n + 1/2) O2 → (n + 1) H2O + n CO2
In the absence of sufficient oxygen, carbon monoxide or even soot can be formed, as shown below:
For example, methane:
- 2 CH4 + 3 O2 → 2 CO + 4 H2O
- CH4 + 3/2 O2 → CO + 2 H2O
See the
Reactions with halogens
Alkanes react with
These reactions are an important industrial route to halogenated hydrocarbons. There are three steps:
- Initiation the halogen radicals form by homolysis. Usually, energy in the form of heat or light is required.
- Chain reaction or Propagation then takes place—the halogen radical abstracts a hydrogen from the alkane to give an alkyl radical. This reacts further.
- Chain termination where the radicals recombine.
Experiments have shown that all halogenation produces a mixture of all possible isomers, indicating that all hydrogen atoms are susceptible to reaction. The mixture produced, however, is not a statistical mixture: Secondary and tertiary hydrogen atoms are preferentially replaced due to the greater stability of secondary and tertiary free-radicals. An example can be seen in the monobromination of propane:[15]

Cracking
Cracking breaks larger molecules into smaller ones. This can be done with a thermal or catalytic method. The thermal cracking process follows a
Isomerization and reformation
Dragan and his colleague were the first to report about isomerization in alkanes.
Other reactions
Alkanes will react with
Occurrence
This section needs additional citations for verification. (January 2021) |
Occurrence of alkanes in the Universe


Alkanes form a small portion of the
Occurrence of alkanes on Earth
Traces of methane gas (about 0.0002% or 1745 ppb) occur in the Earth's atmosphere, produced primarily by methanogenic microorganisms, such as Archaea in the gut of ruminants.[29]
The most important commercial sources for alkanes are natural gas and
- C6H12O6 → 3 CH4 + 3 CO2
These hydrocarbon deposits, collected in porous rocks trapped beneath impermeable cap rocks, comprise commercial
Methane is also present in what is called
Alkanes have a low solubility in water, so the content in the oceans is negligible; however, at high pressures and low temperatures (such as at the bottom of the oceans), methane can co-crystallize with water to form a solid methane clathrate (methane hydrate). Although this cannot be commercially exploited at the present time, the amount of combustible energy of the known methane clathrate fields exceeds the energy content of all the natural gas and oil deposits put together. Methane extracted from methane clathrate is, therefore, a candidate for future fuels.
Biological occurrence
Acyclic alkanes occur in nature in various ways.
- Bacteria and archaea

Certain types of bacteria can metabolize alkanes: they prefer even-numbered carbon chains as they are easier to degrade than odd-numbered chains.[30]
On the other hand, certain
- CO2 + 4 H2 → CH4 + 2 H2O
Methanogens are also the producers of
- Fungi and plants
Alkanes also play a role, if a minor role, in the biology of the three eukaryotic groups of organisms: fungi, plants and animals. Some specialized yeasts, e.g., Candida tropicale, Pichia sp., Rhodotorula sp., can use alkanes as a source of carbon or energy. The fungus Amorphotheca resinae prefers the longer-chain alkanes in aviation fuel, and can cause serious problems for aircraft in tropical regions.[34]
In plants, the solid long-chain alkanes are found in the plant cuticle and epicuticular wax of many species, but are only rarely major constituents.[35] They protect the plant against water loss, prevent the leaching of important minerals by the rain, and protect against bacteria, fungi, and harmful insects. The carbon chains in plant alkanes are usually odd-numbered, between 27 and 33 carbon atoms in length[35] and are made by the plants by decarboxylation of even-numbered fatty acids. The exact composition of the layer of wax is not only species-dependent but changes also with the season and such environmental factors as lighting conditions, temperature or humidity.[35]
More volatile short-chain alkanes are also produced by and found in plant tissues. The
Edible vegetable oils also typically contain small fractions of biogenic alkanes with a wide spectrum of carbon numbers, mainly 8 to 35, usually peaking in the low to upper 20s, with concentrations up to dozens of milligrams per kilogram (parts per million by weight) and sometimes over a hundred for the total alkane fraction.[38]
- Animals
Alkanes are found in animal products, although they are less important than unsaturated hydrocarbons. One example is the shark liver oil, which is approximately 14%
Ecological relations

One example, in which both plant and animal alkanes play a role, is the ecological relationship between the
Production
This section needs additional citations for verification. (January 2021) |
Petroleum refining

