User:Thingg/neptunium
Refs: critical mass; health effects; general info (German). (These are not great sources, but links to info for which we should search for sources. There are some errors in the German article...and no, they're not typos, although I did see at least one of those!)
Physical properties refs: specific heat allotropes electrical resistivity, thermoelectric power in 300–900 K
Some properties (sorry, don't have fulltext for this one): [1]
Yoshida et al. (see bibliography at bottom) is a very rich source of material when it comes to chemistry.
Phase diagrams for Np chemical thermodynamics of Np and Pu
Header
 | |||||||||||||||||||||||||||||||||||||
| Neptunium | |||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pronunciation | /nɛpˈtjuːniəm/ | ||||||||||||||||||||||||||||||||||||
| Appearance | silvery metallic | ||||||||||||||||||||||||||||||||||||
| Mass number | [237] | ||||||||||||||||||||||||||||||||||||
| Neptunium in the periodic table | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
kJ/mol | |||||||||||||||||||||||||||||||||||||
| Heat of vaporization | 336 kJ/mol | ||||||||||||||||||||||||||||||||||||
| Molar heat capacity | 29.46 J/(mol·K) | ||||||||||||||||||||||||||||||||||||
Vapor pressure
| |||||||||||||||||||||||||||||||||||||
| Atomic properties | |||||||||||||||||||||||||||||||||||||
Philip H. Abelson (1940) | |||||||||||||||||||||||||||||||||||||
| Isotopes of neptunium | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
Neptunium is a
Although many false claims of its discovery were made over the years, the element was first synthesized by
The most stable isotope of neptunium, neptunium-237, is a by-product of nuclear reactors and plutonium production, and it and the isotope neptunium-239 are also found in trace amounts in uranium ores due to neutron capture reactions and beta decay.[5]
Characteristics
Physical
Neptunium is a
Neptunium is found in at least three
| Neptunium allotrope | α | β (measured at 313 °C) | γ (measured at 600 °C) |
|---|---|---|---|
| Transition temperature | (α→β) 282 °C | (β→γ) 583 °C | (γ→liquid) 639 °C |
| Symmetry | Orthorhombic
|
Tetragonal
|
Body-centered cubic
|
| Density (g/cm3) | 20.45 | 19.36 | 18.0 |
| Space group | Pnma | P42 | Im3m |
pm )
|
a = 666.3 b = 472.3 c = 488.7 |
a = 489.7 c = 338.8 |
a = 351.8 |

α-neptunium takes on an
β-neptunium takes on a distorted tetragonal close-packed structure. Four atoms of neptunium make up a unit cell, and the Np–Np bond lengths are 276 pm.
Neptunium metal is similar to uranium in terms of physical workability. When exposed to air at normal temperatures, it forms a thin oxide layer. This reaction proceeds more rapidly as the temperature increases. The metal has been determined to melt at 639±3 °C; and although the boiling point is not empirically known, the currently accepted value of 4174 °C is from an extrapolation of the vapor pressure of the element. If accurate, this would give neptunium the largest liquid range of any element (3535 °C).[6][13]
Alloys
Due to the presence of valence 5f electrons, the actinides and their alloys exhibit very interesting magnetic behavior. These can range from the itinerant band-like character characteristic of the
One neptunium-based superconductor alloy has been discovered with formula NpPd5Al2. This occurence in Np compounds is somewhat surprising because they often exhibit strong magnetism, which usually destroys superconductivity. The alloy has a tetragonal structure with a superconductivity transition temperature of −268 °C (4.9 K).[19][20]
Chemical
Neptunium has five ionic
Atomic
A neptunium atom has 93 electrons, arranged in the
Isotopes

20 neptunium
The isotopes of neptunium range in
The isotopes neptunium-235, -236, and -237 are predicted to be
Occurrence
Since all isotopes of neptunium have half-lives that are many times shorter than the
Most neptunium (and plutonium) now encountered in the environment is due to atmospheric nuclear explosions that took place between the detonation of the
Once in the environment, neptunium generally
History
Background and early claims
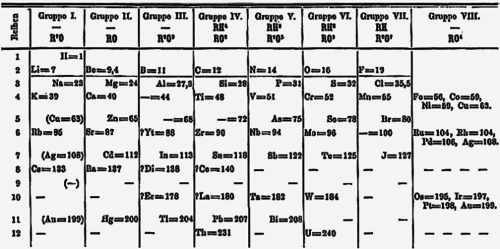
When the first
After several months of work, Fermi's group had tentatively determined that lighter elements would disperse the energy of the captured neutron by emitting a

Although the many different and unknown radioactive half-lives in the experiment's results showed that several nuclear reactions were occurring, Fermi's group could not prove that element 93 was being created unless they could isolate it chemically. They and many other scientists attempted to accomplish this, including
While the question of whether Fermi's experiment had produced element 93 was stalemated, two additional claims of the discovery of the element appeared; although unlike Fermi they both claimed to have observed it in nature. The first of these claims was by Czech engineer
Although by 1938 some scientists, including
Discovery

As research on nuclear fission progressed in early 1939,
However, as more information about fission became available, the possibility that the fragments of nuclear fission could still have been present in the target became more remote. McMillan and several scientists, including
- (The times are half-lives.)
