Clonidine
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˈklɒnədiːn/ |
| Trade names | Catapres, Kapvay, Nexiclon, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682243 |
| License data | |
| Pregnancy category |
|
| Routes of administration | Oral, epidural, intravenous (IV), transdermal, topical |
| Drug class | Centrally acting α2A-agonist hypotensive agent |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 70–80% (oral),[2][3] 60–70% (transdermal)[4] |
| Protein binding | 20–40%[5] |
| Metabolism | Liver to inactive metabolites,[5] 2/3 CYP2D6[9] |
| Onset of action | IR: 30–60 minutes after an oral dose[6] |
| Elimination half-life | IR: 12–16 hours; 41 hours in kidney failure,[7][8] 48 hours for repeated dosing[4] |
| Excretion | Urine (72%)[5] |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Clonidine, sold under the brand name Catapres among others, is an α2A-adrenergic agonist[10] medication used to treat high blood pressure, ADHD, drug withdrawal (alcohol, opioids, or nicotine), menopausal flushing, diarrhea, spasticity, and certain pain conditions.[11] The drug is often prescribed off-label for tics. It is used orally (by mouth), by injection, or as a transdermal skin patch.[11] Onset of action is typically within an hour with the effects on blood pressure lasting for up to eight hours.[11]
Common side effects include dry mouth, dizziness, headaches, hypotension, and sleepiness.
Clonidine was patented in 1961 and came into medical use in 1966.
Medical uses

Clonidine is used to treat high blood pressure, attention deficit hyperactivity disorder (ADHD), drug withdrawal (alcohol, opioids, or smoking), menopausal flushing, diarrhea, and certain pain conditions. It also sees some use off-label for episodic insomnia, restless-legs syndrome, and anxiety, among other uses.[11]
Resistant hypertension
Clonidine may be effective for lowering blood pressure in people with resistant hypertension.[18]
Clonidine works by slowing the
Attention deficit hyperactivity disorder
Clonidine may improve symptoms of attention deficit hyperactivity disorder in some people but causes many adverse effects and the beneficial effect is modest.[20] In Australia, clonidine is an accepted but not approved use for ADHD by the TGA.[21] Clonidine, along with methylphenidate, has been studied for treatment of ADHD.[22][23][24] While not as effective as methylphenidate in treating ADHD, clonidine does offer some benefit;[22] it can also be useful in combination with stimulant medications.[25] Some studies show clonidine to be more sedating than guanfacine, which may be better at bedtime along with an arousing stimulant in the morning.[26][27] Clonidine has been used to reduce sleep disturbances in ADHD, including to help offset stimulant-associated insomnia.[28][29][30][31] Unlike stimulant medications, clonidine is regarded as having no abuse potential, and may even be used to reduce abuse of drugs including nicotine and cocaine.[32]
In the US, only the extended-release form of clonidine is approved for ADHD treatment.[33]
Drug withdrawal
Clonidine may be used to ease drug withdrawal symptoms associated with abruptly stopping the long-term use of
Clonidine has also been suggested as a treatment for rare instances of dexmedetomidine withdrawal.[39]
Spasticity
Clonidine has some role in the treatment of spasticity, acting principally by inhibiting excessive sensory transmission below the level of injury[
Clonidine suppression test
The reduction in circulating norepinephrine by clonidine was used in the past as an investigatory test for
Other uses
Clonidine also has several
Injection of α2 receptor agonists into the knee joint space, including clonidine, may reduce the severity of knee pain after arthroscopic knee surgery.[63]
Light-activated derivatives of clonidine (adrenoswitches) have been developed for research purposes and shown to control pupillary reflex with light in blind mice by topical application.[64]
Pregnancy and breastfeeding
It is classified by the TGA of Australia as pregnancy category B3, which means that it has shown some detrimental effects on fetal development in animal studies, although the relevance of this to human beings is unknown.[65] Clonidine appears in high concentration in breast milk; a nursing infant's serum clonidine concentration is approximately 2/3 of the mother's.[66] Caution is warranted in women who are pregnant, planning to become pregnant, or are breastfeeding.[67]