As stated earlier, the most important source of alkanes is natural gas and
and processed into many products.Fischer–Tropsch
The
Laboratory preparation
There is usually little need for alkanes to be synthesized in the laboratory, since they are usually commercially available. Also, alkanes are generally unreactive chemically or biologically, and do not undergo
- C4H9Li + H2O → C4H10 + LiOH
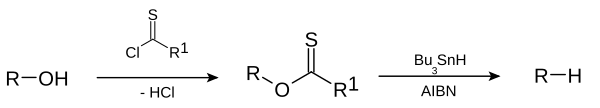
However, at times it may be desirable to make a section of a molecule into an alkane-like functionality (
- RCH=CH2 + H2 → RCH2CH3 (R = alkyl)
Alkanes or alkyl groups can also be prepared directly from
and the Clemmensen reduction[42][43][44][45] removes carbonyl groups from aldehydes and ketones to form alkanes or alkyl-substituted compounds e.g.:
Preparation of alkanes from other organic compounds
Alkanes can be prepared from a variety of organic compounds.These include alkenes, alkynes, haloalkanes, alcohols, aldehydes and ketones and carboxylic acids.
From alkenes and alkynes
When alkenes and alkynes are subjected to hydrogenation reaction by treating them with hydrogen in the presence of palladium or platinum or nickel catalyst, they produce alkanes. In this reaction powdered catalyst is preferred to increase the surface area so that adsorption of hydrogen on the catalyst increases. In this reaction the hydrogen gets attached on the catalyst to form a hydrogen-catalyst bond which leads to weakening of H-H bond, thereby leading to the addition of hydrogen on alkenes and alkynes. The reaction is exothermic because the product alkane is stable as it has more sigma bonds than the reactant alkenes and alkynes due to conversion of pi bond to sigma bonds.[46]
From haloalkanes
Alkanes can be produced from haloalkanes using different methods.
Wurtz reaction
When haloalkane is treated with sodium in dry ether, alkane with double the number of carbon atoms is obtained. This reaction proceeds through free radical intermediate and has possibility of alkene formation in case of tertiary haloalkanes and vicinal dihalides.
Corey-House-Synthesis
When haloalkane is treated with dialkyl lithium cuprite, which is otherwise known as Gilman's reagent, any higher alkane is obtained.
Reaction with metal hydride
When haloalkanes are treated with metal hydride ,eg, sodium hydride and lithium aluminium hydride.
Frankland reaction
When haloalkane is treated with zinc in ester, alkane is obtained.
Fittig reaction
When aryl halide is treated with sodium in dry ether, it forms biphenyl.
Ullmann biaryl synthesis
When aryl halide is treated with copper, it forms biphenyl.
Wurtz-Fittig reaction
When aryl halide is treated with haloalkane, we get alkyl benzene.
Applications
The applications of alkanes depend on the number of carbon atoms. The first four alkanes are used mainly for heating and cooking purposes, and in some countries for electricity generation. Methane and ethane are the main components of natural gas; they are normally stored as gases under pressure. It is, however, easier to transport them as liquids: This requires both compression and cooling of the gas.
From pentane to octane the alkanes are highly volatile liquids. They are used as fuels in internal combustion engines, as they vaporize easily on entry into the combustion chamber without forming droplets, which would impair the uniformity of the combustion. Branched-chain alkanes are preferred as they are much less prone to premature ignition, which causes knocking, than their straight-chain homologues. This propensity to premature ignition is measured by the octane rating of the fuel, where 2,2,4-trimethylpentane (isooctane) has an arbitrary value of 100, and heptane has a value of zero. Apart from their use as fuels, the middle alkanes are also good solvents for nonpolar substances.
Alkanes from nonane to, for instance, hexadecane (an alkane with sixteen carbon atoms) are liquids of higher viscosity, less and less suitable for use in gasoline. They form instead the major part of diesel and aviation fuel. Diesel fuels are characterized by their cetane number, cetane being an old name for hexadecane. However, the higher melting points of these alkanes can cause problems at low temperatures and in polar regions, where the fuel becomes too thick to flow correctly.
Alkanes from hexadecane upwards form the most important components of
Alkanes with a chain length of approximately 35 or more carbon atoms are found in bitumen, used, for example, in road surfacing. However, the higher alkanes have little value and are usually split into lower alkanes by cracking.
Some synthetic polymers such as polyethylene and polypropylene are alkanes with chains containing hundreds or thousands of carbon atoms. These materials are used in innumerable applications, and billions of kilograms of these materials are made and used each year.