This proved that the unknown radioactive source originated from the decay of uranium and, coupled with the previous observation that the source was different chemically from all known elements, proved beyond all doubt that a new element had been discovered. McMillan and Abelson published their results in a paper entitled Radioactive Element 93 in the Physical Review on May 27, 1940.[59] They did not propose a name for the element in the paper, but they soon decided on the name neptunium since Neptune is the next planet beyond Uranus in our solar system.[19][60][61][62] Although it was also realized that the beta decay of 239Np must produce an isotope of element 94 (now called plutonium), the quantities involved in McMillan and Abelson's original experiment were too small to isolate and identify plutonium along with neptunium.[63]
Subsequent developments
Although the new element's unique radioactive characteristics allowed it to be traced as it moved through various compounds in chemical reactions, at first this was the only method available to prove that its chemistry was different from other elements. As the first isotope of neptunium to be discovered has such a short half-life, McMillan and Abelson were unable to prepare a sample that was large enough to perform chemical analysis of the new element using the technology that was then available. However, after the discovery of the long-lived 237Np isotope in 1942 by
Much of the research into the properties of neptunium since then has been focused on understanding how to confine it as a portion of nuclear waste. Because it has isotopes with very long half-lives, it is of particular concern in the context of designing confinement facilities that can last for thousands of years. It has found some limited uses as a radioactive tracer and a precursor for various nuclear reactions to produce useful plutonium isotopes. However, most of the neptunium that is produced as a reaction byproduct in nuclear power stations is considered to be a waste product.[19][64]
Production

Synthesis
The vast majority of the neptunium that currently exists on Earth was produced in human-induced nuclear reactions. Neptunium-237 is the most commonly synthesized isotope due to it being the only one that can both be created via neutron capture and also has a half-life long enough to allow weighable quantities to be easily isolated. As such, it is by far the most common isotope to be utilized in chemical studies of the element.[26]
- When an gamma radiation. Further neutron capture creates 237U which has a half-life of 7 days and thus quickly decays to 237Np through beta decay. During beta decay, the excited 237U emits an electron, while the atomic weak interaction converts a neutron to a proton, thus creating 237Np.[26]
- 237U is also produced via an (n,2n) reaction with 238U. This only happens with very energetic neutrons.[26]
- 237Np is the product of alpha decay of 241Am, which is produced through neutron irradiation of uranium-238.[26]
Heavier isotopes of neptunium decay quickly, and lighter isotopes of neptunium cannot be produced by neutron capture, so chemical separation of neptunium from cooled
Artificial 237Np metal is usually isolated through a reaction of 237NpF3 with liquid
- 2 NpF3 + 3 Ba → 2 Np + 3 BaF2
By weight, neptunium-237 discharges are about 5% as great as plutonium discharges and about 0.05% of spent nuclear fuel discharges.[67] However, even this fraction still amounts to more than fifty tons per year globally.[68]
Purification methods
Recovering uranium and plutonium from spent nuclear fuel for reuse is one of the major processes of the nuclear fuel cycle. As it has a long half-life of 2.144 ×106 years, the alpha emitter 237Np is one of the major isotopes separated from spent nuclear fuel.[69] Many separation methods have been used to separate out the neptunium, operating on small and large scales. The small-scale purification operations have the goals of preparing pure neptunium as a precursor of metallic neptunium and its compounds, and also to isolate and preconcentrate neptunium in samples for analysis.[69]
Most methods that separate neptunium ions exploit the differing chemical behaviour of the differing oxidation states of neptunium (from +3 to +6 or sometimes even +7) in solution.
Chemistry and compounds
Solution chemistry

When it is in an aqueous solution, neptunium can exist in any of its possible oxidation states and each of these show a characteristic color. The stability of each oxidation state is strongly dependent on various factors, such as the presence of oxidizing or reducing agents, pH of the solution, presence of coordination complex-forming ligands, and even the concentration of neptunium in the solution.[71]
In
2, NpO2+
2, and NpO+
3. In basic solutions, they exist as the oxides and hydroxides Np(OH)3, NpO2, NpO2OH, NpO2(OH)2, and NpO3−
5. Not as much work has been done to characterize neptunium in basic solutions.[71] Np3+ and Np4+ can easily be reduced and oxidized to each other, as can NpO+
2 and NpO2+
2.[72]
- Neptunium(III)
Np(III) or Np3+ exists as hydrated complexes in acidic solutions, Np(H
2O)3+
n.
- Neptunium(IV)
Np(IV) or Np4+ is pale yellow-green in acidic solutions,[19] where it exists as hydrated complexes (Np(H
2O)4+
n). It is quite unstable to hydrolysis in acidic aqueous solutions at pH 1 and above, forming NpOH3+.[74] In basic solutions, Np4+ tends to hydrolyze to form the neutral neptunium(IV) hydroxide (Np(OH)4) and neptunium(IV) oxide (NpO2).[74]
- Neptunium(V)
Np(V) or NpO+
2 is green-blue in aqueous solution,
2 and PuO+
2, NpO+
2 does not spontaneously disproportionate except at very low pH and high concentration:[72]
- 2 NpO+
2 + 4 H+ ⇌ Np4+ + NpO2+
2 + 2 H2O
It hydrolyzes in basic solutions to form NpO2OH and NpO
2(OH)−
2.[74]
- Neptunium(VI)
Np(VI) or NpO2+
2, the neptunyl ion, shows a light pink or reddish color in an acidic solution and yellow-green otherwise.[19] It is a strong Lewis acid[71] and is the main neptunium ion encountered in solutions of pH 3–4.[74] Though stable in acidic solutions, it is quite easily reduced to the Np(V) ion,[71] and it is not as stable as the homologous hexavalent ions of its neighbours uranium and plutonium (the uranyl and plutonyl ions). It hydrolyzes in basic solutions to form the oxo and hydroxo ions NpO2OH+, (NpO
2)
2(OH)2+
2, and (NpO
2)
3(OH)+
5.[74]
- Neptunium(VII)
Np(VII) is dark green in a strongly
3; water quickly reduces this to Np(VI).[71] Its hydrolysis products are uncharacterized.[74]
Hydroxides
