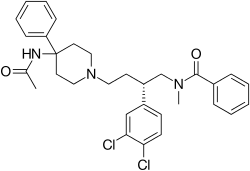
Saredutant
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Saredutant (SR-48,968) is a
phase III clinical trials. However, in May 2009, Sanofi-Aventis published its quarterly results and announced the cessation of 14 research/development projects, among which was saredutant for the treatment of major depressive disorder.[2]
See also
- GR-159,897
- Ibodutant
- Nepadutant
References
- PMID 22776916.
- ^ "Letter to the stockholders of Sanofi-Aventis" (PDF). May 2009. Archived from the original (PDF) on 21 December 2010.
