Anabolic steroid
| Anabolic–androgenic steroids | ||
|---|---|---|
Chemical class Steroids; Androstanes; Estranes | | |
| Clinical data | ||
| Drugs.com | Drug Classes | |
| External links | ||
| MeSH | D045165 | |
| Legal status | ||
| Legal status |
| |
| In Wikidata | ||
Anabolic steroids, also known as anabolic-androgenic steroids (AAS), are a class of drugs that are structurally related to testosterone, the main male sex hormone, and produce effects by binding to the androgen receptor. Anabolic steroids have a number of medical uses,[1] but are also used by athletes to increase muscle size, strength, and performance.
Health risks can be produced by long-term use or excessive doses of AAS.
Uses
Medical

Since the discovery and synthesis of testosterone in the 1930s, AAS have been used by physicians for many purposes, with varying degrees of success. These can broadly be grouped into anabolic, androgenic, and other uses.
Anabolic
- growth hormone, which has fewer side effects, makes this a secondary treatment.
- Stimulation of
- Stimulation of lean body mass and prevention of insulin sensitivity, or quality of life.[17]
- Prevention or treatment of
- Aiding
- Counteracting the catabolic effect of long-term corticosteroid therapy.[22][23]
- Oxandrolone improves both short-term and long-term outcomes in people recovering from severe burns, and is well-established as a safe treatment for this indication.[24][25]
- Treatment of idiopathic short stature, hereditary angioedema, alcoholic hepatitis, and hypogonadism.[26][27]
- Methyltestosterone is used in the treatment of
Androgenic
- Androgen replacement therapy for men with low levels of testosterone, such as those associated with late-onset hypogonadism;[31] also effective in improving libido for elderly males.[32][33][34][35]
- Induction of male puberty: Androgens are given to many boys distressed about extreme delay of puberty. Testosterone is now nearly the only androgen used for this purpose and has been shown to increase height, weight, and fat-free mass in boys with delayed puberty.[36]
Other
- Treatment of breast cancer in women, although they are now very rarely used for this purpose due to their marked virilizing side effects.[42][18][43]
- In low doses as a component of transgender women, for instance to increase energy, well-being, libido, and quality of life, as well as to reduce hot flashes.[44][45][46][47] Testosterone is usually used for this purpose, although methyltestosterone is also used.[47][48]
- Male hormonal contraception; currently experimental, but potential for use as effective, safe, reliable, and reversible male contraceptives.[49]
- Assistant in the treatment of vasodilators, which can significantly improve bloodflow in individuals prone to vasoconstriction.[50]
Enhancing performance

Most steroid users are not athletes.[51] In the United States, between 1 million and 3 million people (1% of the population) are thought to have used AAS.[52] Studies in the United States have shown that AAS users tend to be mostly middle-class men with a median age of about 25 who are noncompetitive bodybuilders and non-athletes and use the drugs for cosmetic purposes.[53] "Among 12- to 17-year-old boys, use of steroids and similar drugs jumped 25 percent from 1999 to 2000, with 20 percent saying they use them for looks rather than sports, a study by insurer Blue Cross Blue Shield found."(Eisenhauer) Another study found that non-medical use of AAS among college students was at or less than 1%.[54] According to a recent survey, 78.4% of steroid users were noncompetitive bodybuilders and non-athletes, while about 13% reported unsafe injection practices such as reusing needles, sharing needles, and sharing multidose vials,[55] though a 2007 study found that sharing of needles was extremely uncommon among individuals using AAS for non-medical purposes, less than 1%.[56] Another 2007 study found that 74% of non-medical AAS users had post-secondary degrees and more had completed college and fewer had failed to complete high school than is expected from the general populace.[56] The same study found that individuals using AAS for non-medical purposes had a higher employment rate and a higher household income than the general population.[56] AAS users tend to research the drugs they are taking more than other controlled-substance users;[citation needed] however, the major sources consulted by steroid users include friends, non-medical handbooks, internet-based forums, blogs, and fitness magazines, which can provide questionable or inaccurate information.[57]
AAS users tend to be unhappy with the portrayal of AAS as deadly in the media and in politics.[58] According to one study, AAS users also distrust their physicians and in the sample 56% had not disclosed their AAS use to their physicians.[59] Another 2007 study had similar findings, showing that, while 66% of individuals using AAS for non-medical purposes were willing to seek medical supervision for their steroid use, 58% lacked trust in their physicians, 92% felt that the medical community's knowledge of non-medical AAS use was lacking, and 99% felt that the public has an exaggerated view of the side-effects of AAS use.[56] A recent study has also shown that long term AAS users were more likely to have symptoms of muscle dysmorphia and also showed stronger endorsement of more conventional male roles.[60] A recent study in the Journal of Health Psychology showed that many users believed that steroids used in moderation were safe.[61]
AAS have been used by men and women in many different kinds of professional sports to attain a competitive edge or to assist in recovery from injury. These sports include bodybuilding, weightlifting, shot put and other track and field, cycling, baseball, wrestling, mixed martial arts, boxing, football, and cricket. Such use is prohibited by the rules of the governing bodies of most sports. AAS use occurs among adolescents, especially by those participating in competitive sports. It has been suggested that the prevalence of use among high-school students in the U.S. may be as high as 2.7%.[62]
Dosages
| Medication | Route | Dosage range[a] | ||
|---|---|---|---|---|
| Danazol | Oral | 100–800 mg/day | ||
| Drostanolone propionate | Injection | 100 mg 3 times/week | ||
| Ethylestrenol | Oral | 2–8 mg/day | ||
| Fluoxymesterone | Oral | 2–40 mg/day | ||
| Mesterolone | Oral | 25–150 mg/day | ||
| Metandienone | Oral | 2.5–15 mg/day | ||
| Metenolone acetate | Oral | 10–150 mg/day | ||
| Metenolone enanthate | Injection | 25–100 mg/week | ||
| Methyltestosterone | Oral | 1.5–200 mg/day | ||
| Nandrolone decanoate | Injection | 12.5–200 mg/week[b] | ||
| Nandrolone phenylpropionate | Injection | 6.25–200 mg/week[b] | ||
| Norethandrolone | Oral | 20–30 mg/day | ||
| Oxandrolone | Oral | 2.5–20 mg/day | ||
| Oxymetholone | Oral | 1–5 mg/kg/day or 50–150 mg/day | ||
| Stanozolol | Oral | 2–6 mg/day | ||
| Injection | 50 mg up to every two weeks | |||
| Testosterone | Oral[c] | 400–800 mg/day[b] | ||
| Injection | 25–100 mg up to three times weekly | |||
| Testosterone cypionate | Injection | 50–400 mg up to every four weeks | ||
| Testosterone enanthate | Injection | 50–400 mg up to every four weeks | ||
| Testosterone propionate | Injection | 25–50 mg up to three times weekly | ||
| Testosterone undecanoate | Oral | 80–240 mg/day[b] | ||
| Injection | 750–1000 mg up to every 10 weeks | |||
| Trenbolone HBC | Injection | 75 mg every 10 days | ||
| Sources: [63][64][65][66][18][67][68][69][70][71] | ||||
Available forms
The AAS that have been used most commonly in medicine are
Routes of administration

There are four common forms in which AAS are administered: oral pills; injectable steroids; creams/gels for topical application; and skin patches. Oral administration is the most convenient. Testosterone administered by mouth is rapidly absorbed, but it is largely converted to inactive metabolites, and only about one-sixth is available in active form. In order to be sufficiently active when given by mouth, testosterone derivatives are alkylated at the 17α position, e.g. methyltestosterone and fluoxymesterone. This modification reduces the liver's ability to break down these compounds before they reach the systemic circulation.
Testosterone can be administered
Transdermal patches (adhesive patches placed on the skin) may also be used to deliver a steady dose through the skin and into the bloodstream. Testosterone-containing creams and gels that are applied daily to the skin are also available, but absorption is inefficient (roughly 10%, varying between individuals) and these treatments tend to be more expensive. Individuals who are especially physically active and/or bathe often may not be good candidates, since the medication can be washed off and may take up to six hours to be fully absorbed. There is also the risk that an intimate partner or child may come in contact with the application site and inadvertently dose themselves; children and women are highly sensitive to testosterone and can develop unintended masculinization and health effects, even from small doses. Injection is the most common method used by individuals administering AAS for non-medical purposes.[56]
The traditional routes of administration do not have differential effects on the efficacy of the drug. Studies indicate that the anabolic properties of AAS are relatively similar despite the differences in pharmacokinetic principles such as
Adverse effects
Known possible side effects of AAS include:[6][77][78][79][80]
- fluid retention/edema.
- Reproductive/endocrine: libido changes, reversible infertility, hypogonadotropic hypogonadism.
- Male-specific: prostate hypertrophy, prostate cancer.
- Female-specific: teratogenicity (in female fetuses).
- Child-specific: premature contrasexual precocityin girls.
- Musculoskeletal: muscle hypertrophy, muscle strains, tendon ruptures, rhabdomyolysis.
- hepatic steatosis, hepatocellular adenoma, hepatocellular carcinoma, cholestasis, peliosis hepatis; all mostly or exclusively with 17α-alkylated AAS.[85]
- acute renal failure (secondary to rhabdomyolysis), focal segmental glomerulosclerosis, renal cell carcinoma.
- Others: immune dysfunction.[86]
Physiological
Depending on the length of drug use, there is a chance that the immune system can be damaged. Most of these side-effects are dose-dependent, the most common being elevated blood pressure, especially in those with pre-existing hypertension.[87] In addition to morphological changes of the heart which may have a permanent adverse effect on cardiovascular efficiency.
AAS have been shown to alter fasting blood sugar and glucose tolerance tests.
A number of severe side effects can occur if adolescents use AAS. For example, AAS may prematurely stop the lengthening of bones (premature
Cancer
WHO organization
Cardiovascular
Other side-effects can include alterations in the structure of the
AAS use can cause harmful changes in cholesterol levels: Some steroids cause an increase in LDL cholesterol and a decrease in HDL cholesterol.[100]
Growth defects
AAS use in adolescents quickens bone maturation and may reduce adult height in high doses.[citation needed] Low doses of AAS such as oxandrolone are used in the treatment of idiopathic short stature, but this may only quicken maturation rather than increasing adult height.[101]
Feminization

Although all anabolic steroids have
Prolonged use of androgenic-anabolic steroids by men results in temporary shut down of their natural testosterone production due to an inhibition of the hypothalamic–pituitary–gonadal axis. This manifests in testicular atrophy, inhibition of the production of sperm, sexual function and infertility.[103][104][105] A short (1–2 months) use of androgenic-anabolic steroids by men followed by a course of testosterone-boosting therapy (e.g. clomifene and human chorionic gonadotropin) usually results in return to normal testosterone production.[106])
Masculinization
Female-specific side effects include
Kidney problems
Kidney tests revealed that nine of the ten steroid users developed a condition called focal segmental glomerulosclerosis, a type of scarring within the kidneys. The kidney damage in the bodybuilders has similarities to that seen in morbidly obese patients, but appears to be even more severe.[109]
Liver problems
High doses of oral AAS compounds can cause liver damage.[3] Peliosis hepatis has been increasingly recognised with the use of AAS.