Adverse effects
The principal adverse effects of clonidine are sedation, dry mouth, and hypotension (low blood pressure).[5]
Very common (>10% frequency):
- Dizziness
- Orthostatic hypotension
- Somnolence (dose-dependent)
- Dry mouth
- Headache (dose-dependent)
- Fatigue
- Skin reactions (if given transdermally)
- Hypotension
Common (1–10% frequency):
- Anxiety
- Constipation
- Sedation (dose-dependent)
- Nausea/vomiting
- Malaise
- Abnormal LFTs
- Rash
- Weight gain/loss
- Pain below the ear (from salivary gland)
- Erectile dysfunction
Uncommon (0.1–1% frequency):
- Delusional perception
- Hallucination
- Nightmare
- Paresthesia
- Sinus bradycardia
- Raynaud's phenomenon
- Pruritus
- Urticaria
Rare (<0.1% frequency):
- Gynaecomastia
- Impaired ability to cry
- Atrioventricular block
- Nasal dryness
- Colonic pseudo-obstruction
- Alopecia
- Hyperglycemia
Withdrawal
Because clonidine suppresses sympathetic outflow, resulting in lower blood pressure, sudden discontinuation can result in acute hypertension due to a rebound in sympathetic outflow. In extreme cases, this can result in a hypertensive crisis, which is a medical emergency.[69]
Clonidine therapy should generally be gradually tapered when discontinuing therapy to avoid
Pharmacology
| Site | Ki (nM) | Species | Ref |
|---|---|---|---|
| NET | >1,000 | Human | [73] |
| 5-HT1B | >10,000 | Rat | [74] |
| 5-HT2A | >10,000 | Human | [72] |
| α1A | 316.23 | Human | [73] |
| α1B | 316.23 | Human | [73] |
| α1D | 125.89 | Human | [73] |
| α2A | 35.48 – 61.65 | Human | [73][75] |
| α2B | 69.18 – 309.0 | Human | [75][73] |
| α2C | 134.89 – 501.2 | Human | [75][73] |
D1 |
> 10,000 | Rat | [76] |
| I1 | 31.62 | Bovine | [73] |
| I2 (cortex) | >1,000 | Rat | [73] |
| MAO-A | >1,000 | Rat | [73] |
| MAO-B | >1,000 | Rat | [73] |
| σ | >10,000 | Guinea Pig | [77] |
| The Ki refers to a drug's affinity for a receptor. The smaller the Ki, the higher the affinity for that receptor.[78] Reported imidazoline-2 binding is measured in the cortex — I2 receptor bindings measured in stomach membranes are much lower.[79] | |||
Mechanism of action
Clonidine crosses the blood–brain barrier.[7]
High blood pressure
Clonidine treats high blood pressure by stimulating
Clonidine also acts as an agonist at imidazoline-1 (I1) receptors in the brain, and it is hypothesized that this effect may contribute to reducing blood pressure by reducing signaling in the sympathetic nervous system; this effect acts upstream of the central α2 agonist effect of clonidine.[10]: 201–203 [80]
Clonidine may also cause bradycardia, theoretically by increasing signaling through the vagus nerve. When given intravenously, clonidine can temporarily increase blood pressure by stimulating α1 receptors in smooth muscles in blood vessels.[81] This hypertensive effect is not usual when clonidine is given orally or by the transdermal route.[10]: 201–203
Plasma concentration of clonidine exceeding 2.0 ng/mL does not provide further blood pressure reduction.[82]
Attention deficit hyperactivity disorder
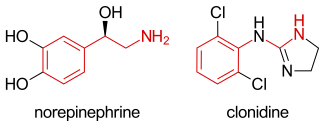
In the setting of attention deficit hyperactivity disorder (ADHD), clonidine's molecular mechanism of action occurs due to its agonism at the α2A adrenergic receptor, the subtype of the adrenergic receptor that is most principally found in the brain. Within the brain, the α2A adrenergic receptors are found within the prefrontal cortex (PFC), among other areas. The α2A adrenergic receptors are found on the presynaptic cleft of a given neuron, and, when activated by an agonist, the effect on downstream neurons is inhibitory. The inhibition is accomplished by preventing the secretion of the neurotransmitter norepinephrine. Thus, clonidine's agonism on α2A adrenergic receptors in the PFC inhibits the action of downstream neurons by preventing the secretion of norepinephrine.[83]
This mechanism is similar to the brain's physiological inhibition of PFC neurons by the
Growth hormone test
Clonidine stimulates release of GHRH hormone from the hypothalamus, which in turn stimulates pituitary release of growth hormone.[84] This effect has been used as part of a "growth hormone test," which can assist with diagnosing growth hormone deficiency in children.[85]
Pharmacokinetics
After being ingested, clonidine is absorbed into the blood stream rapidly with an overall