Environmental transformations
This section needs expansion. You can help by adding to it. (August 2014) |
Alkanes are chemically very inert apolar molecules which are not very reactive as organic compounds. This inertness yields serious ecological issues if they are released into the environment. Due to their lack of functional groups and low water solubility, alkanes show poor bioavailability for microorganisms.[48]
There are, however, some microorganisms possessing the metabolic capacity to utilize n-alkanes as both carbon and energy sources.[49] Some bacterial species are highly specialised in degrading alkanes; these are referred to as hydrocarbonoclastic bacteria.[50]
Hazards
This section needs expansion. You can help by adding to it. (September 2017) |
Methane is flammable, explosive and dangerous to inhale; because it is a colorless, odorless gas, special caution must be taken around methane.[51] Ethane is also extremely flammable, explosive, and dangerous to inhale.[52] Both of them may cause suffocation.[51][52] Propane, too, is flammable and explosive,[53] and may cause drowsiness or unconsciousness if inhaled.[53] Butane presents the same hazards as propane.[54]
Alkanes also pose a threat to the environment. Branched alkanes have a lower biodegradability than unbranched alkanes.[55] Methane is considered to be the greenhouse gas that is most dangerous to the environment,[56] although the amount of methane in the atmosphere is relatively low.[56]
See also
- Alkene
- Alkyne
- Cycloalkane
- Higher alkanes
References
- ^
- ^ "Alkanes". Chemistry LibreTexts. 28 November 2016.
- ISBN 9788183561426.
- ^ On-Line Encyclopedia of Integer Sequences (sequence A000602 in the OEIS)
- ISBN 978-0-632-03488-8. Retrieved 12 February 2007.
- ^ Alkane Nomenclature Archived 2 February 2012 at the Wayback Machine
- ^ Thus, the ending "-diene" is applied in some cases where von Hofmann had "-ine"
- ISBN 978-0-85404-182-4.
- ^ a b William Reusch. "Nomenclature – Alkanes". Virtual Textbook of Organic Chemistry. Archived from the original on 21 May 2016. Retrieved 5 April 2007.
- ^ William Reusch. "Examples of the IUPAC Rules in Practice". Virtual Textbook of Organic Chemistry. Archived from the original on 21 May 2016. Retrieved 5 April 2007.
- ^ "IUPAC Rules". www.chem.uiuc.edu. Retrieved 13 August 2018.
- ISBN 1420044397, p. 206
- ^ "Archived copy" (PDF). Archived from the original (PDF) on 29 October 2013. Retrieved 17 February 2014.
{{cite web}}: CS1 maint: archived copy as title (link) - ^ "13. Hydrocarbons | Textbooks". textbook.s-anand.net. Archived from the original on 8 May 2011. Retrieved 3 October 2014.
- ^ ISBN 978-0-13-643669-0.
- ^ "Physical Properties of Cycloalkanes". Chemistry LibreTexts. 29 November 2015. Archived from the original on 2 February 2018. Retrieved 2 February 2018.
- .
- ^ visualization of crystal structures.
- ^ )
- PMID 17607688.
- ^ PMID 16438539.
- PMID 23868617.
- PMID 21086970.
- PMID 17165895.
- ^ a b c Asinger, Friedrich (1967). Paraffins; Chemistry and Technology. Oxford: Pergamon Press.
- .
- ^ Emily Lakdawalla. "Titan: Arizona in an Icebox?". Archived from the original on 6 April 2008. Retrieved 21 January 2004.
- S2CID 27362518.
- PMID 18424540.
- ^ "Metabolism of Alkanes and Fatty Acids – eQuilibrator 0.2 beta documentation". equilibrator.weizmann.ac.il. Retrieved 11 April 2018.
- ^ TodayIFoundOut.com, Matt Blitz -. "Do Cow Farts Actually Contribute to Global Warming?". Gizmodo. Retrieved 11 April 2018.
- PMID 28168033.
- ^ Society, National Geographic (24 July 2012). "natural gas". National Geographic Society. Retrieved 11 April 2018.
- .
- ^ ISBN 0-12-199920-3
- PMID 11192171.
- S2CID 94021819. Archived from the original(PDF) on 13 March 2013.
- PMID 11760997.
- PMID 17713987.)
{{cite journal}}: CS1 maint: unflagged free DOI (link - .
- .
- ISBN 0471264180.
- ^ Buchanan, J. G. St. C.; Woodgate, P. D. Quart. Rev. 1969, 23, 522, (Review).
- ISBN 0471264180.
- ^ Yamamura, S.; Nishiyama, S. Compr. Org. Synth. 1991, 8, 309–313, (Review).
- ^ Harikiran.B, K E Mannanam, Kottayam
- ^ "Using propane as a fuel" (PDF). Archived from the original (PDF) on 12 October 2013. Retrieved 27 November 2012.
- ISBN 978-3-642-23788-1.
- ISSN 1521-3846.
- PMID 25740314.
- ^ a b "CDC - METHANE - International Chemical Safety Cards - NIOSH". www.cdc.gov. Retrieved 19 September 2017.
- ^ a b "CDC - ETHANE - International Chemical Safety Cards - NIOSH". www.cdc.gov. Retrieved 19 September 2017.
- ^ a b "CDC - PROPANE - International Chemical Safety Cards - NIOSH". www.cdc.gov. Archived from the original on 23 November 2017. Retrieved 19 September 2017.
- ^ "CDC - BUTANE - International Chemical Safety Cards - NIOSH". www.cdc.gov. Retrieved 19 September 2017.
- ISBN 9780471109327.
- ^ a b "Pollutant Fact Sheet". apps.sepa.org.uk. Retrieved 19 September 2017.