The oxides and hydroxides of neptunium are closely related to its ions. In general, Np hydroxides at various oxidation levels are less stable than the actinides before it on the periodic table such as thorium and uranium and more stable than those after it such as plutonium and americium. This phenomenon is caused by the fact that the stability of an ion increases as the ratio of atomic number to the radius of the ion increases. Thus actinides higher on the periodic table will more readily undergo hydrolysis.[74][71]
Neptunium(III) hydroxide is quite stable in acidic solutions and in environments that lack oxygen, but it will rapidly oxidize to the IV state in the presence of air. It is not not soluble in water.[64] Np(IV) hydroxides exist mainly as the electrically neutral Np(OH)4 and its mild solubility in water is not affected at all by the pH of the solution. This suggests that the other Np(IV) hydroxide, Np(OH)−
5, does not have a significant presence.[74][75]
Because the Np(V) ion NpO+
2 is very stable, it can only form a hydroxide in high acidity levels. When placed in a 0.1 M sodium perchlorate solution, it does not react significantly for a period of months, although a higher molar concentration of 3.0 M will result in it reacting to the solid hydroxide NpO2OH almost immediately. Np(VI) hydroxide is more reactive but it is still fairly stable in acidic solutions. It will form the compound NpO3· H2O in the presence of ozone under various carbon dioxide pressures. Np(VII) has not been well-studied and no neutral hydroxides have been reported. It probably exists mostly as [NpO
4(OH)
2]3−
.[74][76][77][78]
Oxides
Three nonhydrous neptunium oxides have been reported, NpO2, Np2O5, and Np5O8; although some studies[79] have stated that only the first two of these exist, suggesting that claims of Np5O8 are actually the result of mistaken analysis of Np2O5. However as the full extent of the reactions that occur between neptunium and oxygen has yet to be researched, it is not certain which of these claims is accurate. Although pure neptunium oxide has not been produced in as high an oxidation state as is possible with the adjacent actinide uranium, neptunium oxides are more stable at lower oxidation levels. This behavior is illustrated by the fact that NpO2 can be produced by simply burning salts of oxy acid in air.[19][80][81][82]
NpO2 is very stable over a large range of pressures and temperatures and does not undergo phase transitions at low temperatures. It does show a phase transition from face-centered cubic to orthorhombic at around 33-37GPa, although it returns to is original phase when pressure is released. It remains stable under oxygen pressures up to 2.84 MPa and temperatures up to 400°C. Np2O5 is brown in color and
Neptunium also forms a large number of oxide compounds with a wide variety of elements, although the neptunate oxides formed with
- NpO2 + 3 MO2 → MNpO5 (M = K, Cs, Rb)
The oxide compounds KNpO4, CsNpO4, and RbNpO4 are formed by reacting Np(VII) ([NpO
4(OH)
2]3−
) with a compound of the alkali metal nitrate and ozone. Additional compounds have been producted by reacting NpO3 and water with solid alkali and alkaline peroxides at temperatures of 400 - 600°C for 15-30 hours. Some of these include Ba3(NpO5)2, Ba2NaNpO6, and Ba2LiNpO6. Also, a considerable number of hexavelant neptunium oxides are formed by reacting solid-state NpO2 with various alkali or alkaline earth oxides in an environment of flowing oxygen. Many of the resulting compounds also have an equivalent compound that substitutes uranium for neptunium. Some compounds that have been characterized include Na2Np2O7, Na4NpO5, Na6NpO6, and Na2NpO4. These can be obtained by heating different combinations of NpO2 and Na2O to various temperature thresholds and further heating will also cause these compounds to exhibit different neptunium allotropes. The lithium neptunate oxides Li6NpO6 and Li4NpO5 can be obtained with similar reactions of NpO2 and Li2O.[85][86][87][88][89][90][91][92]
A large number of additional alkali and alkaline neptunium oxide compounds such as Cs4Np5O17 and Cs2Np3O10 have been characterized with various production methods. Neptunium has also been observed to bond with oxides of many additional elements in groups 3 through 7, although these are much less well studied.[85][93][94]
Halides
Although neptunium halide compounds have not been nearly as well studied as its oxides, a fairly large number have been successfully characterized. Of these, neptunium fluorides have been the most extensively researched, largely because of their potential use in separating the element from nuclear waste products. Four binary neptunium fluoride compounds, NpF3, NpF4, NpF5, and NpF6, have been reported. The first two are fairly stable and were first prepared in 1947 through the following reactions:
- NpO2 + ½ H2 + 3 HF → NpF3 + 2 H2O (400°C)
- NpF3 + ¼ O2 + HF → NpF4 + ½ H2O (400°C)
Later, NpF4 was obtained directly by heating NpO2 to various temperatures in mixtures of either hydrogen fluoride or pure fluorine gas. NpF5 is much more difficult to create and most known preparation methods involve reacting NpF4 or NpF6 compounds with various other fluoride compounds. NpF5 will decompose into NpF4 and NpF6 when heated to around 320°C.[95][96][97][98]
NpF6 or
Four neptunium oxyfluoride compounds, NpO2F, NpOF3, NpO2F2, and NpOF4; have been reported although none of them have been extensively studied. NpO2F2 is a pinkish solid and can be prepared by reacting NpO3 · H2O and Np2F5 with pure fluorine at around 330°C. NpOF3 and NpOF4 can be produced by reacting neptunium oxides with anhydrous hydrogen fluoride at various temperatures. Neptunium also forms a wide variety of fluoride compounds with various elements. Some of these that have been characterized include CsNpF6, Rb2NpF7, Na3NpF8, and K3NpO2F5.[96][98][102][103][104][105][106]
Two neptunium chlorides, NpCl3 and NpCl4, have been characterized and although several attempts to create NpCl5 have been made, they have not been successful. NpCl3 is created by reducing neptunium dioxide with hydrogen and carbon tetrachloride (CCl4) and NpCl4 by reacting a neptunium oxide with CCl4 at around 500°C. Other neptunium chloride compounds have also been reported, including NpOCl2, Cs2NpCl6, Cs3NpO2Cl4, and Cs2NaNpCl6. Neptunium bromides NpBr3 and NpBr4 have also been created; the latter by reacting aluminium bromide with NpO2 at 350°C and the former in an almost identical procedure but with zinc present. The neptunium iodide NpI3 has also been prepared by the same method as NpBr3.[107][108][109]