Neuropsychiatric

A 2005 review in
Diagnostic Statistical Manual assertion
Personality profiles
Cooper, Noakes, Dunne, Lambert, and Rochford identified that AAS-using individuals are more likely to score higher on
Mood and anxiety
Affective disorders have long been recognised as a complication of AAS use. Case reports describe both hypomania and mania, along with irritability, elation, recklessness, racing thoughts and feelings of power and invincibility that did not meet the criteria for mania/hypomania.[117] Of 53 bodybuilders who used AAS, 27 (51%) reported unspecified mood disturbance.[118]
Aggression and hypomania
From the mid-1980s onward, the media reported "roid rage" as a side effect of AAS.[119]: 23
A 2005 review determined that some, but not all, randomized controlled studies have found that AAS use correlates with hypomania and increased aggressiveness, but pointed out that attempts to determine whether AAS use triggers violent behavior have failed, primarily because of high rates of non-participation.[120] A 2008 study on a nationally representative sample of young adult males in the United States found an association between lifetime and past-year self-reported AAS use and involvement in violent acts. Compared with individuals that did not use steroids, young adult males that used AAS reported greater involvement in violent behaviors even after controlling for the effects of key demographic variables, previous violent behavior, and polydrug use.[121] A 1996 review examining the blind studies available at that time also found that these had demonstrated a link between aggression and steroid use, but pointed out that with estimates of over one million past or current steroid users in the United States at that time, an extremely small percentage of those using steroids appear to have experienced mental disturbance severe enough to result in clinical treatments or medical case reports.[122]
The relationship between AAS use and depression is inconclusive. A 1992 review[needs update] found that AAS may both relieve and cause depression, and that cessation or diminished use of AAS may also result in depression, but called for additional studies due to disparate data.[123]
Reproductive
AAS consumption disrupts the
Pharmacology
Mechanism of action

The pharmacodynamics of AAS are unlike peptide hormones. Water-soluble peptide hormones cannot penetrate the fatty cell membrane and only indirectly affect the nucleus of target cells through their interaction with the cell's surface receptors. However, as fat-soluble hormones, AAS are membrane-permeable and influence the nucleus of cells by direct action. The pharmacodynamic action of AAS begin when the exogenous hormone penetrates the membrane of the target cell and binds to an androgen receptor (AR) located in the cytoplasm of that cell. From there, the compound hormone-receptor diffuses into the nucleus, where it either alters the expression of genes[129] or activates processes that send signals to other parts of the cell.[130] Different types of AAS bind to the AAR with different affinities, depending on their chemical structure.[7]
The effect of AAS on muscle mass is caused in at least two ways:
Molecular Interaction of AAS with Androgen Receptors
Anabolic steroids interact with ARs across various tissues, including muscle, bone, and reproductive systems.[133] Upon binding to the AR, anabolic steroids trigger a translocation of the hormone-receptor complex to the cell nucleus, where they either alter gene expression or activate cellular signaling pathways; this results in increased protein synthesis, enhanced muscle growth, and reduced muscle catabolism.[134]
Anabolic steroids influence cellular differentiation while favoring the development of muscle cells over fat-storage cells.[135] Research in this field has shown that structural modifications in anabolic steroids are critical in determining their binding affinity to ARs and their resulting anabolic and androgenic activities.[79] These modifications affect a steroid's ability to influence gene expression and cellular processes, highlighting the complex biophysical interactions of anabolic steroids at the cellular level.[133]
Anabolic and androgenic effects
| Medication | Ratioa |
|---|---|
| Testosterone | ~1:1 |
| Androstanolone (DHT) | ~1:1 |
| Methyltestosterone | ~1:1 |
| Methandriol | ~1:1 |
| Fluoxymesterone | 1:1–1:15 |
| Metandienone | 1:1–1:8 |
| Drostanolone | 1:3–1:4 |
| Metenolone | 1:2–1:30 |
| Oxymetholone | 1:2–1:9 |
| Oxandrolone | 1:3–1:13 |
| Stanozolol | 1:1–1:30 |
| Nandrolone | 1:3–1:16 |
| Ethylestrenol | 1:2–1:19 |
| Norethandrolone | 1:1–1:20 |
| Notes: In rodents. Footnotes: a = Ratio of androgenic to anabolic activity. Sources: See template. | |
As their name suggests, AAS have two different, but overlapping, types of effects: anabolic, meaning that they promote anabolism (cell growth), and androgenic (or virilizing), meaning that they affect the development and maintenance of masculine characteristics.
Some examples of the anabolic effects of these hormones are increased
The androgenic effects of AAS are numerous. Depending on the length of use, the side effects of the steroid can be irreversible. Processes affected include pubertal growth,
A commonly used protocol for determining the androgenic:anabolic ratio, dating back to the 1950s, uses the relative weights of ventral
Body composition and strength improvements
Anabolic steroids notably influence muscle fiber characteristics, affecting both the size and type of muscle fibers. This alteration significantly contributes to enhanced muscle strength and endurance.[141] Anabolic-androgenic steroids (AAS) cause these changes by directly impacting the muscle tissue's cellular components. Studies have shown that these changes are not merely superficial but represent a profound transformation in the muscle's structural and functional properties. This transformation is a key factor in the steroids' ability to enhance physical performance and endurance.[142]
Body weight in men may increase by 2 to 5 kg as a result of short-term (<10 weeks) AAS use, which may be attributed mainly to an increase of lean mass. Animal studies also found that fat mass was reduced, but most studies in humans failed to elucidate significant fat mass decrements. The effects on lean body mass have been shown to be dose-dependent. Both
The upper region of the body (thorax, neck, shoulders, and upper arm) seems to be more susceptible for AAS than other body regions because of predominance of ARs in the upper body.[
Strength improvements in the range of 5 to 20% of baseline strength, depending largely on the drugs and dose used as well as the administration period. Overall, the exercise where the most significant improvements were observed is the bench press.[7] For almost two decades, it was assumed that AAS exerted significant effects only in experienced strength athletes.[143][144] A randomized controlled trial demonstrated, however, that even in novice athletes a 10-week strength training program accompanied by testosterone enanthate at 600 mg/week may improve strength more than training alone does.[7][145] This dose is sufficient to significantly improve lean muscle mass relative to placebo even in subjects that did not exercise at all.[145] The anabolic effects of testosterone enanthate were highly dose dependent.[7][146]
Dissociation of effects
Endogenous/natural AAS like testosterone and DHT and synthetic AAS mediate their effects by binding to and activating the AR.
The measurement of the dissociation between anabolic and androgenic effects among AAS is based largely on a simple but outdated and unsophisticated model using rat tissue bioassays.
Intracellular metabolism
Testosterone is
Natural AAS like testosterone and DHT and synthetic AAS are analogues and are very similar structurally.[73] For this reason, they have the capacity to bind to and be metabolized by the same steroid-metabolizing enzymes.[73] According to the intracellular metabolism explanation, the androgenic-to-anabolic ratio of a given AR agonist is related to its capacity to be transformed by the aforementioned enzymes in conjunction with the AR activity of any resulting products.[73] For instance, whereas the AR activity of testosterone is greatly potentiated by local conversion via 5α-reductase into DHT in tissues where 5α-reductase is expressed, an AAS that is not metabolized by 5α-reductase or is already 5α-reduced, such as DHT itself or a derivative (like mesterolone or drostanolone), would not undergo such potentiation in said tissues.[73] Moreover, nandrolone is metabolized by 5α-reductase, but unlike the case of testosterone and DHT, the 5α-reduced metabolite of nandrolone has much lower affinity for the AR than does nandrolone itself, and this results in reduced AR activation in 5α-reductase-expressing tissues.[73] As so-called "androgenic" tissues such as skin/hair follicles and male reproductive tissues are very high in 5α-reductase expression, while skeletal muscle is virtually devoid of 5α-reductase, this may primarily explain the high myotrophic–androgenic ratio and dissociation seen with nandrolone, as well as with various other AAS.[73]
Aside from 5α-reductase, aromatase may inactivate testosterone signaling in skeletal muscle and adipose tissue, so AAS that lack aromatase affinity, in addition to being free of the potential side effect of gynecomastia, might be expected to have a higher myotrophic–androgenic ratio in comparison.[73] In addition, DHT is inactivated by high activity of 3α-HSD in skeletal muscle (and cardiac tissue), and AAS that lack affinity for 3α-HSD could similarly be expected to have a higher myotrophic–androgenic ratio (although perhaps also increased long-term cardiovascular risks).[73] In accordance, DHT, mestanolone (17α-methyl-DHT), and mesterolone (1α-methyl-DHT) are all described as very poorly anabolic due to inactivation by 3α-HSD in skeletal muscle, whereas other DHT derivatives with other structural features like metenolone, oxandrolone, oxymetholone, drostanolone, and stanozolol are all poor substrates for 3α-HSD and are described as potent anabolics.[68]
The intracellular metabolism theory explains how and why remarkable dissociation between anabolic and androgenic effects might occur despite the fact that these effects are mediated through the same signaling receptor, and why this dissociation is invariably incomplete.
| Compound | rAR (%) | hAR (%) | ||||||
|---|---|---|---|---|---|---|---|---|
| Testosterone | 38 | 38 | ||||||
| 5α-Dihydrotestosterone | 77 | 100 | ||||||
| Nandrolone | 75 | 92 | ||||||
| 5α-Dihydronandrolone | 35 | 50 | ||||||
| Ethylestrenol | ND | 2 | ||||||
| Norethandrolone | ND | 22 | ||||||
| 5α-Dihydronorethandrolone | ND | 14 | ||||||
| Metribolone | 100 | 110 | ||||||
| Sources: See template. | ||||||||
Functional selectivity
An animal study found that two different kinds of
Non-genomic mechanisms
Testosterone signals not only through the nuclear AR, but also through mARs, including
Antigonadotropic effects
Changes in endogenous testosterone levels may also contribute to differences in myotrophic–androgenic ratio between testosterone and synthetic AAS.
