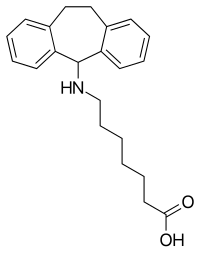
Amineptine
 | |
| Clinical data | |
|---|---|
| Trade names | Survector, others |
| Other names | S-1694 |
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
Renal | |
| Identifiers | |
| |
JSmol) | |
SMILES
| |
| |
| (verify) | |
Amineptine, formerly sold under the brand name Survector among others, is an atypical antidepressant of the tricyclic antidepressant (TCA) family.[4][5] It acts as a selective and mixed dopamine reuptake inhibitor and releasing agent, and to a lesser extent as a norepinephrine reuptake inhibitor.[4][5]
Amineptine was developed by the French Society of Medical research in the 1960s. effect experienced by some patients.
After its release into the European market, cases of hepatotoxicity emerged, some serious. This, along with the potential for abuse, led to the suspension of the French marketing authorization for Survector in 1999.[8]
Amineptine is illegal in both Germany and the United States.
Medical uses
Amineptine was approved in France for severe
Contraindications
- Chorea
- Hypersensitivity: Known hypersensitivity to amineptine, in particular antecedents of hepatitis after dosage of the product.
- MAO inhibitors
Precautions for use
Warnings and precautions before taking amineptine:[10]
- Breast feeding
- Children less than 15-year of age
- General anaesthesia: Discontinue the drug 24 to 48 hours before anaesthesia.[citation needed]
- Official sports/Olympic Games: Prohibited substance.
- 7 March Official Journal 2000.
- Pregnancy (first trimester)[citation needed]
Effects on the fetus
- Lacking information in humans
- Non-teratogenicin rodents
Side effects
Dermatological
Severe
Psychiatric
Psychomotor excitation can very rarely occur with this drug.
- Insomnia
- Irritability
- Nervousness
- Suicidalideation. Seen early in the treatment, by lifting of psychomotor inhibition.
Abuse and dependence
The risk of
Withdrawal
Pharmacodependence is very common with amineptine compared to other antidepressants.[22] A variety of psychological symptoms can occur during withdrawal from amineptine,[23] such as anxiety and agitation.[24]
Cardiovascular
Very rarely:
- Arterial hypotension
- Palpitations
- Vasomotor episode
Hepatic
Amineptine can rarely cause hepatitis, of the cytolytic, cholestatic varieties.[25] Amineptine-induced hepatitis, which is sometimes preceded by a rash, is believed to be due to an immunoallergic reaction.[26] It resolves upon discontinuation of the offending drug.[25] The risk of getting this may or may not be genetically determined.[27]
Additionally, amineptine is known to rarely elevate transaminases, alkaline phosphatase, and bilirubin.[28]
Mixed hepatitis, which is very rare, generally occurs between the 15th and 30th day of treatment of amineptine. Often preceded by sometimes intense abdominal pains, nausea, vomiting or a rash, the jaundice is variable. Hepatitis is either of mixed type or with cholestatic prevalence. The evolution was, in all the cases, favorable to the discontinuation of the drug. The mechanism is discussed (immunoallergic and/or toxic).[29]
In circa 1994 Spain, there was a case associating acute pancreatitis and mixed hepatitis, after three weeks of treatment.[30]
Lazaros and colleagues at the Western Attica General Hospital in Athens, Greece reported two cases of drug induced hepatitis 18 and 15 days of treatment.[31]
One case of cytolytic hepatitis occurred after ingestion of only one tablet.[32]
Gastrointestinal
- Acute pancreatitis (very rare) A case associating acute pancreatitis and mixed hepatitis after three weeks of treatment.[30]
Immunological
A case of
Pharmacology
Pharmacodynamics
| Site | Ki (nM) | Species | Ref |
|---|---|---|---|
| SERT | >100,000 (IC50) | Rat | [35] |
| NET | 10,000 (IC50) 3,560 |
Rat Canine |
[35][36] [37] |
| DAT | 1,000–1,400 (IC50) 3,330 |
Rat Canine |
[35][38][36] [37] |
| 5-HT1A | >100,000 | Rat | [39] |
| 5-HT2A | 74,000 | Rat | [39] |
| α1 | >100,000 | Rat | [39] |
| α2 | >100,000 | Rat | [39] |
β |
>100,000 | Rat | [39] |
D1 |
>100,000 | Canine | [35] |
D2 |
>100,000 | Rat/canine | [35][39] |
| H1 | >100,000 13,000 |
Rat Guinea pig |
[39] [40] |
| mACh | >100,000 | Rat | [39] |
| Values are Ki (nM), unless otherwise noted. The smaller the value, the more strongly the drug binds to the site. | |||
Amineptine
No human data appear to be available for binding or inhibition of the monoamine transporters by amineptine.[42]
Pharmacokinetics
Society and culture
Brand names
Amineptine has been sold under a variety of brand names including Survector, Maneon, Directim, Neolior, Provector, and Viaspera.