Chalcogenides, pnictides, and carbides
Neptunium chalcogen and pnictogen compounds have been well studied primarily as part of research into their electronic and magnetic properties and their interactions in the natural environment. Pnictide and carbide compounds have also attracted interest because of their presence in the fuel of several advanced nuclear reactor designs, although the latter group has not had nearly as much research as the former.[110]
- Chalcogenides
A wide variety of neptunium sulfide compounds have been characterized, including the pure sulfide compounds NpS, NpS3, Np2S5, Np3S5, Np2S3, and Np3S4. Of these, Np2S3, prepared by reacting NpO2 with hydrogen sulfide and carbon disulfide at around 1000°C, is the most well-studied and three allotropic forms are known. The α form exists up to around 1230°C, the β up to 1530°C, and the γ form, which can also exist as Np3S4, at higher temperatures. NpS can be created by reacting Np2S3 and neptunium metal at 1600°C and Np3S5 can be prepared by the decomposition of Np2S3 at 500°C or by reacting sulfur and neptunium hydride at 650°C. Np2S5 is made by heating a mixture of Np3S5 and pure sulfur to 500°C. All of the neptunium sulfides except for the β and γ forms of Np2S3 are isostructural with the equivalent uranium sulfide and several, including NpS, α−Np2S3, and β−Np2S3 are also isostructural with the equivalent plutonium sulfide. The oxysulfides NpOS, Np4O4S, and Np2O2S have also been created, although the latter three have not been well studied. NpOS was first prepared in 1985 by vacuum sealing NpO2, Np3S5, and pure sulfur in a quartz tube and heating it to 900°C for one week.[110][111][112][113][114][115][116]
Neptunium selenide compounds that have been reported include NpSe, NpSe3, Np2Se3, Np2Se5, Np3Se4, and Np3Se5. All of these have only been obtained by heating neptunium hydride and selenium metal to various temperatures in a vacuum for an extended period of time and Np2Se3 is only known to exist in the γ allotrope at relatively high temperatures. Two neptunium oxyselenide compounds are known, NpOSe and Np2O2Se, are formed with similar methods by replacing the neptunium hydride with neptunium dioxide. The known neptunium telluride compounds NpTe, NpTe3, Np3Te4, Np2Te3, and Np2O2Te are formed by similar procedures to the selenides and Np2O2Te is isostructural to the equivalent uranium and plutonium compounds. No neptunium−polonium compounds have been reported.[110][116][117][118][119]
- Pnictides and carbides
Neptunium
Three neptunium arsenide compounds have been prepared, NpAs, NpAs2, and Np3As4. The first two were first created by heating arsenic and neptunium hydride in a vacuum-sealed tube for about a week. Later, NpAs was also made by confining neptunium metal and arsenic in a vacuum tube, separating them with a quartz membrane, and heating them to just below neptunium's melting point of 639°C, which is slightly higher than the arsenic's sublimation point of 615°C. Np3As4 is prepared by a similar procedure using iodine as a transporting agent. NpAs2 crystals are brownish gold and Np3As4 is black. The neptunium antimonide compound NpSb was created in 1971 by placing equal quantities of both elements in a vacuum tube, heating them to the melting point of antimony, and then heating it further to 1000°C for sixteen days. This procedure also created trace amounts of an additional antimonide compound Np3Sb4. One neptunium-bismuth compound, NpBi, has also been reported.[120][121][123][124][125][126]
The neptunium carbides NpC, Np2C3, and NpC2 (tentative) have been reported, but have not characterized in detail despite the high importance and utility of actinide carbides as advanced nuclear reactor fuel. NpC is a non-stoichiometric compound, and could be better labelled as NpCx (0.82 ≤ x ≤ 0.96). It may be obtained from the reaction of neptunium hydride with graphite at 1400 °C or by heating the constituent elements together in an electric arc furnace using a tungsten electrode. It reacts with excess carbon to form pure Np2C3. NpC2 is formed from heating NpO2 in a graphite crucible at 2660–2800 °C.[120][121][127][128]
Other inorganic
- Hydrides
Neptunium reacts with
- Phosphates, sulfates, and carbonates
Being chemically stable, neptunium
Organometallic

A few organoneptunium compounds are known and chemically characterized, although not as many as for
- NpCl4 + 4 KC5H5 → Np(C5H5) + 4 KCl
It is soluble in benzene and THF, and is less sensitive to oxygen and water than Pu(C5H5)3 and Am(C5H5)3.[132] Other Np(IV) cyclopentadienyl compounds are known for many ligands: they have the general formula (C5H5)3NpL, where L represents a ligand.[132]
Coordination complexes
There is much interest in the
Solid state
Few neptunium(III) coordination compounds are known, because Np(III) is readily oxidized by atmospheric oxygen while in aqueous solution. However,
2(C
2O
4)
3·11H2O, Np
2(C
6H
5AsO
3)
3·H2O, and Np
2[C
6H
4(OH)COO]
3.[133]
Many neptunium(IV) coordination compounds have been reported, the first one being (Et
4N)Np(NCS)
8, which is isostructural with the anologous uranium(IV) coordination compound.[133] Other Np(IV) coordination compounds are known, some involving other metals such as cobalt (CoNp
2F
10·8H2O, formed at 400 K) and copper (CuNp
2F
10·6H2O, formed at 600 K).[133] Complex nitrate compounds are also known: the experimenters who produced them in 1986 and 1987 produced single crystals by slow evaporation of the Np(IV) solution at ambient temperature in concentrated nitric acid and excess 2,2′-pyrimidine.[133]
The coordination chemistry of neptunium(V) has been extensively researched due to the presence of
4(NpO
4)
2C
12O
12·8H2O and neptunium glycolate, both of which form green crystals.[133]
Neptunium(VI) compounds range from the simple oxalate NpO
2C
2O
4 (which is unstable, usually becoming Np(IV)) to such complicated compounds as the green (NH
4)
4NpO
2(CO
3)
3.[133] Extensive study has been performed on compounds of the form M
4AnO
2(CO
3)
3, where M represents a monovalent cation and An is either uranium, neptunium, or plutonium.[133]
Since 1967, when neptunium(VII) was discovered, some coordination compounds with neptunium in the +7 oxidation state have been prepared and studied. The first reported such compound was initially characterized as Co(NH
3)
6NpO
5·nH2O in 1968, but was suggested in 1973 to actually have the formula [Co(NH
3)
6][NpO
4(OH)
2]·2H2O based on the fact that Np(VII) occurs as [NpO
4(OH)
2]3−
in aqueous solution.