GABAA receptor modulation
Some AAS, such as testosterone, DHT, stanozolol, and methyltestosterone, have been found to modulate the
Comparison of AAS
AAS differ in a variety of ways including in their capacities to be
| Compound | Class | 5α-R | AROM | 3-HSD | AAR | Estr | Prog |
Oral | Hepat |
|---|---|---|---|---|---|---|---|---|---|
| Androstanolone | DHT | – | – | + | * | – | – | – | – |
| Boldenone | T | – | ± | – | ** | ± | – | – | – |
| Drostanolone | DHT | – | – | – | *** | – | – | – | – |
| Ethylestrenol | 19-NT; 17α-A | + (↓) | ± | – | *** | + | + | + | + |
| Fluoxymesterone | T; 17α-A | + (↑) | – | – | * | – | – | + | + |
| Mestanolone | DHT; 17α-A | – | – | + | * | – | – | + | + |
| Mesterolone | DHT | – | – | + | * | – | – | ± | – |
| Metandienone | T; 17α-A | – | ± | – | ** | + | – | + | + |
| Metenolone | DHT | – | – | – | ** | – | – | ± | – |
| Methyltestosterone | T; 17α-A | + (↑) | + | – | * | + | – | + | + |
| Nandrolone | 19-NT | + (↓) | ± | – | *** | ± | + | – | – |
| Norethandrolone | 19-NT; 17α-A | + (↓) | ± | – | *** | + | + | + | + |
| Oxandrolone | DHT; 17α-A | – | – | – | *** | – | – | + | ± |
| Oxymetholone | DHT; 17α-A | – | – | – | *** | + | – | + | + |
| Stanozolol | DHT; 17α-A | – | – | – | *** | – | – | + | + |
| Testosterone | T | + (↑) | + | – | * | + | – | ±a | – |
| Trenbolone | 19-NT | – | – | – | *** | – | + | – | – |
| Key: + = Yes. ± = Low. – = No. ↑ = Potentiated. ↓ = Inactivated. *** = High. ** = Moderate. * = Low. Abbreviations: 5α-R = Metabolized by Progestogenic. Oral = Oral activity. Hepat = Hepatotoxicity. Footnotes: a = As testosterone undecanoate . Sources: See template.
| |||||||||
| Steroid | Chemical name | Relative binding affinities (%)
| ||||||
|---|---|---|---|---|---|---|---|---|
| PR | AR | ER | GR | MR | SHBG | CBG
| ||
| Androstanolone | DHT | 1.4–1.5 | 60–120 | <0.1 | <0.1–0.3 | 0.15 | 100 | 0.8 |
| Boldenone | Δ1-T | <1 | 50–75 | ? | <1 | ? | ? | ? |
| Danazol | 2,3-Isoxazol-17α-Ety-T | 9 | 8 | ? | <0.1a | ? | 8 | 10 |
| Dienolone | ∆9-19-NT | 17 | 134 | <0.1 | 1.6 | 0.3 | ? | ? |
| Dimethyldienolone | ∆9-7α,17α-DiMe-19-NT | 198 | 122 | 0.1 | 6.1 | 1.7 | ? | ? |
| Dimethyltrienolone | ∆9,11-7α,17α-DiMe-19-NT | 306 | 180 | 0.1 | 22 | 52 | ? | ? |
| Drostanolone | 2α-Me-DHT | ? | ? | ? | ? | ? | 39 | ? |
| Ethisterone | 17α-Ety-T | 35 | 0.1 | <1.0 | <1.0 | <1.0 | 25–92 | 0.3 |
| Ethylestrenol | 3-DeO-17α-Et-19-NT | ? | ? | ? | ? | ? | <1 | ? |
| Fluoxymesterone | 9α-F-11β-OH-17α-Me-T | ? | ? | ? | ? | ? | ≤3 | ? |
| Gestrinone | ∆9,11-17α-Ety-18-Me-19-NT | 75–76 | 83–85 | <0.1–10 | 77 | 3.2 | ? | ? |
| Levonorgestrel | 17α-Ety-18-Me-19-NT | 170 | 84–87 | <0.1 | 14 | 0.6–0.9 | 14–50 | <0.1 |
| Mestanolone | 17α-Me-DHT | 5–10 | 100–125 | ? | <1 | ? | 84 | ? |
| Mesterolone | 1α-Me-DHT | ? | ? | ? | ? | ? | 82–440 | ? |
| Metandienone | ∆1-17α-Me-T | ? | ? | ? | ? | ? | 2 | ? |
| Metenolone | ∆1-1-Me-DHT | ? | ? | ? | ? | ? | 3 | ? |
| Methandriol | 17α-Me-A5 | ? | ? | ? | ? | ? | 40 | ? |
| Methasterone | 2α,17α-DiMe-DHT | ? | ? | ? | ? | ? | 58 | ? |
| Methyldienolone | ∆9-17α-Me-19-NT | 71 | 64 | <0.1 | 6 | 0.4 | ? | ? |
| Methyltestosterone | 17α-Me-T | 3 | 45–125 | <0.1 | 1–5 | ? | 5–64 | <0.1 |
| Methyl-1-testosterone | ∆1-17α-Me-DHT | ? | ? | ? | ? | ? | 69 | ? |
| Metribolone | ∆9,11-17α-Me-19-NT | 208–210 | 199–210 | <0.1 | 10–26 | 18 | 0.2–0.8 | ≤0.4 |
| Mibolerone | 7α,17α-DiMe-19-NT | 214 | 108 | <0.1 | 1.4 | 2.1 | 6 | ? |
| Nandrolone | 19-NT | 20 | 154–155 | <0.1 | 0.5 | 1.6 | 1–16 | 0.1 |
| Norethandrolone | 17α-Et-19-NT | ? | ? | ? | ? | ? | 3 | ? |
| Norethisterone | 17α-Ety-19-NT | 155–156 | 43–45 | <0.1 | 2.7–2.8 | 0.2 | 5–21 | 0.3 |
| Norgestrienone | ∆9,11-17α-Ety-19-NT | 63–65 | 70 | <0.1 | 11 | 1.8 | ? | ? |
| Normethandrone | 17α-Me-19-NT | 100 | 146 | <0.1 | 1.5 | 0.6 | 7 | ? |
| Oxandrolone | 2-Oxa-17α-Me-DHT | ? | ? | ? | ? | ? | <1 | ? |
| Oxymetholone | 2-OHMeEne-17α-Me-DHT | ? | ? | ? | ? | ? | ≤3 | ? |
| RU-2309 (17α-Me-THG) | ∆9,11-17α,18-DiMe-19-NT | 230 | 143 | <0.1 | 155 | 36 | ? | ? |
| Stanozolol | 2,3-Pyrazol-17α-Me-DHT | ? | ? | ? | ? | ? | 1–36 | ? |
| Testosterone | T | 1.0–1.2 | 100 | <0.1 | 0.17 | 0.9 | 19–82 | 3–8 |
| 1-Testosterone | ∆1-DHT | ? | ? | ? | ? | ? | 98 | ? |
| Tibolone | 7α-Me-17α-Ety-19-N-5(10)-T | 12 | 12 | 1 | ? | ? | ? | ? |
| Δ4-Tibolone | 7α-Me-17α-Ety-19-NT | 180 | 70 | 1 | <1 | 2 | 1–8 | <1 |
| Trenbolone | ∆9,11-19-NT | 74–75 | 190–197 | <0.1 | 2.9 | 1.33 | ? | ? |
| Trestolone | 7α-Me-19-NT | 50–75 | 100–125 | ? | <1 | ? | 12 | ? |
| Notes: Values are percentages (%). Reference CBG . Footnotes: a = 1-hour incubation time (4 hours is standard for this assay; may affect affinity value). Sources: See template.
| ||||||||
| Medication | Form | Major brand names | Duration |
|---|---|---|---|
| Testosterone | Aqueous suspension | Andronaq, Sterotate, Virosterone | 2–3 days |
| Testosterone propionate | Oil solution | Androteston, Perandren, Testoviron | 3–4 days |
| Testosterone phenylpropionate | Oil solution | Testolent | 8 days |
| Testosterone isobutyrate | Aqueous suspension | Agovirin Depot, Perandren M | 14 days |
| Mixed testosterone estersa | Oil solution | Triolandren | 10–20 days |
| Mixed testosterone estersb | Oil solution | Testosid Depot | 14–20 days |
| Testosterone enanthate | Oil solution | Delatestryl | 14–28 days |
| Testosterone cypionate | Oil solution | Depovirin | 14–28 days |
| Mixed testosterone estersc | Oil solution | Sustanon 250 | 28 days |
| Testosterone undecanoate | Oil solution | Aveed, Nebido | 100 days |
| Testosterone buciclated | Aqueous suspension | 20 Aet-1, CDB-1781e | 90–120 days |
| Nandrolone phenylpropionate | Oil solution | Durabolin | 10 days |
| Nandrolone decanoate | Oil solution | Deca Durabolin | 21–28 days |
| Methandriol | Aqueous suspension | Notandron, Protandren | 8 days |
| Methandriol bisenanthoyl acetate | Oil solution | Notandron Depot | 16 days |
| Metenolone acetate | Oil solution | Primobolan | 3 days |
| Metenolone enanthate | Oil solution | Primobolan Depot | 14 days |
| Note: All are via i.m. injection. Footnotes: a = TP, TV, and TUe. b = TP and TKL. c = TP, TPP, TiCa, and TD. d = Studied but never marketed. e = Developmental code names. Sources: See template. | |||
| Testosterone ester | Form | Route | Tmax |
t1/2 |
MRT
|
|---|---|---|---|---|---|
| Testosterone undecanoate | Oil-filled capsules | Oral | ? | 1.6 hours | 3.7 hours |
| Testosterone propionate | Oil solution | Intramuscular injection | ? | 0.8 days | 1.5 days |
| Testosterone enanthate | Castor oil solution | Intramuscular injection | 10 days | 4.5 days | 8.5 days |
| Testosterone undecanoate | Tea seed oil solution | Intramuscular injection | 13.0 days | 20.9 days | 34.9 days |
| Testosterone undecanoate | Castor oil solution | Intramuscular injection | 11.4 days | 33.9 days | 36.0 days |
| Testosterone buciclatea | Aqueous suspension | Intramuscular injection | 25.8 days | 29.5 days | 60.0 days |
| Notes: Testosterone cypionate has similar pharmacokinetics to Testosterone enanthate. Footnotes: a = Never marketed. Sources: See template. | |||||
5α-Reductase and androgenicity
Testosterone can be robustly converted by
The capacity to be metabolized by 5α-reductase and the AR activity of the resultant metabolites appears to be one of the major, if not the most important determinant of the
Aromatase and estrogenicity
Testosterone can be
The major effect of estrogenicity is
Progestogenic activity
Many 19-nortestosterone derivatives, including nandrolone, trenbolone, ethylestrenol (ethylnandrol), metribolone (R-1881), trestolone, 11β-MNT, dimethandrolone, and others, are potent agonists of the progesterone receptor (PR) and hence are progestogens in addition to AAS.[73][175] Similarly to the case of estrogenic activity, the progestogenic activity of these drugs serves to augment their antigonadotropic activity.[175] This results in increased potency and effectiveness of these AAS as antispermatogenic agents and male contraceptives (or, put in another way, increased potency and effectiveness in producing azoospermia and reversible male infertility).[175]
Oral activity and hepatotoxicity
Non-17α-alkylated testosterone derivatives such as testosterone itself, DHT, and nandrolone all have poor oral
