GABAB receptor
(Redirected from
GABAB
)Chr. 6 p21.3 | |||||||
|---|---|---|---|---|---|---|---|
| |||||||
Chr. 9 q22.1-22.3 | |||||||
|---|---|---|---|---|---|---|---|
| |||||||
GABAB receptors (GABABR) are
G-proteins to potassium channels.[1] The changing potassium concentrations hyperpolarize the cell at the end of an action potential. The reversal potential of the GABAB-mediated IPSP (inhibitory postsynaptic potential) is −100 mV, which is much more hyperpolarized than the GABAA IPSP. GABAB receptors are found in the central nervous system and the autonomic division of the peripheral nervous system.[2]
The receptors were first named in 1981 when their distribution in the CNS was determined, which was determined by Norman Bowery and his team using radioactively labelled baclofen.[3]
Functions
GABABRs stimulate the opening of
GIRKs, which brings the neuron closer to the equilibrium potential of K+. This reduces the frequency of action potentials which reduces neurotransmitter release.[citation needed
] Thus GABAB receptors are inhibitory receptors.
GABAB receptors also reduces the activity of
Gi/G0 α subunits.[4]
GABAB receptors are involved in behavioral actions of ethanol,[5][6] gamma-hydroxybutyric acid (GHB),[7] and possibly in pain.[8] Recent research suggests that these receptors may play an important developmental role.[9]
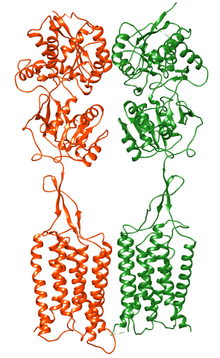
Structure
GABAB Receptors are similar in structure to and in the same receptor family with
Cryo-electron microscopy structures of the full length GABAB receptor in different conformational states from inactive apo to fully active have been obtained. Unlike Class A and B GPCRs, phospholipids bind within the transmembrane bundles and allosteric modulators bind at the interface of GABAB1 and GABAB2 subunits.[13][14][15][16][17][18][19]
Ligands
GABA
GHB
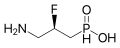
Agonists
- GABA
- analogue which acts as a selective agonist of GABAB receptors, and is used as a muscle relaxant. However, it can aggravate absence seizures, and so is not used in epilepsy.
- gamma-Hydroxybutyrate(GHB)
- Phenibut
- 4-Fluorophenibut
- Isovaline
- 3-Aminopropylphosphinic acid
- Lesogaberan
- SKF-97541: 3-Aminopropyl(methyl)phosphinic acid, 10× more potent than baclofen as GABAB agonist, but also GABAA-ρantagonist
- Taurine
- CGP-44532

Positive Allosteric Modulators


Antagonists
- Homotaurine[24]
- Ginsenosides[25]
- 2-OH-saclofen
- Saclofen
- Phaclofen
- SCH-50911
- 2-Phenethylamine
- CGP-35348
- CGP-52432: 3-([(3,4-Dichlorophenyl)methyl]amino]propyl) diethoxymethyl)phosphinic acid, CAS# 139667-74-6
- CGP-55845: (2S)-3-([(1S)-1-(3,4-Dichlorophenyl)ethyl]amino-2-hydroxypropyl)(phenylmethyl)phosphinic acid, CAS# 149184-22-5
- SGS-742[26][27]
See also
- GABA receptor
- GABAA receptor
References
- S2CID 6433030.
- PMID 21833169.
- S2CID 4335907.
- OCLC 903234097.
- PMID 12692303.
- PMID 15564584.
- PMID 16129424.
- PMID 15140638.
- PMID 16054235.
- ^ a b MRC (Medical Research Council). 2003. Glutamate receptors: Structures and functions. University of Brisotol Centre for Synaptic Plasticity.
- ^ Purves D, Augustine GJ, Fitzpatrick D, Katz LC, LaMantia AS, McNamara JO, Williams SM (2001). "7. Neurotransmitter Receptors and Their Effects". Neuroscience (Second ed.). Sinauer Associates, Inc.
- ^ S2CID 4345443.
- PMID 34049877.
- PMID 32555460.
- PMID 32580208.
- S2CID 219183617.
- S2CID 220050861.
- S2CID 222841520.
- PMID 33911284.
- PMID 11641424.
- PMID 17894647.
- PMID 18445779.
- S2CID 26152839.
- PMID 6652358.
- PMID 8026706.
- PMID 15451390.
- PMID 15675610.