In aqueous solution
Most neptunium
Analogously to its neighbours, uranium and plutonium, the order of the neptunium ions in terms of complex formation ability is Np4+ > NpO2+
2 ≥ Np3+ > NpO+
2. (The relative order of the middle two neptunium ions depends on the ligands and solvents used.)[134] The stability sequence for Np(IV), Np(V), and Np(VI) complexes with monovalent inorganic ligands is F− > H
2PO−
4 > SCN− > NO−
3 > Cl− > ClO−
4; the order for divalent inorganic ligands is CO2−
3 > HPO2−
4 > SO2−
4. These follow the strengths of the corresponding acids. The divalent ligands are more strongly complexing than the monovalent ones.[134] NpO+
2 can also form the complex ions [NpO+
2X3+
] (X = Al, Ga, Sc, In, Fe, Cr, Rh) in perchloric acid solution: the strength of interaction between the two cations follows the order Fe > In > Sc > Ga > Al.[134] The neptunyl and uranyl ions can also form a complex together.[134]
Applications
Precursor in plutonium production
237Np is irradiated with neutrons to create
238Pu also exists in sizable quantities in
Weapons
Neptunium is
In September 2002, researchers at the Los Alamos National Laboratory briefly created the first known nuclear critical mass using neptunium in combination with shells of enriched uranium (uranium-235), discovering that the critical mass of a bare sphere of neptunium-237 "ranges from kilogram weights in the high fifties to low sixties,"[138] showing that it "is about as good a bomb material as [uranium-235]."[16] The United States Federal government made plans in March 2004 to move America's supply of separated neptunium to a nuclear-waste disposal site in Nevada.
Physics
237Np is used in devices for detecting high-energy (MeV) neutrons.[139]
Role in nuclear waste
Neptunium-237 is the most mobile actinide in the deep geological repository environment.[140] This makes it and its predecessors such as americium-241 candidates of interest for destruction by nuclear transmutation.[141] Neptunium accumulates in commercial household ionization-chamber smoke detectors from decay of the (typically) 0.2 microgram of americium-241 initially present as a source of ionizing radiation. With a half-life of 432 years, the americium-241 in a smoke detector includes about 3% neptunium after 20 years, and about 15% after 100 years.
Due to its long half-life, neptunium becomes the major contributor of the total radiation in 10,000 years. As it is unclear what happens to the containment in that long time span, an extraction of the neptunium would minimize the contamination of the environment if the nuclear waste could be mobilized after several thousand years.[142][143]
Biological role and precautions
Neptunium does not have any biological role. Animal tests showed that it is not absorbed via the
Finely divided neptunium metal presents a fire hazard because neptunium is pyrophoric; small grains will ignite spontaneously in air at room temperature.[129]
References
- ^ ISBN 978-1-62708-155-9.
- PMID 28553487.
- ^ Magnetic susceptibility of the elements and inorganic compounds, in Handbook of Chemistry and Physics 81st edition, CRC press.
- .
- ^ ISBN 0-8493-0485-7.
- ^ a b c d Yoshida et al., p. 718
- )
- ISBN 5-7695-2533-9.
- .
- ^ Lemire,R.J. et al.,Chemical Thermodynamics of Neptunium and Plutonium, Elsevier, Amsterdam, 2001
- ^ "Crystal Lattice Structures: The αNp (Ac) Structure". United States Naval Research Laboratory Center for Computational Materials Science. Retrieved 2013-10-16.
- ^ a b c d Yoshida et al., p. 719
- ^ a b Theodore Gray. The Elements. Page 215
- ^ Hindman JC 1968, 'Neptunium', in CA Hampel (ed.), The encyclopedia of the chemical elements, Reinhold, New York, pp. 434
- ^ Dunlap BD, Brodsky MB, Shenoy GK & Kalvius GM 1970, 'Hyperfine interactions and anisotropic lattice vibrations of 237Np in α-Np metal', Physical Review B, vol. 1, no. 1, pp. 44. 46, doi:10.1103/PhysRevB.1.44
- ^ a b c d "Evaluation of nuclear criticality safety data and limits for actinides in transport" (PDF). Institut de Radioprotection et de Sûreté Nucléaire. p. 15. Retrieved 2010-12-20. Cite error: The named reference "critical" was defined multiple times with different content (see the help page).
- )
- ^ a b c Yoshida et al., pp. 719–20
- ^ a b c d e f g h i j k l "Periodic Table Of Elements: LANL - Neptunium". Los Alamos National Laboratory. Retrieved 2013-10-13.
- doi:10.1088/1742-6596/150/4/042119.)
{{cite journal}}: CS1 maint: multiple names: authors list (link) CS1 maint: numeric names: authors list (link - ^ V.A. Mikhailov, ed. (1971). Analytical chemistry of neptunium. Moscow: Nauka.
- ^ Golub, A. M. (1971). Общая и неорганическая химия (General and Inorganic Chemistry). Vol. 2. pp. 222–7.
- ^ Martin, W. C.; Hagan, Lucy; Reader, Joseph; Sugan, Jack (1974). "Ground Levels and Ionization Potentials for Lanthanide and Actinide Atoms and Ions" (PDF). J. Phys. Chem. Ref. Data. 3 (3): 771–9. Retrieved 19 October 2013.
- ^ David R. Lide (ed), CRC Handbook of Chemistry and Physics, 84th Edition. CRC Press. Boca Raton, Florida, 2003; Section 10, Atomic, Molecular, and Optical Physics; Ionization Potentials of Atoms and Atomic Ions
- ^ a b c d Nucleonica (2007–2013). "Universal Nuclide Chart". Nucleonica: Web Driven Nuclear Science. Retrieved 15 October 2013. (registration required)
- ^ a b c d e f g h Yoshida et al., p. 700–2
- )
- ^ doi:10.2307/4014034. Retrieved 19 October 2013.