In addition to oral activity, 17α-alkylation also confers a high potential for
Aside from prohormones and testosterone undecanoate, almost all orally active AAS are 17α-alkylated.[185] A few AAS that are not 17α-alkylated are orally active.[73] Some examples include the testosterone 17-ethers cloxotestosterone, quinbolone, and silandrone,[citation needed] which are prodrugs (to testosterone, boldenone (Δ1-testosterone), and testosterone, respectively), the DHT 17-ethers mepitiostane, mesabolone, and prostanozol (which are also prodrugs), the 1-methylated DHT derivatives mesterolone and metenolone (although these are relatively weak AAS),[73][72] and the 19-nortestosterone derivatives dimethandrolone and 11β-MNT, which have improved resistance to first-pass hepatic metabolism due to their 11β-methyl groups (in contrast to them, the related AAS trestolone (7α-methyl-19-nortestosterone) is not orally active).[73][175] As these AAS are not 17α-alkylated, they show minimal potential for hepatotoxicity.[73]
Neurosteroid activity
DHT, via its metabolite
Chemistry
AAS are
- 17α-Alkylation: methyltestosterone, metandienone, fluoxymesterone, oxandrolone, oxymetholone, stanozolol, norethandrolone, ethylestrenol
- 19-Demethylation: nandrolone, trenbolone, norethandrolone, ethylestrenol, trestolone, dimethandrolone
- 5α-Reduction: androstanolone, drostanolone, mestanolone, mesterolone, metenolone, oxandrolone, oxymetholone, stanozolol
- 3β- and/or 17β-esterification: testosterone enanthate, nandrolone decanoate, drostanolone propionate, boldenone undecylenate, trenbolone acetate
As well as others such as
| Classes | Androgen | Structure | Chemical name | Features |
|---|---|---|---|---|
| Testosterone | 4-Hydroxytestosteronea | 4-Hydroxytestosterone | – | |
| Androstenediola | 5-Androstenediol (androst-5-ene-3β,17β-diol) | Prohormone | ||
| Androstenedionea | 4-Androstenedione (androst-4-ene-3,17-dione) | Prohormone | ||
| Boldenone | 1-Dehydrotestosterone | – | ||
| Boldionea | 1-Dehydro-4-androstenedione | Prohormone | ||
| Clostebol | 4-Chlorotestosterone | – | ||
| Cloxotestosterone | Testosterone 17-chloral hemiacetal ether | Ether | ||
| Prasterone | 5-Dehydroepiandrosterone (androst-5-en-3β-ol-17-one) | Prohormone | ||
| Quinbolone | 1-Dehydrotestosterone 17β-cyclopentenyl enol ether | Ether | ||
| Silandronea | Testosterone 17β-trimethylsilyl ether | Ether | ||
| Testosterone | Androst-4-en-17β-ol-3-one | – | ||
| 17α-Alkylated testosterone | Bolasterone | 7α,17α-Dimethyltestosterone | – | |
| Calusterone | 7β,17α-Dimethyltestosterone | – | ||
| Chlorodehydromethylandrostenediola | 1-Dehydro-4-chloro-17α-methyl-4-androstenediol | Prohormone | ||
| Chlorodehydromethyltestosterone | 1-Dehydro-4-chloro-17α-methyltestosterone | – | ||
| Chloromethylandrostenediola | 4-Chloro-17α-methyl-4-androstenediol | – | ||
| Enestebola | 1-Dehydro-4-hydroxy-17α-methyltestosterone | – | ||
| Ethyltestosteronea | 17α-Ethyltestosterone | – | ||
| Fluoxymesterone | 9α-Fluoro-11β-hydroxy-17α-methyltestosterone | – | ||
| Formebolone | 1-Dehydro-2-formyl-11α-hydroxy-17α-methyltestosterone | – | ||
| Hydroxystenozolea | 17α-Methyl-2'H-androsta-2,4-dieno[3,2-c]pyrazol-17β-ol | Ring-fused | ||
| Metandienone | 1-Dehydro-17α-methyltestosterone | – | ||
| Methandriol | 17α-Methyl-5-androstenediol | Prohormone | ||
| Methylclostebola | 4-Chloro-17α-methyltestosterone | – | ||
| Methyltestosterone | 17α-Methyltestosterone | – | ||
Methyltestosterone hexyl ether
|
17α-Methyltestosterone 3-hexyl enol ether | Ether | ||
| Oxymesterone | 4-Hydroxy-17α-methyltestosterone | – | ||
| Penmesterol | 17α-Methyltestosterone 3-cyclopentyl enol ether | Ether | ||
| Tiomesterone | 1α,7α-Diacetylthio-17α-methyltestosterone | – | ||
| Other 17α-substituted testosterone | Danazol | 2,3-Isoxazol-17α-ethynyltestosterone | Ring-fused | |
| Dihydrotestosterone | 1-Testosteronea | 1-Dehydro-4,5α-dihydrotestosterone | – | |
| Androstanolone | 4,5α-Dihydrotestosterone | – | ||
| Bolazine | C3 azine dimer of drostanolone | Dimer | ||
| Drostanolone | 2α-Methyl-4,5α-dihydrotestosterone | – | ||
| Epitiostanol | 2α,3α-Epithio-3-deketo-4,5α-dihydrotestosterone | Ring-fused | ||
| Mepitiostane | 2α,3α-Epithio-3-deketo-4,5α-dihydrotestosterone 17β-(1-methoxycyclopentane) ether | Ring-fused; Ether | ||
| Mesabolonea | 1-Dehydro-4,5α-Dihydrotestosterone 17β-(1-methoxycyclohexane) ether | Ether | ||
| Mesterolone | 1α-Methyl-4,5α-dihydrotestosterone | – | ||
| Metenolone | 1-Dehydro-1-methyl-4,5α-dihydrotestosterone | – | ||
| Prostanozola | 2'H-5α-Androst-2-eno[3,2-c]pyrazol-17β-ol 17β-tetrahydropyran ether | Ether | ||
| Stenbolone | 1-Dehydro-2-methyl-4,5α-dihydrotestosterone | – | ||
| 17α-Alkylated dihydrotestosterone | Androisoxazole | 17α-Methyl-5α-androstano[3,2-c]isoxazol-17β-ol | Ring-fused | |
| Desoxymethyltestosteronea | 2-Dehydro-3-deketo-4,5α-dihydro-17α-methyltestosterone | – | ||
| Furazabol | 17α-Methyl-5α-androstano[2,3-c][1,2,5]oxadiazol-17β-ol | Ring-fused | ||
| Mebolazine | C3 azine dimer of methasterone | Dimer | ||
| Mestanolone | 4,5α-Dihydro-17α-methyltestosterone | – | ||
| Methasteronea | 2α,17α-Dimethyl-4,5α-dihydrotestosterone | – | ||
| Methyl-1-testosteronea | 1-Dehydro-4,5α-dihydro-17α-methyltestosterone | – | ||
| Methyldiazinola | 3-Deketo-3-azi-4,5α-dihydro-17α-methyltestosterone | – | ||
| Methylepitiostanola | 2α,3α-Epithio-3-deketo-4,5α-dihydro-17α-methyltestosterone | – | ||
| Methylstenbolonea | 1-Dehydro-2,17α-dimethyl-4,5α-dihydrotestosterone | – | ||
| Oxandrolone | 2-Oxa-4,5α-dihydro-17α-methyltestosterone | – | ||
| Oxymetholone | 2-Hydroxymethylene-4,5α-dihydro-17α-methyltestosterone | – | ||
| Stanozolol | 17α-Methyl-2'H-5α-androst-2-eno[3,2-c]pyrazol-17β-ol | Ring-fused | ||
| 19-Nortestosterone | 11β-Methyl-19-nortestosteronea | 11β-Methyl-19-nortestosterone | – | |
| 19-Nor-5-androstenediola | 19-Nor-5-androstenediol | Prohormone | ||
| 19-Nordehydroepiandrosteronea | 19-Nor-5-dehydroepiandrosterone | Prohormone | ||
| Bolandiola | 19-Nor-4-androstenediol | Prohormone | ||
| Bolandionea | 19-Nor-4-androstenedione | Prohormone | ||
| Bolmantalatea | 19-Nortestosterone 17β-adamantoate | Ester | ||
| Dienedionea | 9-Dehydro-19-nor-4-androstenedione | Prohormone | ||
| Dienolonea | 9-Dehydro-19-nortestosterone | – | ||
| Dimethandrolonea | 7α,11β-Dimethyl-19-nortestosterone | – | ||
| Methoxydienonea | 2,5(10)-Didehydro-18-methyl-19-norepiandrosterone 3-methyl ether | Prohormone; Ether | ||
| Nandrolone | 19-Nortestosterone | – | ||
| Norclostebol | 4-Chloro-19-nortestosterone | – | ||
| Oxabolone | 4-Hydroxy-19-nortestosterone | – | ||
| Trestolonea | 7α-Methyl-19-nortestosterone | – | ||
| Trenbolone | 9,11-Didehydro-19-nortestosterone | – | ||
| Trendionea | 9,11-Didehydro-19-nor-4-androstenedione | Prohormone | ||
Trestione a
|
7α-Methyl-19-nor-4-androstenedione | Prohormone | ||
| 17α-Alkylated 19-nortestosterone | Dimethyltrienolonea | 7α,17α-Dimethyl-9,11-didehydro-19-nortestosterone | – | |
| Dimethyldienolonea | 7α,17α-Dimethyl-9-dehydro-19-nortestosterone | – | ||
| Ethyldienolonea | 9-Dehydro-17α-ethyl-19-nortestosterone | – | ||
| Ethylestrenol | 17α-Ethyl-3-deketo-19-nortestosterone | – | ||
| Methyldienolonea | 9-Dehydro-17α-methyl-19-nortestosterone | – | ||
| Methylhydroxynandrolonea | 4-Hydroxy-17α-methyl-19-nortestosterone | – | ||
| Metribolonea | 9,11-Didehydro-17α-methyl-19-nortestosterone | – | ||
| Mibolerone | 7α,17α-Dimethyl-19-nortestosterone | – | ||
| Norboletonea | 17α-Ethyl-18-methyl-19-nortestosterone | – | ||
| Norethandrolone | 17α-Ethyl-19-nortestosterone | – | ||
| Normethandrone | 17α-Methyl-19-nortestosterone | – | ||
| Propetandrol | 17α-Ethyl-19-nortestosterone 3-propionate | Ester | ||
| RU-2309a | 9,11-Didehydro-17α,18-dimethyl-19-nortestosterone | – | ||
| Tetrahydrogestrinonea | 9,11-Didehydro-17α-ethyl-18-methyl-19-nortestosterone | – | ||
| Other 17α-substituted 19-nortestosterone | Gestrinone | 9,11-Didehydro-17α-ethynyl-18-methyl-19-nortestosterone | – | |
| Tibolone | 5(10)-Dehydro-7α-methyl-17α-ethynyl-19-nortestosterone | – | ||
| Vinyltestosteronea | 17α-Ethenyltestosterone | – | ||
| Notes: Esters of androgens and anabolic steroids are mostly not included in this table; see here instead. Footnotes: a = Never marketed.
