EA-1763
The topic of this article may not meet Wikipedia's general notability guideline. (May 2022) |
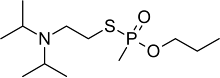
| |
| Names | |
|---|---|
| Other names
N-[2-[methyl(propoxy)phosphoryl]sulfanylethyl]-N-propan-2-ylpropan-2-amine
| |
| Identifiers | |
3D model (
JSmol ) |
|
PubChem CID
|
|
| |
| |
| Properties | |
| C12H28NO2PS | |
| Molar mass | 281.39 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
EA-1763,
Chemical characteristics
The effects of the propyl group are based on the effects of propyl
V1 has a boiling point of 316,5 degrees C. Its larger paraffin chain pushes its melting point above that of VX. The estimated solubility of V1 in water is 4 times lower compared to VX (6.8 g/L of water at 25 °C). V1 has high solubility in organic solvents and other non-polar compounds. The stability of V1 is considerably the same as that of VX in either environment. Higher insolubility and lower volatility can slow down the process. A vapor pressure at least 3 times lower than VX is speculated.[8]
A methyl group on the β-carbon adjacent to the O-bonded α-carbon tends to stabilize the induction of electrons from P to O, making P less electrophilic.[8] It is expected that the persistence of V1 is slightly higher than that of VX, estimating the hydrolysis rate between ethyl and n-propyl paraoxon, is 1.6 times more persistent in neutral medium.[9]
The lower volatility and minimal persistence difference makes VX preferable to V1.[8]
Preparation
The manufacture of S-(2-dialkylaminoethyl) O-alkyl alkylphosphonothiolates essentially involves the conditions reported by Eckhaus et al.[10]
References
- ^ a b John B. Samuel, Elwin C. Penski, John J. Callahan. PHYSICAL PROPERTIES OF STANDARD AGENTS, CANDIDATE AGENTS, AND RELATED COMPOUNDS AT SEVERAL TEMPERATURES (U). p 24 and 279.
- PMID 27000540.
- ^ PubChem. "Propyl acetate". pubchem.ncbi.nlm.nih.gov. Retrieved 2022-05-29.
- ^ PubChem. "Ethyl acetate". pubchem.ncbi.nlm.nih.gov. Retrieved 2022-05-29.
- ^ PubChem. "O-Ethyl S-(2-diisopropylaminoethyl) methylphosphonothioate". pubchem.ncbi.nlm.nih.gov. Retrieved 2022-05-29.
- ^ a b Ledgard, J. A Laboratory History of Chemical Warfare Agents. p 223-225.
- ^ Coulter, P. B.; Callahan, J. J.; Link, R.S. Physical Constants of Thirteen V Agents. U. S. Army Chemical Warfare Laboratories Technical Report (Report). CWLR-2346.
- ^ a b c cit-OPDC. The preparatory manual to chemical warfare. Vol 1: V1
- ^ CHEMICAL REVIEWS VOLUME6 4, NUMBER 4. JULY 24, 1964. page 318.
- ^ Eckhaus et al. US 3,911,059