- ^ a b c d e f Yoshida et al., pp. 703–4
- ^ a b c d Emsley, pp. 345-347
- ^ a b c d Thompson, Roy C. (1982). "Neptunium: The Neglected Actinide: A Review of the Biological and Environmental Literature". Radiation Research. 90. Radiation Research Society: 1–4. Retrieved 2013-10-15.
- ^ Foster, R.F. (1963). Environmental behavior of chromium and neptunium in Radioecology. New York: Reinhold. p. 569-576.
- ^ a b Atwood, section 4
- ^ Atwood, section 1
- ^ "Human Health Fact Sheet - Neptunium" (PDF). Health Physics Society. 2001. Retrieved 2013-10-15.
- .
- ^ Rhodes, pp. 201-202
- ^ Rhodes, pp. 209-210
- doi:10.1038/133898a0.
- ^ Hoffman, pp. 120-123
- .
- ^ Rhodes, pp. 210-220
- ^ Rhodes, pp. 221-222
- ^ Rhodes, p. 349
- doi:10.1038/134055b0.
- ^ Hoffman, p. 118
- PMID 17798409.
- ^ Fontani, Marco (2005). "The Twilight of the Naturally-Occurring Elements: Moldavium (Ml), Sequanium (Sq) and Dor (Do)". International Conference on the History of Chemistry. Lisbon. pp. 1–8. Archived from the original on 2006-02-24. Retrieved 2013-10-13.
{{cite conference}}: Unknown parameter|booktitle=ignored (|book-title=suggested) (help) - ^ Hulubei, H. (1939). "Nouvelles recherches sur l'élément 93 naturel". Comptes rendus. 209: 476–479.
{{cite journal}}: Unknown parameter|coauthor=ignored (|author=suggested) (help) - )
- ^ Rhodes, pp.264-267
- ^ Rhodes, p. 346
- ^ "The Nobel Prize in Physics 1938". Nobel Foundation. Retrieved 2013-10-13.
- doi:10.1038/143239a0.
- ^ Otto Hahn (1958). "Discovery of fission". Scientific American.
- ^ Segrè, Emilio (1939). "An Unsuccessful Search for Transuranium Elements". Physical Review. 55: 1104–5.
- ^ Rhodes, pp. 346-350
- ^ Yoshida et al, pp. 699-700
- .
- ^ Seaborg, G. T. (1994). "Ch. 118. Origin of the actinide concept". In K.A. Gschneidner, Jr., L, Eyring, G.R. Choppin and G.H. Landet (ed.). Handbook on the Physics and Chemistry of Rare Earths. Vol. 18 – Lanthanides/Actinides: Chemistry. Elsevier. pp. 4–6, 10–14.
{{cite book}}: CS1 maint: multiple names: editors list (link) - ^ Rhodes, pp. 348-350
- ^ Yoshida et al, p. 700
- .
- ^ a b c Burney, G. A; Harbour, R. M; Subcommittee On Radiochemistry, National Research Council (U.S.); Technical Information Center, U.S. Atomic Energy Commission (1974). Radiochemistry of neptunium.
- ISBN 978-87-550-1535-7.
- ^ a b Yoshida et al., p. 710
- ^ "Separated Neptunium 237 and Americium" (PDF). Retrieved 2009-06-06.
- ^ a b http://www.rsc.org/chemistryworld/podcast/interactive_periodic_table_transcripts/neptunium.asp
- ^ a b c Yodshida et al., pp. 704–5
- ^ Yoshida et al., pp. 705–17
- ^ a b c d e f g h i Yoshida et al., pp. 752–4
- ^ a b Yoshida et al., p. 759
- ^ ISBN 978-0-08-037941-8.
- ^ a b c d e f g h i j k Yoshida et al., p. 766–70
- ^ Trygve E Eriksen, Pierre Ndalamba, Daqing Cui, Jordi Bruno, Marco Caceci, Kastriot Spahiu (1993). "Solubility of the redox-sensitive radionuclides 99Tc and 237Np under reducing conditions in neutral to alkaline solutions" (PDF). SKB Technical Report. 93–18: 1–32.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Neck, V., Kim, J. I., and Kanellakopulos, B. (1992). "Solubility and hydrolysis behaviour of neptunium(V)". Radiochim. Acta. 56: 25–30.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Kato, Y., Kimura, T., Yoshida, Z., and Nitani, N. (1996). "Solid-Liquid Phase-Equilibria Of Np(VI) And Of U(VI) Under Controlled CO2 Partial Pressures". Radiochim. Acta. 74: 21–25.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Nikonov, M.V.; Bessonov, A.A.; Krot, N.N.; Perminov, V.P. (1994). "Synthesis and characterization of Np(VI, VII) mixed-valence compound". Radiochemistry. 36: 237–8.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Fahey, J. A., Turcotte, R. P., and Chikalla, T. D. (1976). "Decomposition, stoichiometry and structure of neptunium oxides". Journal of Inorganic Nuclear Chemistry. 38: 495–500.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Yoshida et al., 724-726
- ^ Sharma, B. K. Nuclear and Radiation Chemistry - Transuranium elements. Krishna Prakashan Media. p. 128-9.
{{cite book}}: Cite has empty unknown parameter:|1=(help) - ^ a b Richter K, Sari C. (1987). "Phase relationships in the neptunium-oxygen system". Journal of Nuclear Materials. 148: 266–71.
- ^ Benedict, U., Dabos, S., Dufour, C., and Spirelet, J. C. (1986). "Neptunium compounds under high pressure". Journal of Less Common Metals. 121: 461–68.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Fahey, J. A. (1986). "Neptunium". In Katz, J. J.; Seaborg, G. T.; Morss, L. R. (eds.). The Chemistry of the Actinide Elements. Vol. 1. New York: Chapman & Hall. p. 456.
- ^ a b Yoshida et al, pp. 728-730
- ^ Keller, C., Kock, L., and Walter, K. H. (1965). Journal of Inorganic Nuclear Chemistry. 27: 1205–23.
