T-2 mycotoxin
This article needs additional citations for verification. (October 2008) |

| |

| |
| Names | |
|---|---|
| IUPAC name
(2α,3α,4β,8α)-4,15-bis(acetyloxy)-3-hydroxy-12,13-epoxytrichothec-9-en-8-yl 3-methylbutanoate
| |
| Other names
T-2 Toxin
Fusariotoxin T 2 Insariotoxin Mycotoxin T 2 | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.040.255 |
PubChem CID
|
|
RTECS number
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C24H34O9 | |
| Molar mass | 466.527 g·mol−1 |
| Insoluble | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
T-2 mycotoxin is a
History
Alimentary toxic aleukia (ATA), a disease which is caused by trichothecenes like T-2 mycotoxin, killed many thousands of USSR citizens in the
In 1981, the
T-2 mycotoxin is also thought to be a cause of
Chemical properties
This compound has a tetracyclic
Mechanism of action
The
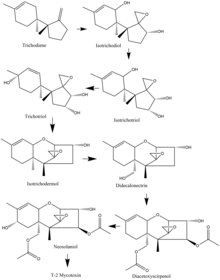
Synthesis
T-2 mycotoxin is produced naturally by Fusarium fungi of which the most important species are: F. sporotrichioides, F. langsethiae, F. acuminatum and F. poae. These fungi are found in grains such as
In the Fusarium species, biosynthesis of the T-2 mycotoxin often starts with trichodiene, and many of the species share a common route of oxidizations and cyclizations. As an example, from the F. sporotrichioides species, the important oxidation steps that occur start from trichodiene and goes to isotrichodiol. From there, the eleventh carbon atom is oxidized to form isotrichotriol. The ninth carbon is then oxidized, and trichotriol is formed, which then cyclizes to make isotrichodermol. After that, the fifteenth carbon is oxidized to form didecalonectrin, which leads to the fourth carbon being oxidized, and diacetoxyscirpenol is formed. The second to last step is the oxidation of the eighth carbon to make neosolaniol, which then undergoes slight modification to create the T-2 toxin.[17]
Toxicity
ADME properties
Absorption and exposure
Humans and animals are generally exposed to T-2 mycotoxins through food. Certain grains can contain the toxin which makes it a threat to human health and an economic burden.
The
Distribution
T-2 mycotoxin is distributed uniformly throughout the body without preference to a specific organ or site. In rodents, plasma concentration levels peak around roughly thirty minutes after exposure, and in one study, the half-life of the T-2 toxin was seen to be less than twenty minutes. In a different study involving pigs, the distribution after four hours of IV injection was seen to be 15–24% in the GI tract and 4.7–5.2% in various other tissues.[20]
Metabolism
Once absorbed and distributed to various tissues, the T-2 mycotoxin goes through various metabolic reactions before it gets excreted. In vivo studies showed that the most occurring reactions are
Excretion
Following absorption, distribution, and metabolism, T-2 mycotoxin is excreted fairly quickly, where 80–90% of it is excreted within 48 hours.[20] The main methods of excretion seem to be from the urine and feces,[21] where excretion through bile contributes heavily to the feces route of excretion.[14] There is also very little of the parent T-2 mycotoxin in the excretions, meaning most of the initial compound is metabolized beforehand.[21]
Toxic effects
T-2 is highly toxic when inhaled. Acute toxic symptoms include vomiting, diarrhea, skin irritation, itching, rash, blisters, bleeding and
At first the patient experiences a burning sensation in the mouth, throat and stomach. After a few days the person will suffer from an acute
In the end the following symptoms might occur: a high fever,
Effects on animals
T-2 mycotoxin is also toxic to animals. The compound is known for having lethal and sub-lethal effects on farm animals. It is often found in contaminated cereal grains that are fed to these animals.
The compound also seems to reduce the fertility of ewes and heifers. Research has shown that a high dose of T-2 delays the
T-2 also has an effect on the fertility of bulls. In 1998 it was discovered that moldy hay influenced the quality of semen of bulls. Analysis of the moldy hay showed that T-2 was present. The compound decreased sperm motility and testosterone levels and increased the frequency of morphological abnormalities in the sperm cells.