| ||||
| Androgen | Structure | Ester | Relative mol. weight |
Relative T contentb |
logPc | ||||
|---|---|---|---|---|---|---|---|---|---|
| Position(s) | Moiet(ies) | Type | Lengtha | ||||||
| Testosterone | – | – | – | – | 1.00 | 1.00 | 3.0–3.4 | ||
| Testosterone propionate | C17β | Propanoic acid |
Straight-chain fatty acid | 3 | 1.19 | 0.84 | 3.7–4.9 | ||
| Testosterone isobutyrate | C17β | Isobutyric acid | Branched-chain fatty acid | – (~3) | 1.24 | 0.80 | 4.9–5.3 | ||
| Testosterone isocaproate | C17β | Isohexanoic acid |
Branched-chain fatty acid | – (~5) | 1.34 | 0.75 | 4.4–6.3 | ||
| Testosterone caproate | C17β | Hexanoic acid |
Straight-chain fatty acid | 6 | 1.35 | 0.75 | 5.8–6.5 | ||
| Testosterone phenylpropionate | C17β | Phenylpropanoic acid | Aromatic fatty acid | – (~6) | 1.46 | 0.69 | 5.8–6.5 | ||
| Testosterone cypionate | C17β | Cyclopentylpropanoic acid |
Cyclic carboxylic acid | – (~6) | 1.43 | 0.70 | 5.1–7.0 | ||
| Testosterone enanthate | C17β | Heptanoic acid |
Straight-chain fatty acid | 7 | 1.39 | 0.72 | 3.6–7.0 | ||
| Testosterone decanoate | C17β | Decanoic acid |
Straight-chain fatty acid | 10 | 1.53 | 0.65 | 6.3–8.6 | ||
| Testosterone undecanoate | C17β | Undecanoic acid |
Straight-chain fatty acid | 11 | 1.58 | 0.63 | 6.7–9.2 | ||
| Testosterone buciclated | C17β | Bucyclic acid e |
Cyclic carboxylic acid | – (~9) | 1.58 | 0.63 | 7.9–8.5 | ||
| Footnotes: a = Length of Bucyclic acid = trans-4-Butylcyclohexane-1-carboxylic acid. Sources: See individual articles.
| |||||||||
| Anabolic steroid | Structure | Ester | Relative mol. weight |
Relative AAS contentb |
Durationc | ||||
|---|---|---|---|---|---|---|---|---|---|
| Position | Moiety | Type | Lengtha | ||||||
| Boldenone undecylenate | C17β | Undecylenic acid | Straight-chain fatty acid | 11 | 1.58 | 0.63 | Long | ||
| Drostanolone propionate | C17β | Propanoic acid |
Straight-chain fatty acid | 3 | 1.18 | 0.84 | Short | ||
| Metenolone acetate | C17β | Ethanoic acid |
Straight-chain fatty acid | 2 | 1.14 | 0.88 | Short | ||
| Metenolone enanthate | C17β | Heptanoic acid |
Straight-chain fatty acid | 7 | 1.37 | 0.73 | Long | ||
| Nandrolone decanoate | C17β | Decanoic acid |
Straight-chain fatty acid | 10 | 1.56 | 0.64 | Long | ||
| Nandrolone phenylpropionate | C17β | Phenylpropanoic acid | Aromatic fatty acid | – (~6–7) | 1.48 | 0.67 | Long | ||
| Trenbolone acetate | C17β | Ethanoic acid |
Straight-chain fatty acid | 2 | 1.16 | 0.87 | Short | ||
| Trenbolone enanthated | C17β | Heptanoic acid |
Straight-chain fatty acid | 7 | 1.41 | 0.71 | Long | ||
| Footnotes: a = Length of oil solution . d = Never marketed. Sources: See individual articles.
| |||||||||
Structural conversions of anabolic steroids
Testosterone to derivatives
Conversion to DHT,[187] nandrolone,[73] metandienone (Dianabol),[188] chlorodehydromethyltestosterone (Turinabol),[189] fluoxymesterone (Halotestin),[190] and boldenone (Equipoise):[191]

DHT to derivatives
DHT to stanozolol (Winstrol),[192] metenolone acetate (Primobolan),[193] oxymetholone (Anadrol),[194] and methasterone (Superdrol):[195]

Nandrolone to derivatives
Nandrolone to trestolone,[196] trenbolone,[197] norboletone,[198] and ethylestrenol:[199]

Detection in body fluids
The most commonly employed human physiological specimen for detecting AAS usage is urine, although both blood and hair have been investigated for this purpose. The AAS, whether of endogenous or exogenous origin, are subject to extensive hepatic biotransformation by a variety of enzymatic pathways. The primary urinary metabolites may be detectable for up to 30 days after the last use, depending on the specific agent, dose and route of administration. A number of the drugs have common metabolic pathways, and their excretion profiles may overlap those of the endogenous steroids, making interpretation of testing results a very significant challenge to the analytical chemist. Methods for detection of the substances or their excretion products in urine specimens usually involve gas chromatography–mass spectrometry or liquid chromatography-mass spectrometry.[200][201][202][203]
History
| Generic name | Class[a] | Brand name | Route[b] | Intr. | ||
|---|---|---|---|---|---|---|
| Androstanolone[c][d] | DHT | Andractim | PO,[e] IM, TD | 1953 | ||
| Boldenone undecylenate[f] | Ester | Equipoise[g] | IM | 1960s | ||
| Danazol | Alkyl | Danocrine | PO | 1971 | ||
| Drostanolone propionate[e] | DHT Ester | Masteron | IM | 1961 | ||
| Ethylestrenol[d] | 19-NT Alkyl | Maxibolin[g] | PO | 1961 | ||
| Fluoxymesterone[d] | Alkyl | Halotestin[g] | PO | 1957 | ||
| Mestanolone[e] | DHT Alkyl | Androstalone[g] | PO | 1950s | ||
| Mesterolone | DHT | Proviron | PO | 1967 | ||
| Metandienone[d] | Alkyl | Dianabol | PO, IM | 1958 | ||
| Metenolone acetate[d] | DHT Ester | Primobolan | PO | 1961 | ||
| Metenolone enanthate[d] | DHT Ester | Primobolan Depot | IM | 1962 | ||
| Methyltestosterone[d] | Alkyl | Metandren | PO | 1936 | ||
| Nandrolone decanoate | 19-NT Ester | Deca-Durabolin | IM | 1962 | ||
| Nandrolone phenylpropionate[d] | 19-NT Ester | Durabolin | IM | 1959 | ||
| Norethandrolone[d] | 19-NT Alkyl | Nilevar[g] | PO | 1956 | ||
| Oxandrolone[d] | DHT Alkyl | Oxandrin[g] | PO | 1964 | ||
| Oxymetholone[d] | DHT Alkyl | Anadrol[g] | PO | 1961 | ||
| Prasterone[h] | Prohormone | Intrarosa[g] | PO, IM, vaginal | 1970s | ||
| Stanozolol[e] | DHT Alkyl | Winstrol[g] | PO, IM | 1962 | ||
| Testosterone cypionate | Ester | Depo-Testosterone | IM | 1951 | ||
| Testosterone enanthate | Ester | Delatestryl | IM | 1954 | ||
| Testosterone propionate | Ester | Testoviron | IM | 1937 | ||
| Testosterone undecanoate | Ester | Andriol[g] | PO, IM | 1970s | ||
| Trenbolone acetate[f] | 19-NT Ester | Finajet[g] | IM | 1970s | ||
| ||||||
Discovery of androgens
The use of
In the 1930s, it was already known that the
Clinical trials on humans, involving either PO doses of methyltestosterone or injections of testosterone propionate, began as early as 1937.[205] There are often reported rumors that German soldiers were administered AAS during the Second World War, the aim being to increase their aggression and stamina, but these are, as yet, unproven.[119]: 6 Adolf Hitler himself, according to his physician, was injected with testosterone derivatives to treat various ailments.[210] AAS were used in experiments conducted by the Nazis on concentration camp inmates,[210] and later by the allies attempting to treat the malnourished victims that survived Nazi camps.[119]: 6 President John F. Kennedy was administered steroids both before and during his presidency.[211]
Development of synthetic AAS
The development of muscle-building properties of testosterone was pursued in the 1940s, in the Soviet Union and in
Three major ideas governed modifications of testosterone into a multitude of AAS:
Society and culture
Etymology
Androgens were discovered in the 1930s and were characterized as having effects described as androgenic (i.e., virilizing) and anabolic (e.g., myotrophic, renotrophic).[68][73] The term anabolic steroid can be dated as far back as at least the mid-1940s, when it was used to describe the at-the-time hypothetical concept of a testosterone-derived steroid with anabolic effects but with minimal or no androgenic effects.[214] This concept was formulated based on the observation that steroids had ratios of renotrophic to androgenic potency that differed significantly, which suggested that anabolic and androgenic effects might be dissociable.[214]
In 1953, a testosterone-derived steroid known as
Although anabolic steroid was originally intended to specifically describe testosterone-derived steroids with a marked dissociation of anabolic and androgenic effect, it is applied today indiscriminately to all steroids with AR agonism-based anabolic effects regardless of their androgenic potency, including even non-synthetic and non-preferentially-anabolic steroids like testosterone.[68][73][215] While many anabolic steroids have diminished androgenic potency in comparison to anabolic potency, there is no anabolic steroid that is exclusively anabolic, and hence all anabolic steroids retain at least some degree of androgenicity.[68][73][215] (Likewise, all "androgens" are inherently anabolic.)[68][73][215] Indeed, it is probably not possible to fully dissociate anabolic effects from androgenic effects, as both types of effects are mediated by the same signaling receptor, the AR.[73] As such, the distinction between the terms anabolic steroid and androgen is questionable, and this is the basis for the revised and more recent term anabolic–androgenic steroid (AAS).[68][73][215]
David Handelsman has criticized terminology and understanding surrounding AAS in many publications.[221][222][223][224][225][226][227][228] According to Handelsman, the pharmaceutical industry attempted to dissociate the so-called "androgenic" and "anabolic" effects of AAS in the mid-20th-century in order to create non-masculinizing anabolic agents that would be more suitable for use in women and children.[221] However, this effort failed comprehensively and was abandoned by the 1970s.[221][222] This failure was due to the subsequent discovery of a singular androgen receptor (AR) mediating the effects of AAS in both muscle and reproductive tissue, along with misinterpretation of flawed animal androgen bioassays employed to distinguish between androgenic or virilizing effects and anabolic or myotrophic effects (i.e., the Hershberger assay involving the unrepresentative levator ani muscle).[221][222] In reality, all AAS have essentially similar AR-mediated effects,[228] even if some may differ in potency to a degree in certain tissues (e.g., skin, hair follicles, prostate gland) based on susceptibility to 5α-reduction and associated metabolic amplification or inactivation or lack thereof.[228][8] Per Handelsman, the terms "anabolic steroid" and "anabolic–androgenic steroid" are obsolete, meaningless, and falsely distinguish these agents from androgens when there is no physiological basis for such distinction.[221][222] In fact, it has been noted that the use and distinction of the concepts "anabolic" and "androgenic" as well as the term "anabolic–androgenic steroid" are oxymoronic, as anabolic refers to muscle-building while androgenic refers to induction and maintenance of male secondary sexual characteristics (which in principle would include anabolic or muscle-building effects).[221][222][229] Handelsman has argued that these terms should be discarded and instead, AAS should all simply be referred to as "androgens", with him using this term exclusively to refer to these agents in his publications.[221][222] Although the term "anabolic–androgenic steroid" is technically valid in describing two types of actions of these agents, Handelsman considers the term unnecessary and redundant and likens it to hypothetical never-used terms like "luteal–gestational progestins" or "mammary–uterine estrogens".[221] Handelsman also notes that "anabolic steroid" is easily and unnecessarily confusable with corticosteroids.[221] Aside from AAS, Handelsman has criticized the term "selective androgen receptor modulator (SARM)" and claims about these agents as well.[223][221][222][227]
Legal status
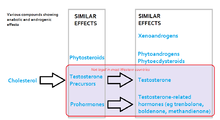
The legal status of AAS varies from country to country: some have stricter controls on their use or prescription than others though in many countries they are not illegal. In the U.S., AAS are currently listed as Schedule III
In Canada, researchers have concluded that steroid use among student athletes is extremely widespread. A study conducted in 1993 by the Canadian Centre for Drug-Free Sport found that nearly 83,000 Canadians between the ages of 11 and 18 use steroids.