{{cite journal}}: Missing or empty|title=(help)CS1 maint: multiple names: authors list (link) - doi:10.1021/ic50034a034.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ Morss, L. R., Appelman, E. H., Gerz, R. R., and Martin‐Rovet, D. (1994). "Structural studies of Li5ReO6, Li4NpO5 and Li5NpO6 by neutron and X-ray powder diffraction". Journal of Alloys and Compounds. 203: 289–95.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Keller, C. and Seiffert, H. (1969). Inorganic Nuclear Chemistry Letters. 5: 51–7.
{{cite journal}}: Missing or empty|title=(help)CS1 maint: multiple names: authors list (link) - ^ Awasthi, S. K., Martinot, L., Fuger, J., and Duyckaerts, G. (1971). "Preparation and Characterisation of some Np(VII) Compounds". Inorganic Nuclear Chemistry Letters. 7: 145–51.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Pagès, M., Nectoux, F., and Freundlich, W. (1971). Radiochemical and Radioanalytical Letters. 7: 155–62.
{{cite journal}}: Missing or empty|title=(help)CS1 maint: multiple names: authors list (link) - ^ Mefod’eva, M. P., Krot, N. N., Smirnova, T. V., and Gel’man, A.D (1969). "Oxalate Compounds of Hexavalent Neptunium". Radiokhimiya (in Russian). 11: 193–200.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Hoekstra, H. R. and Gebert, E. (1977). Journal of Inorganic Nuclear Chemistry. 39: 2219–21.
{{cite journal}}: Missing or empty|title=(help)CS1 maint: multiple names: authors list (link) - ^ Tabuteau, A.; Pagès, M. (1985). "Neptunium". In Freeman, A. J.; Keller, C. (eds.). Handbook on the Physics and Chemistry of the Actinides. Vol. 3. Amsterdam: North-Holland. p. 184-241.
{{cite book}}: line feed character in|title=at position 45 (help) - ^ S. Fried, N. R. Davidson: The Basic Dry Chemistry of Neptunium, (1947) Report MDDC-1332, United States Atomic Energy Commission - Argonne National Laboratory, Declassified: July 18, 1947
- ^ a b c Yoshida et al, pp. 730-736
- ^ Kleinschmidt, P. D., Lau, K. H., Hildenbrand, D. L. (1992). "Sublimation studies of NpF4". Journal of Chemical Physics. 97: 1950–3.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Drobyshevskii, Y.V., Serik, V.F., Sokolov, V.B., Tul'skii, M.N. (1978). "Synthesis and some properties of neptunium oxide tetrafluoride and neptunium pentafluoride". Radiokhimiya (in Russian). 20: 238–243.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Seaborg, G. T. and Brown, H. S. (1961) US Patent No. 2,982,604
- ^ Florin, A. E. (1943) Report MUC‐GTS-2165, Declassified: January 23, 1946
- doi:10.1021/j150570a009.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ Fried, S. (1954). "Neptunium". In Seaborg, G. T.; Katz, J. J. (eds.). The Actinide Elements. New York: McGraw-Hill. p. 471.
- ^ Eller, P. G., Asprey, L. B., Kinkead, S. A., Swanson, B. I., and Kissane, R. J. (1998). "Reaction of Dioxygen Difluoride with Neptunium Oxides and Fluorides". Journal of Alloys and Compounds. 269: 63–6.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Kleinschmidt, P. D., Lau, K. H., and Hildenbrand, D. L. (1992). "Sublimation studies of NpO2F2". Journal of Physical Chemistry. 97: 2417–21.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Peacock, R. D. and Edelstein, N. (1976). "Some reactions of neptunium hexafluoride". Journal of Inorganic Nuclear Chemistry. 38: 771–3.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Brown, D. (1972). "Transuranium". Gmelin Handbuck der Anorganischen Chemie, Suppl. Work. Vol. 4. Weinheim, Germany: Verlag Chemie.
- ^ Yoshida et al, pp.736-738
- ^ Fried, S. and Davidson, N. R. (1951) US Patent No. 2,578,416
- ^ Lemire, pp.143-155
- ^ a b c Yoshida et al, pp. 739-742
- ^ Marcon, J. P. (1967) C. R. Acad. Sci. Paris 265 Series C 235
- ^ Fried, S. and Davidson, N. R. (1948). Journal of the American Chemical Society. 70: 3539–47.
{{cite journal}}: Missing or empty|title=(help)CS1 maint: multiple names: authors list (link) - ^ Zachariasen, W. H. (1949). "Crystal chemical studies of the tf-series of elements. X. Sulfides and ox\-sulfides". Acta Crystallographica. 2: 291–6.
- ^ Charvillat, J. P., Benedict, U., Damien, D., Novion, D., Wojakowski, A., and Muller, W. (1976). "Neptunium". In Muller, W.; Linder, R. (eds.). Tranplutonium Elements. Amsterdam: North-Holland. p. 79.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ Peacock, R. D. and Edelstein, N. (1997). "High pressure X-ray diffraction experiments on NpS and PUS up to 60 GPa". High Pressure Research. 15: 387–92.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Thevenin, T.; Jove, J.; Pages, M. (1985). "Crystal chemistry and ^2^3^7Np mossbauer investigations on neptunium oxide chalcogenides NpOS and NpOSe". Materials Research Bulletin. 20: 723–30.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Damien, D. and Berger, R. (1976). Spitsyn, V. I.; Katz, J. J. (eds.). Supplement to Journal of Inorganic Nuclear Chemistry - Moscow Symposium - On the Chemistry of Transuranium Elements. Oxford: Pergamon Press. p. 109-16.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ Thevenin, T.; Pages, M. (1982). "Crystallographic and magnetic studies of a new neptunium selenide: Np2Se5". Journal of Less Common Metals. 84: 133–7.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Damien, D., Wojakowski, A. (1975). "Preparation et parametres de maille des monoseleniures et monotellurures de neptunium et d'americium". Radiochemical and Radioanalytical Letters (in French). 23: 145–54.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b c Yoshida et al, pp. 742-744
- ^ a b c Sheft, I. and Fried, S. (1953). "Neptunium Compounds". Journal of the American Chemical Society. 75: 1236–7.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Olson, W. M. and Mulford R. N. R. (1966). "The melting point and decomposition pressure of neptunium mononitride". Journal of Physical Chemistry. 70: 2932.