The liver is another target for the mycotoxin. It is one of the first organs where the compound passes through after ingestion. Here it causes a reduced expression of
All of the mentioned effects happen when T-2 is ingested in high doses. Animals are able to metabolize the compound with enzymes from the CYP3A family, just like humans.
Treatments
At the moment, there is no specific therapy for T-2 mycotoxin poisonings.
Application
There are currently no applications, aside from war, for T-2 mycotoxins; however, there are some plausible therapeutic uses. Due to their abilities, research shows possible uses for the mycotoxin as growth promoters,
See also
References
- ^ "T-2 Toxin: essential data". CBWInfo.com. Archived from the original on October 12, 2008.
- PMID 22749975.
- ^ Pitt JI (July 1989). Semple RL, Frio AS, Hicks PA, Lozare JW (eds.). An introduction to mycotoxins. Mycotoxin Prevention and kontrol in Food Grains. Proceedimgs of Assistance for The Training Course. Bangkok.
- ^ Shultz GP (1982). Chemical warfare in Southeast Asia and Afghanistan: an update (Report). US Department of State, Bureau of Public Affairs, Office of Public Communication, Editorial Division.
- S2CID 4263047.
- ^ a b "Yellow Rain Falls". New York Times. September 3, 1987. Archived from the original on November 9, 2012. Retrieved January 20, 2022.
Yellow rain is the excrement of jungle bees. It's yellow from digested pollen grains, and it rains down from swarms of bees too high to be seen. His theory turns out to be exactly right. The Government's own studies, still unpublished, prove that the source is bees, not bombs.
- ^ Meselson MS, Robinson JP (June 2008). "The Yellow Rain Affair: Lessons from a Discredited Allegation". In Clunan AL, Lavoy PR, Martin SB (eds.). Terrorism, War, or Disease? Unraveling the Use of Biological Weapons. Stanford: Stanford University Press. pp. 72–96. Archived from the original on 2014-07-27. Retrieved 2015-09-03.
- ^ Zhang Z (1977). "A Study of the Origin and the Pollen Analysis of "Yellow Rain" in Northern Jiangsu". Kexue Tongbao. 22: 409–12.
- PMID 9244334.
- ^ CBRNE - T-2 Mycotoxins at eMedicine
- PMID 23907020.
- S2CID 13540977.
- S2CID 14146122.
- ^ PMID 21417259.
- ISBN 978-0-429-12493-8.
- ^ "T-2 toxin from fusarium sp., powder, ≥98% (HPLC)". Sigma-Aldridge.
- PMID 8246841.
- PMID 25588579.
- PMID 25680507.
- ^ PMID 28430618.
- ^ ISBN 9789997320919.
- ^ Kalantari H, Mousavi M (2010). "Review on T-2 Toxin". Jundishapur Journal of Natural Pharmaceutical Products. 5 (1): 26–38.
- ^ Semple RL, Frio AS, Hicks PA, Lozare JV (1989). Mycotoxin prevention and control in foodgrains. UNDP/FAO Regional Network Inter-Country Cooperation on Preharvest Technology and Quality Control of Foodgrains (REGNET) and the ASEAN Grain Postharvest Programme (Report). Thailand.
- PMID 23916251.
- PMID 25079423.
- PMID 31941063.
- PMID 23524315.
Further reading
- "Mycotoxin (T-2)". Medical Management of Biological Casualties: Handbook. US Army Medical Research Institute of Infectious Diseases (USAMRIID). 1998. pp. 107–111.
- Bamburg JR, Riggs NV, Strong FM (1968). "The structure of toxins from two stains of Fusarium tricinctum". Tetrahedron. 24 (8): 3329–3336. PMID 5648271.
- Bamburg JR, Strong FM (1971). "12,13-Epoxytrichothecenes.". In Kadis S, Ciegler A, Ajl SJ (eds.). Microbial Toxins. Vol. VII. New York, NY: Academic Press. pp. 207–292.