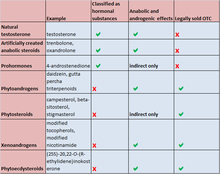
| Substance | Example | Classified as hormonal substances | Anabolic and androgenic effects | Legally sold OTC |
|---|---|---|---|---|
| Natural testosterone | testosterone | hormonal | yes | not legal |
| Artificially created anabolic steroids | trenbolone, oxandrolone | hormonal | yes | not legal |
| Prohormones | 4-androstenedione | hormonal | indirect only | not legal |
| Phytoandrogens | daidzein, gutta-percha triterpenoids | no | yes | legal |
| Phytosteroids | campesterol, beta-sitosterole, stigmasterol | no | indirect only | legal |
| Xenoandrogens | modified tocopherols, modified nicotinamide | no | yes | legal |
| Phytoecdysteroids | (25S)-20, 22-O-(R-ethylidene)inokosterone | no | yes | legal |
Selective androgen receptor modulators
|
ostarine |
anabolic[234] | not for human consumption[235][236] |
United States

The history of the U.S. legislation on AAS goes back to the late 1980s, when the
The same act also introduced more stringent controls with higher criminal penalties for offenses involving the illegal distribution of AAS and human growth hormone. By the early 1990s, after AAS were scheduled in the U.S., several pharmaceutical companies stopped manufacturing or marketing the products in the U.S., including
Even though they can still be prescribed by a medical doctor in the U.S., the use of anabolic steroids for injury recovery purposes has been a taboo subject, even amongst the majority of sports medicine doctors and endocrinologists.
United Kingdom
In the United Kingdom, AAS are classified as class C drugs, which puts them in the same class as
Part 1 drugs are subject to full import and export controls with possession being an offence without an appropriate prescription. There is no restriction on the possession when it is part of a medicinal product. Part 2 drugs require a Home Office licence for importation and export unless the substance is in the form of a medicinal product and is for self-administration by a person.[239]
Status in sports
AAS are banned by all major sports bodies including
Usage
Law enforcement
United States federal law enforcement officials have expressed concern about AAS use by police officers. "It's a big problem, and from the number of cases, it's something we shouldn't ignore. It's not that we set out to target cops, but when we're in the middle of an active investigation into steroids, there have been quite a few cases that have led back to police officers," says Lawrence Payne, a spokesman for the United States Drug Enforcement Administration.[247] The FBI Law Enforcement Bulletin stated that "Anabolic steroid abuse by police officers is a serious problem that merits greater awareness by departments across the country".[248] It is also believed that police officers across the United Kingdom "are using criminals to buy steroids" which he claims to be a top risk factor for police corruption.
Professional wrestling
Following the
Economics

AAS are frequently produced in pharmaceutical laboratories, but, in nations where stricter laws are present, they are also produced in small home-made underground laboratories, usually from raw substances imported from abroad.[252] In these countries, the majority of steroids are obtained illegally through black market trade.[253][254] These steroids are usually manufactured in other countries, and therefore must be smuggled across international borders. As with most significant smuggling operations, organized crime is involved.[255]
In the late 2000s, the worldwide trade in illicit AAS increased significantly, and authorities announced record captures on three continents. In 2006, Finnish authorities announced a record seizure of 11.8 million AAS tablets. A year later, the DEA seized 11.4 million units of AAS in the largest U.S. seizure ever. In the first three months of 2008, Australian customs reported a record 300 seizures of AAS shipments.[111]
In the U.S., Canada, and Europe, illegal steroids are sometimes purchased just as any other illegal drug, through dealers who are able to obtain the drugs from a number of sources. Illegal AAS are sometimes sold at gyms and competitions, and through the mail, but may also be obtained through pharmacists, veterinarians, and physicians.[256] In addition, a significant number of counterfeit products are sold as AAS, in particular via mail order from websites posing as overseas pharmacies. In the U.S., black-market importation continues from Mexico, Thailand, and other countries where steroids are more easily available, as they are legal.[257]
Research
AAS, alone and in combination with
Topical androgens have been used and studied in the treatment of
See also
- Selective androgen receptor modulator
- Antiandrogen
- Mossman-Pacey paradox
- Steroid rosacea
- Steroid use in Bollywood
- Bigger, Stronger, Faster
- Juiced: Wild Times, Rampant 'Roids, Smash Hits & How Baseball Got Big
- List of androgens/anabolic steroids available in the United States
References
- PMID 29494025.
Endogenous anabolic steroids such as testosterone and dihydrotestosterone and synthetic anabolic steroids mediate their effects by binding to and activating androgen receptors.
- ^ PMID 7556805.
- ^ S2CID 83319949.
- ^ S2CID 19425569.
- ^ PMID 19751025.
- ^ PMID 21443513.
- ^ S2CID 15234016.
- ^ S2CID 24339701.

- ISBN 978-1-55642-901-9. Archivedfrom the original on 22 December 2016. Retrieved 17 October 2016.
- ^ PMID 11701661.
[...] in a recent animal study, Hsiao et al. (10) found two different kinds of androgen response elements that could respond differentially to T and DHT. Therefore, it is possible that a selective androgen response element sequence may play a role in differential T vs. DHT AR trans-activation.
- PMID 2429792.
- ^ S2CID 25911263.
- S2CID 9832782.
- PMID 11320105.
- PMID 17824721.
- S2CID 24170276.
- S2CID 42844580.
- ^ ISBN 978-0-8036-2027-8. Archivedfrom the original on 14 April 2021. Retrieved 25 June 2017.
- ISBN 978-1-86016-079-0. Archivedfrom the original on 14 April 2021. Retrieved 25 June 2017.
- S2CID 23520067.
- ISBN 978-0-7864-1128-3. Archivedfrom the original on 14 April 2021. Retrieved 25 June 2017.
- ^ a b "Oxandrolone Tablets, USP – Rx only" (PDF). Drugs@FDA. U.S. Food and Drug Administration. 1 December 2006. Archived (PDF) from the original on 26 August 2016. Retrieved 21 June 2016.
- ^ a b "Oxandrin (oxandrolone tablets, USP)" (PDF). Drugs@FDA. BTG Pharmaceuticals, U.S. Food and Drug Administration. 21 April 2003. Archived (PDF) from the original on 1 March 2017. Retrieved 21 June 2016.
- S2CID 24139354.
- PMID 23121414.
- S2CID 207323793.
- PMID 22541704.
- ISBN 978-1-4200-4744-8. Archivedfrom the original on 14 April 2021. Retrieved 27 July 2018.
- ISBN 978-0-323-07824-5. Archivedfrom the original on 14 April 2021. Retrieved 27 July 2018.
- ^ "Android® C-III Label" (PDF). Archived (PDF) from the original on 10 February 2017. Retrieved 27 July 2018.
- PMID 35266057.
- PMID 17403329.
- PMID 17367445.
- S2CID 31976796.
- S2CID 19174381.
- S2CID 5031396.
- PMID 12915619.
- ^ Notaro K (24 October 2013). "Genderqueer, Pansexual, LGBTQ: Will Gender Exist 100 Years From Now? – Rebooted". Institute for Ethics and Emerging Technologies (IEET). Archived from the original on 9 December 2014. Retrieved 17 November 2014.
- ^ Leigh S (12 February 2014). "Young people exploring nonbinary gender roles". SF Gate. Archived from the original on 8 December 2014. Retrieved 17 November 2014.
- S2CID 71737774.
- ^ "What is Intersex – An Intersex FAQ by Inter/Act". Inter/Act Youth. 12 September 2014. Archived from the original on 7 December 2014. Retrieved 5 December 2014.
- ISBN 978-1-4698-0343-2. Archivedfrom the original on 14 April 2021. Retrieved 25 June 2017.
- PMID 3909420.
- PMID 10076169.
- PMID 17087613.
- PMID 18488873.
- ^ S2CID 8030248.
- PMID 11304877.
- ^ (PDF) from the original on 5 December 2020. Retrieved 18 August 2019.
- ^ Baalmann J (19 June 2017). "Testosterone can be part of your treatment plan for Raynaud's Disease". BioBalance Health. Retrieved 9 June 2023.
- ^ "Most steroid users are not athletes: study". Reuters. Reuters. 21 November 2007. Archived from the original on 25 January 2016. Retrieved 3 January 2014.
- S2CID 10762429.
- PMID 8355384.
- PMID 17512138.
- PMID 16679978.
- ^ PMID 17931410.
- S2CID 8699231.
- ^ Eastley T (18 January 2006). "Steroid study debunks user stereotypes". ABC. Archived from the original on 16 July 2014. Retrieved 3 January 2014.
- PMID 15317640.
- S2CID 38738640.
- S2CID 5794238.
- ^ PMID 2199753.
- ^ "Drugs@FDA: FDA Approved Drug Products". United States Food and Drug Administration. Archived from the original on 16 November 2016. Retrieved 1 December 2019.
- ISBN 978-1-284-05671-6. Archivedfrom the original on 13 February 2021. Retrieved 13 September 2020.