- ^ Blaise, A., Damien, D., and Suski, W. (1981). "Electrical resistivity of neptunium mono and diarsenide". Solid State Communications. 37: 659–62.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Dabos, S., Dufour, C., Benedict, U., Spirlet, J. C., and Pages, M. (1986). "High-pressure X-ray diffraction on neptunium compounds: Recent results for NpAs". Physica B. 144: 79–83.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Aldred, A. T., Dunlap, B. D., Harvey, A. R., Lam, D. J., Lander, G. H., and Mueller, M. H. (1974). Physical Review B. 9: 3766–78.
{{cite journal}}: Missing or empty|title=(help)CS1 maint: multiple names: authors list (link) - ^ Burlet, P., Bourdarot, F., Rossat‐ Mignod, J., Sanchez, J. P., Spirlet, J. C., Rebizant, J., and Vogt, O. (1992). "Neutron diffraction study of the magnetic ordering in NpBi". Physica B. 180: 131–2.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ De Novion, C. H. and Lorenzelli, R. (1968). "Proprietes electroniques du monocarbure et du mononitrure de neptunium". Journal of Physics and Chemistry of Solids. 29: 1901–5.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Holley, C. E. Jr, Rand, M. H., and Storms, E. K. (1984). The Chemical Thermodynamics of Actinide Elements and Compounds, part 6. The Actinide Carbides. Vienna: International Atomic Energy Agency. pp. 49–51.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ a b Yoshida et al., pp. 722–4
- ^ Lemire et al., pp. 177-180
- ^ Yoshida et al., pp. 744–5
- ^ a b c d e f g h Yoshida et al., pp. 750–2
- ^ a b c d e f g h i j k Yoshida et al., pp. 745–750
- ^ a b c d e f g Yoshida et al., pp. 771–82
- .
- ^ a b Yoshida et al., pp. 702–3
- ^ "Restricted Data Declassification Decisions from 1946 until Present", accessed Sept 23, 2006
- ^ Cite error: The named reference
critical1was invoked but never defined (see the help page). - ISBN 3-11-014467-0.
- ^ "Yucca Mountain" (PDF). Retrieved 2009-06-06.
- .
- ^ Yarris, Lynn (2005-11-29). "Getting the Neptunium out of Nuclear Waste". Berkeley laboratory, U.S. Department of Energy. Retrieved 05-12-2008.
{{cite web}}: Check date values in:|accessdate=(help) - ^ J. I. Friese; E. C. Buck; B. K. McNamara; B. D. Hanson; S. C. Marschman (January 06-2003). "Existing Evidence for the Fate of Neptunium in the Yucca Mountain Repository" (PDF). Pacific northwest national laboratory, U.S. Department of Energy. Retrieved 05-12-2008.
{{cite web}}: Check date values in:|accessdate=and|date=(help)CS1 maint: multiple names: authors list (link)
Bibliography
Atwood, David A. (2013). Radionuclides in the Enviornment. John Wiley and Sons.
Emsley, John (2011). Nature's Building Blocks: An A-Z Guide to the Elements. New York: Oxford University Press, USA.
Hoffman, Klaus (2001). Otto Hahn: Achievement and Responsibility. Springer.
Lemire, Robert J. (2001). Chemical Thermodynamics of Neptunium and Plutonium. Amsterdam: Elsevier.
Rhodes, Richard (2012). The Making of the Atomic Bomb: 25th Anniversary Edition. New York: Simon & Schuster.
Yoshida, Zenko; Johnson, Stephen G.; Kimura, Takaumi; Krsul, John R. (2006). "Neptunium". In Morss, Lester R.; Edelstein, Norman M.; Fuger, Jean (eds.). The Chemistry of the Actinide and Transactinide Elements (PDF). Vol. 3 (3rd ed.). Dordrecht, the Netherlands: Springer. pp. 699–812. .


![{\displaystyle \mathrm {^{238}_{\ 92}U\ +\ _{0}^{1}n\ \longrightarrow \ _{\ 92}^{239}U\ {\xrightarrow[{23\ min}]{\beta ^{-}}}\ _{\ 93}^{239}Np\ {\xrightarrow[{2.355\ days}]{\beta ^{-}}}\ _{\ 94}^{239}Pu} }](https://wikimedia.org/api/rest_v1/media/math/render/svg/1a5a44653a2a3da3b2f932bc69eab157f2c193c9)
![{\displaystyle \mathrm {^{235}_{\ 92}U\ +\ _{0}^{1}n\ \longrightarrow \ _{\ 92}^{236}U_{m}\ {\xrightarrow[{120\ ns}]{}}\ _{\ 92}^{236}U\ +\ \gamma } }](https://wikimedia.org/api/rest_v1/media/math/render/svg/4048c0b6e9530b699cddcb3f677768354e643a48)
![{\displaystyle \mathrm {^{236}_{\ 92}U\ +\ _{0}^{1}n\ \longrightarrow \ _{\ 92}^{237}U\ {\xrightarrow[{6.75\ d}]{\beta ^{-}}}\ _{\ 93}^{237}Np} }](https://wikimedia.org/api/rest_v1/media/math/render/svg/787533788d653792c7d5cb842d045a094de89902)
![{\displaystyle \mathrm {^{237}_{\ 93}Np\ +\ _{0}^{1}n\ \longrightarrow \ _{\ 93}^{238}Np\ {\xrightarrow[{2.117\ d}]{\beta ^{-}}}\ _{\ 94}^{238}Pu} }](https://wikimedia.org/api/rest_v1/media/math/render/svg/b3a8a7663dc2ada26e26f0fc9b7f4bf751f8ae34)