Legal status
It had been proposed that Amineptine become a Schedule I controlled substance in the United States in July 2021.[43] This announcement was followed by the placement of Amineptine into Schedule I. [44]
Research
Wakefulness
Amineptine shows wakefulness-promoting effects in animals and might be useful in the treatment of narcolepsy.[45][46][47][48]
References
- ^ Anvisa (31 March 2023). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 4 April 2023). Archived from the original on 3 August 2023. Retrieved 16 August 2023.
- ^ S2CID 25992333.
- ^ S2CID 31069503.
- ^ S2CID 32862145.
- ^ S2CID 26550661.
- ^ DE 2011806, Fresnes C, Malen JS, "New Tricyclic Derivatives and Process for their Manufacture", issued 3 December 1981, assigned to Science Union et Cie. Societe Francaise de Recherche Medical, Suresnes (Frankreich)
- ISBN 978-0-8155-1144-1. Archived from the original on 23 October 2005. Retrieved 29 October 2005.[page needed]
- ^ "Docket No. 02N-0101". U.S. Food and Drug Administration. 9 April 2002. Retrieved 30 January 2014.
- ^ Doctissimo (2005). "SURVECTOR – Amineptine" (in French). Archived from the original on 9 March 2005. Retrieved 27 October 2005.
- ^ Amineptine Medication – Uses, Side Effects and Precautions of Amineptine. Health-care-information.org. Retrieved on 28 September 2013
- PMID 2977079.
- PMID 2977080.
- PMID 2977081.
- PMID 2977082.
- PMID 2977083.
- S2CID 32936080.
- PMID 2534534.
- ^ S2CID 39394890.
- PMID 2260032.
- PMID 7227285.
- PMID 2265603.
- PMID 9231505.
- PMID 2260032.
- PMID 2265603.
- ^ PMID 7454584.
- PMID 3044468.
- PMID 9204405.
- PMID 10533145.
- ^ Concours Med 1982; 104:5733-5734[verification needed]
- ^ PMID 8189020.
- PMID 8884331.
- PMID 1397859.
- PMID 2488491.
- ^ Roth BL, Driscol, J. "PDSP Ki Database". Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Retrieved 14 August 2017.
- ^ S2CID 10947713.
- ^ PMID 3708219.
- ^ PMID 11382857.
- PMID 8676127.
- ^ PMID 6464782.
- ^ PMID 6141518.
- ^ S2CID 23014661.
- PMID 19557250.
- ^ "2021 - Placement of Amineptine in Schedule I". Archived from the original on 3 December 2021. Retrieved 30 November 2021.
- ^ "Federal Register". Federal Register. National Archives & Drug Enforcement Administration. 17 November 2022.
- ISBN 978-3-319-23738-1.
- PMID 18078360.
- PMID 23065655.
- PMID 11382857.