- ISBN 978-1-60547-633-9. Archivedfrom the original on 14 April 2021. Retrieved 13 September 2020.
- ISBN 978-1-60913-345-0. Archivedfrom the original on 14 April 2021. Retrieved 13 September 2020.
- ISBN 978-1-4684-5499-4. Archivedfrom the original on 14 April 2021. Retrieved 13 September 2020.
- ^ ISBN 978-0-9828280-1-4. Archivedfrom the original on 14 April 2021. Retrieved 2 December 2016.
- ISSN 0091-3847.
- ISBN 978-3-642-68223-0. Archivedfrom the original on 13 February 2021. Retrieved 13 September 2020.
- PMID 6372655.
- ^ ISBN 978-0-7817-1750-2. Archivedfrom the original on 17 May 2020. Retrieved 17 October 2016.
- ^ PMID 18500378.
- ^ S2CID 6999218.
- ^ ISBN 978-0-07-176401-8.
- PMID 11366379.
- PMID 17723870.
- PMID 24423981.
- ^ S2CID 41356223.
- PMID 25805894.
- PMID 16000671.
- ^ S2CID 32243658.
- PMID 20020375.
- PMID 20816133.
- PMID 28379599.
- PMID 21443507.
- PMID 14609147.
- ^ "DailyMed: About DailyMed". Dailymed.nlm.nih.gov. Archived from the original on 12 May 2009. Retrieved 3 November 2008.
- PMID 1586105.
- S2CID 37024092.
- S2CID 13382470.
- PMID 12870172.
- PMID 11927236.
- ^ "Known and Probable Human Carcinogens". American Cancer Society. 29 June 2011. Archived from the original on 17 November 2014. Retrieved 17 November 2014.
- PMID 10499702.
- S2CID 22123696.
- S2CID 45280834.
- S2CID 42891343.
- PMID 4079743.
- ^ Tokar S (February 2006). "Liver Damage And Increased Heart Attack Risk Caused By Anabolic Steroid Use". University of California – San Francisco. Archived from the original on 14 June 2011. Retrieved 24 April 2007.
- PMID 26051296.
- PMID 779604.
- PMID 2104626.
- ^ Hoffman JR, Ratamess NA (1 June 2006). "Medical Issues Associated with Anabolic Steroid Use: Are they Exaggerated?" (PDF). Journal of Sports Science and Medicine. Archived (PDF) from the original on 20 June 2007. Retrieved 8 May 2007.
- S2CID 2400041.
- PMID 2991700.
- PMID 9216474.
- PMID 14576190.
- ^ Herlitz LC, Markowitz GS, Farris AB, Schwimmer JA, Stokes MB, Kunis C, et al. (29 October 2009). Development of FSGS Following Anabolic Steroid Use in Bodybuilders (PDF). 42nd Annual Meeting and Scientific Exposition of the American Society of Nephrology. Archived (PDF) from the original on 7 October 2018. Retrieved 17 November 2014.
- S2CID 5903121.
- ^ PMID 18599224.
- S2CID 25684227.
- ^ .
- PMID 8889121.
- ^ "Dr. Ritchi Morris". Vitalquests.org. Archived from the original on 3 December 2013. Retrieved 1 December 2013.
- PMID 19922565.
- ISBN 978-0-683-05211-4.
- S2CID 22121959.
- ^ ISBN 0-415-28030-3.
- S2CID 2009549.
- PMID 18923108.
- S2CID 23846419.
- S2CID 22571743.
- ^ PMID 26855782.
- PMID 4437350.
- S2CID 42489596.
- S2CID 42489596.
- PMID 16641486.
- PMID 16238547.
- S2CID 43430651.
- PMID 8855787.
- PMID 12960001.
- ^ S2CID 24665772.
- S2CID 28160750.
- PMID 18950759.
- PMID 16424293.
- PMID 10710012.
- ^ PMID 12017555.
- S2CID 29441013.
- S2CID 2628925.
- S2CID 1887613.
- S2CID 229152974.
- S2CID 30590287.
- ^ S2CID 73721690.
- S2CID 2344757.
- ^ PMID 431680.
- ^ PMID 16985920.
- ISBN 0-7668-1142-5. Archivedfrom the original on 19 August 2020. Retrieved 17 October 2016.
- ^ ISBN 978-0-323-24466-4. Archivedfrom the original on 19 August 2020. Retrieved 17 October 2016.
- ISBN 978-1-84593-113-1. Archivedfrom the original on 22 December 2016. Retrieved 17 October 2016.
- ISBN 978-1-84788-339-1. Archivedfrom the original on 19 August 2020. Retrieved 17 October 2016.
- ^ PMID 11343670.
Although both testosterone and dihydrotestosterone activate the same androgen receptor, differences in the sequence of androgen response elements are responsible for differential regulation of these hormones (21).
- PMID 25257522.
- ^ PMID 28479083.
- ^ PMID 26303084.[permanent dead link]
- ^ ISBN 978-0-08-092346-8. Archivedfrom the original on 14 April 2021. Retrieved 21 December 2017.
- .
- PMID 21681779.
- PMID 9920070.
- ^ S2CID 29134676.
- ^ S2CID 54394931.
- ^ PMID 8858992.
- ^ S2CID 31725431.
- ^ PMID 17433821.
- ^ S2CID 26938839.
- (PDF) from the original on 23 August 2017. Retrieved 1 April 2019.
- PMID 20599615.
- ISBN 978-0-08-092346-8.
- ISBN 978-1-4377-2075-4. Archivedfrom the original on 9 January 2020. Retrieved 2 December 2016.
- ^ ISBN 978-3-540-79088-4. Archivedfrom the original on 9 January 2020. Retrieved 2 December 2016.
- ^ PMID 18555683.
- ISBN 978-0-9828280-1-4. Archivedfrom the original on 17 May 2020. Retrieved 2 December 2016.
- ISBN 952-91-2950-5. Archived from the original(PDF) on 11 August 2017. Retrieved 2 December 2016.
Androgens, estrogens and progestins exert a negative feedback effect on the secretion of GnRH and LH by their actions on the pituitary and the hypothalamus. Most of the negative feedback effect of androgens is caused by their estrogenic metabolites produced by aromatization. 5α-Reduction does not seem to be necessary for the negative feedback effect of testosterone. (Rittmaster et al, 1992; Kumar et al, 1995a; Hayes et al, 2000).
- ^ PMID 16497801.
- ISBN 978-1-59259-016-2. Archivedfrom the original on 28 September 2017. Retrieved 2 December 2016.
- ISBN 978-1-4925-0162-6. Archivedfrom the original on 17 February 2018. Retrieved 2 December 2016.
- ISBN 978-1-60913-345-0.
- ^ ISBN 978-1-4200-4211-5. Archivedfrom the original on 9 January 2020. Retrieved 2 December 2016.
- ^ PMID 20153798.
- PMID 3042375.
- ISBN 978-0-323-32195-2. Archivedfrom the original on 9 January 2020. Retrieved 2 December 2016.
- ISBN 978-1-107-01290-5. Archivedfrom the original on 16 May 2020. Retrieved 2 December 2016.
- ISBN 978-3-642-61013-4. Archivedfrom the original on 9 January 2020. Retrieved 2 December 2016.
- PMID 11589254.
- ^ S2CID 30314430.
- PMID 18230794.
- ISSN 0018-019X.
- PMID 3773526.
- PMID 9518142.
- S2CID 206592924.
- ISSN 0002-7863.
- PMID 33948794.
- ISSN 0002-7863.
- PMID 22362568.
- ISSN 0022-3263.
- ISBN 978-3-13-536101-7.
- PMID 12112254.
- PMID 817322.
- PMID 18570179.
- S2CID 10051396.
- PMID 19465014.
- ISBN 978-0-9626523-7-0.
- ^ Randhawa, G. K., & Sharma, R. (2015). Chemotherapeutic potential of cow urine: A review. Journal of intercultural ethnopharmacology, 4(2), 180.
- ^ PMID 7817189.
- ^ PMID 11176375.
- .
- .
- .
- ^ ISBN 978-0-7864-1128-3.
- ^ Suarez R, Senior Correspondent, Kelman J, physician (18 November 2002). "President Kennedy's Health Secrets". PBS NewsHour. Public Broadcasting System. Archived from the original on 22 January 2014. Retrieved 24 August 2017.
- S2CID 6559714.
- ^ Peters J (18 February 2005). "The Man Behind the Juice". Slate. Archived from the original on 7 September 2011. Retrieved 29 April 2008.
- ^ ISSN 0083-6729.
In recent years several laboratories (Kochakian, Albright, Wilkins) have entertained the hope of finding a protein anabolic steroid without any, or with only minor, sexual effects. These studies have received special impetus and encouragement from the observation of Kochakian that certain steroids have greater renotrophic (anabolic?) than androgenic effects.
- ^ ISBN 978-3-642-66353-6. Archivedfrom the original on 14 April 2021. Retrieved 1 April 2019.
- ISBN 978-0-471-89979-2.
- ^ ISBN 978-0-08-056877-5. Archivedfrom the original on 14 April 2021. Retrieved 28 June 2018.
- ^ ISBN 978-1-57028-013-9. Archivedfrom the original on 14 April 2021. Retrieved 28 June 2018.
- ^ United States. Patent Office (1957). Official Gazette of the United States Patent Office. U.S. Patent Office.
- ISBN 978-1-61779-222-9. Archivedfrom the original on 14 April 2021. Retrieved 28 June 2018.
- ^ PMID 21511988.
- ^ PMID 33484556.
However, a third major quest, for the development of a nonvirilizing androgen ("anabolic steroid") suitable for use in women and children, based on dissociating the virilizing from the anabolic effects of androgens failed comprehensively (36). This failure is now understood as being due to the discovery of a singular androgen receptor (AR) together with the misinterpretation of nonspecific whole animal androgen bioassays employed to distinguish between anabolic and virilizing effects (37). The term "androgen" is used herein for both endogenous and synthetic androgens including references to chemicals named elsewhere as "anabolic steroids," "anabolic-androgenic steroids," or "specific AR modulators" (SARM), which continue to make an obsolete and oxymoronic distinction between virilizing and anabolic effects of androgens where there is no difference (36).
- ^ PMID 35277356.
- S2CID 44485330.
These findings also highlight how obsolete is the term "anabolic steroid," when falsely distinguishing from "androgen," a dichotomy devoid of physiological meaning and lingering mainly as a media piñata.10
- ISBN 978-3-319-46084-0.
By definition, all androgens combine intrinsic anabolic and androgenic properties, which have never been meaningfully separated [67], manifest via the androgen receptor, a protein encoded by a single copy gene. Hence the singularity of androgen action means that the terms "anabolic steroid" or "androgenic-anabolic steroids" remain an obsolete terminology making a distinction between androgenic and anabolic effects where there is no real difference [67]. This obsolete yet widely used terminology represent a vestige of the unsuccessful quest by the pharmaceutical industry to dissociate the virilizing from anabolic properties and remains in the public mind mainly as a media piñata. Androgen abuse, a more appropriate term which encompasses illicit use of all available androgens, will be used in this chapter.
- PMID 27347677.
Following the hiatus of World War II, the pharmaceutical industry development of synthetic steroids included pursuing the goal of a nonvirilizing androgen ('anabolic steroid') potentially suitable for use to obtain pharmacological androgen effects in women and children. [...] the industrial quest for an 'anabolic steroid' based on dissociating the virilizing from the anabolic effects of androgens failed. This is now understood in the light of the later discovery of the singular AR together with the flawed interpretations of relatively nonspecific whole animal bioassays then used to screen synthetic steroids for supposedly distinct anabolic and virilizing effects. Yet, despite the industry's abandonment of this fruitless endeavor by 1980, and its recent reincarnation under the guise of developing a 'selective AR modulator' (SARM) [6] , the empty concept of an 'anabolic steroid' persists as an ill-defined and misleading scientific terminology [7, 8] . In this paper, the more accurate and clearer term 'androgen' is used exclusively for both endogenous and synthetic androgens, but includes references to chemicals loosely defined elsewhere as 'anabolic steroids' or 'anabolicandrogenic steroids', which confuse by making an obsolete distinction where there is no difference.
- ^ ISBN 978-1-139-00335-3.
The development of nonsteroidal androgens, marketed as "selective androgen receptor modulators" (SARMs), offers new possibilities for adjuvant pharmacological androgen therapy. In contrast to the full spectrum of androgen effects of testosterone, such SARMs would be pure androgens not subject to tissue-specific activation by aromatization to a corresponding estrogen or to amplification of androgenic potency by 5a-reduction. In this context the endogenous pure androgens nandrolone and DHT can be considered prototype SARMs. SARMs are not the modern embodiment of so-called "anabolic steroids," an outdated term referring to hypothetical but nonexistent non-virilizing androgens targeted exclusively to muscle, a failed concept lacking biological proof of principle (Handelsman 2011).
- ^ PMID 25905231.
The identification of a single gene and protein for the androgen receptor in 1988 (584-586) explains the physiologic observation that, at equivalent doses, all androgens have essentially similar effects (587). The term "anabolic steroid" was invented during the post-WWII golden age of steroid pharmacology to define an idealized androgen lacking virilizing features but maintaining myotrophic properties so that it could be used safely in chidren and women. Although this quest proved illusory and was abandoned after all industry efforts failed to identify such a hypothetical synthetic androgen, the obsolete term "anabolic steroid" persists mainly as a lurid descriptor in popular media despite continuing to make a false distinction where there is no difference. Better understanding of the metabolic activation of androgens via 5α-reduction and aromatization in target tissues and the tissue-specific partial agonist/antagonist properties of some synthetic androgens may lead to more physiological concepts of tissue-specific androgen action ("specific androgen receptor modulator") governed by the physiological processes of pre-receptor androgen activation as well as post-receptor interaction with co-regulator proteins analogous to the development of synthetic estrogen partial agonists with tissue specificity ("specific estrogen receptor modulator") (588). The potential for new clinical therapeutic indications of novel tissue-selective androgens in clinical development remain to be fully evaluated (589).
- PMID 36644692.
Anabolic–androgenic steroids (AAS) are a class of natural and synthetic hormones that owe their name to their chemical structure (the steroid nucleus, see Figure 1) and the biological effects (anabolic and androgenic) they induce. Anabolic refers to the skeletal muscle-building properties of AAS, whereas androgenic refers to the induction and maintenance of male secondary sexual characteristics (which in principle includes the anabolic action, thereby rendering the term an oxymoron (1)).
- ^ "Title 21 United States Code (USC) Controlled Substances Act". US Department of Justice. Archived from the original on 24 July 2009. Retrieved 7 September 2009.
- ^ Controlled Drugs and Substances Act, S.C. 1996, c. 19, s. 4(7) (Controlled Drugs and Substances Act at Department of Justice)
- ^ Deacon J (2 May 1994). "Biceps in a bottle". Maclean's. 107 (18): 52–53.
- ^ "Steroids". Australian Institute of Criminology. 2006. Archived from the original on 5 April 2007. Retrieved 6 May 2007.
- PMID 30503797.
- S2CID 221347916.
- ISSN 2211-2669.
- ^ H.R. 4658
- ^ "News from DEA, Congressional Testimony, 03/16/04". Archived from the original on 6 February 2007. Retrieved 24 April 2007.
- ^ "Patient.info Controlled Drugs". Egton Medical Information Systems Limited. Archived from the original on 13 June 2015. Retrieved 8 August 2013.
- ^ "FIFA Anit-Doping Regulations" (PDF). FIFA. Archived from the original (PDF) on 21 January 2012. Retrieved 1 December 2013.
- ^ "Olympic movement anti-doping code" (PDF). International Olympic Committee. 1999. Archived (PDF) from the original on 3 October 2018. Retrieved 6 May 2007.
- ^ "The NVA and NBPA anti-drug program". NBA Policy. findlaw.com. 1999. Archived from the original on 22 November 2007. Retrieved 6 May 2007.
- ^ "NHL/NHLPA performance-enhancing substances program summary". nhlpa.com. Archived from the original on 2 June 2007. Retrieved 6 May 2007.
- ^ "List of Prohibited Substances" (PDF). nflpa.com. 2006. Archived from the original (PDF) on 20 June 2007. Retrieved 6 May 2007.
- ^ "World anti-doping code" (PDF). WADA. 2003. Archived from the original (PDF) on 7 August 2007. Retrieved 10 July 2007.
- ^ "Prohibited list of 2005" (PDF). WADA. 2005. Archived from the original (PDF) on 20 June 2007. Retrieved 6 May 2007.
- ^ Keeping J (27 December 2010). "Steroid abuse among law enforcement a problem nationwide". The Ann Arbor News. Archived from the original on 4 March 2016. Retrieved 1 December 2013.
- ^ "Anabolic Steroid Use and Abuse by Police Officers: Policy & Prevention". The Police Chief. June 2008. Archived from the original on 3 December 2013. Retrieved 1 December 2013.
- ^ Brian L (1 March 2010). "WWE steroid investigation: A controversy McMahon 'doesn't need'". Greenwich Time. Archived from the original on 31 January 2021. Retrieved 1 March 2010.
- ^ "Talent Wellness Program Program Summary Report 1 July 2007 31 March 2008" (PDF). World Wrestling Entertainment, Inc. Archived from the original (PDF) on 24 December 2010.
- ^ "Deposition details McMahon steroid testimony". News from southeastern Connecticut. The Day. 13 December 2007. Archived from the original on 12 June 2010. Retrieved 14 August 2010.
- ^ Assael S (24 September 2007). "'Raw Deal' busts labs across U.S., many supplied by China". ESPN The Magazine. Archived from the original on 14 October 2007. Retrieved 24 September 2007.
- ISBN 978-0-88011-786-9. Archivedfrom the original on 14 April 2021. Retrieved 3 June 2020.
- S2CID 143442371.
- ISBN 978-0-470-83733-7.
- ^ "Steroids". National Institute on Drug Abuse. GDCADA. Archived from the original on 11 September 2007. Retrieved 13 September 2007.
- ^ "The Drug Enforcement Administration's International Operations (Redacted)". Office of the Inspector General. USDOJ. February 2007. Archived from the original on 30 October 2013. Retrieved 2 January 2014.
- PMID 23063338.
- ^ "Dimethandrolone undecanoate shows promise as a male birth control pill". Endocrine Society. Archived from the original on 21 March 2018. Retrieved 20 March 2018.
- ^ S2CID 37424524.
- S2CID 23592351.
262. Gebauer, J., Hodkovicova, N., Tosnerova, K., Skoupa, K., Batik, A., Bartejsova, I., Charvatova, M., Leva, L., Jarosova, R., Sladek, Z., Faldyna, M., & Stastny, K. (2024). Anabolic steroids induced changes at the level of protein expression: Effects of prolonged administration of testosterone and nandrolone to pigs. Environmental Toxicology and Pharmacology, 107. Scopus. https://doi.org/10.1016/j.etap.2024.104422
263. Wartalski, K., Wiater, J., Maciak, P., Pastuła, A., Lis, G. J., Samiec, M., Trzcińska, M., & Duda, M. (2024). Anabolic Steroids Activate the NF-κB Pathway in Porcine Ovarian Putative Stem Cells Independently of the ZIP-9 Receptor: International Journal of Molecular Sciences. International Journal of Molecular Sciences, 25(5), 2833. https://doi.org/10.3390/ijms25052833
Further reading
- Daniels RC (1 February 2003). The Anabolic Steroid Handbook. RCD Books. p. 80. ISBN 0-9548227-0-6.
- Gallaway S (15 January 1997). The Steroid Bible (3rd Sprl ed.). Belle Intl. p. 125. ISBN 1-890342-00-9.
- Llewellyn W (28 January 2007). Anabolics 2007: Anabolic Steroid Reference Manual (6th ed.). Body of Science. p. 988. ISBN 978-0-9679304-6-6.
- Roberts A, Clapp B (January 2006). Anabolic Steroids: Ultimate Research Guide. Anabolic Books, LLC. p. 394. ISBN 1-59975-100-3.
- Yesalis CE (2000). Anabolic Steroids in Sport and Exercise. Human Kinetics. ISBN 0-88011-786-9.
- Fragkaki AG, Angelis YS, Koupparis M, Tsantili-Kakoulidou A, Kokotos G, Georgakopoulos C (February 2009). "Structural characteristics of anabolic androgenic steroids contributing to binding to the androgen receptor and to their anabolic and androgenic activities. Applied modifications in the steroidal structure". Steroids. 74 (2): 172–197. S2CID 41356223.
- McRobb L, Handelsman DJ, Kazlauskas R, Wilkinson S, McLeod MD, Heather AK (May 2008). "Structure-activity relationships of synthetic progestins in a yeast-based in vitro androgen bioassay". The Journal of Steroid Biochemistry and Molecular Biology. 110 (1–2): 39–47. S2CID 5612000.
- Schänzer W (July 1996). "Metabolism of anabolic androgenic steroids". Clinical Chemistry. 42 (7): 1001–1020. PMID 8674183.
- Tygart TT (December 2009). "Steroids, the Media, and Youth". Prevention Researcher Integrated Research Services, Inc. 16 (7–9). SIRS Researcher. Archived from the original on 29 November 2014. Retrieved 24 November 2013.
- Eisenhauer L (7 November 2005). "Do I Look OK?". St. Louis, MO: St. Louis Post-Dispatch. Archived from the original on 2 December 2013. Retrieved 25 October 2010.
External links
 Media related to Anabolic-androgenic steroids at Wikimedia Commons
Media related to Anabolic-androgenic steroids at Wikimedia Commons