Measurements of the half-life of clonidine vary widely, between 6 and 23 hours, with the half-life being greatly affected by and prolonged in the setting of poor kidney function.[86] Variations in half-life may be partially attributable to CYP2D6 genetics.[9] Some research has suggested the half-life of clonidine is dose dependent and approximately doubles upon chronic dosing,[91] while other work contradicts this.[4] Following a 0.3 mg oral dose, a small study of five patients by Dollery et al. (1976) found half-lives ranging between 6.3 and 23.4 hours (mean 12.7).[92] A similar N=5 study by Davies et al. (1977) found a narrower range of half-lives, between 6.7 and 13 hours (average 8.6 hours),[2] while an N=8 study by Keraäen et al. that included younger patients found a somewhat shorter average half-life of 7.5 hours.[93]
History
Clonidine was introduced in 1966.[94] It was first used as a hypertension treatment under the trade name of Catapres.[95]
Society and culture
Brand names
As of June 2017, clonidine was marketed under many brand names worldwide: Arkamin, Aruclonin, Atensina, Catapin, Catapres, Catapresan, Catapressan, Chianda, Chlofazoline, Chlophazolin, Clonid-Ophtal, Clonidin, Clonidina, Clonidinã, Clonidine, Clonidine hydrochloride, Clonidinhydrochlorid, Clonidini, Clonidinum, Clonigen, Clonistada, Clonnirit, Clophelinum, Dixarit, Duraclon, Edolglau, Haemiton, Hypodine, Hypolax, Iporel, Isoglaucon, Jenloga, Kapvay, Klofelino, Kochaniin, Lonid, Melzin, Menograine, Normopresan, Paracefan, Pinsanidine, Run Rui, and Winpress.[96] It was marketed as a combination drug with chlortalidone as Arkamin-H, Bemplas, Catapres-DIU, and Clorpres, and in combination with bendroflumethiazide as Pertenso.[96]
References
- FDA. Retrieved 22 October 2023.
- ^ S2CID 5566079.
- ^ "Catapres- clonidine hydrochloride tablet". DailyMed. 6 September 2016. Archived from the original on 4 August 2020. Retrieved 21 December 2019.
The pharmacokinetics of clonidine is dose-proportional in the range of 100 to 600 µg.The absolute bioavailability of clonidine on oral administration is 70% to 80%. Peak plasma clonidine levels are attained in approximately 1 to 3 hours.
- ^ S2CID 24783447.
- ^ a b c d "clonidine (Rx) - Catapres, Catapres-TTS, more." Medscape Reference. WebMD. Archived from the original on 4 December 2020. Retrieved 10 November 2013.
- ^ "Catapres- clonidine hydrochloride tablet". DailyMed. 6 September 2016. Archived from the original on 4 August 2020. Retrieved 21 December 2019.
Catapres tablets act relatively rapidly. The patient's blood pressure declines within 30 to 60 minutes after an oral dose, the maximum decrease occurring within 2 to 4 hours.
- ^ a b "Catapres- clonidine hydrochloride tablet". DailyMed. 6 September 2016. Archived from the original on 4 August 2020. Retrieved 21 December 2019.
Following intravenous administration, clonidine displays biphasic disposition with a distribution half-life of about 20 minutes and an elimination half-life ranging from 12 to 16 hours. The half-life increases up to 41 hours in patients with severe impairment of renal function. Clonidine crosses the placental barrier. It has been shown to cross the blood–brain barrier in rats.
- ^ "Kapvay". RxList. Archived from the original on 12 October 2017. Retrieved 30 October 2014.
- ^ PMID 20570945.
- ^ ISBN 9781259584732.
- ^ a b c d e f g h "Clonidine Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Archived from the original on 3 February 2019. Retrieved 2 February 2019.
- ^ ISBN 9780857113382.
- S2CID 40756251.
- .
- ISBN 9783527607495. Archivedfrom the original on 29 April 2023. Retrieved 12 September 2020.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Clonidine - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- PMID 29863319.
- ^ "CATAPRES- clonidine hydrochloride tablet". DailyMed. 6 September 2016. Archived from the original on 4 August 2020. Retrieved 21 December 2019.
Slowing of the pulse rate has been observed in most patients given clonidine, but the drug does not alter normal hemodynamic response to exercise. Other studies in patients have provided evidence of a reduction in plasma renin activity and in the excretion of aldosterone and catecholamines.
- PMID 10596256.
- ISBN 978-0-9805790-9-3.
- ^ PMID 18182963.
- PMID 18182964.
- PMID 23318985.
- PMID 21555501.
- PMID 10192826.
- from the original on 9 August 2014. Retrieved 1 August 2014.
- ^ S2CID 742140.
- S2CID 35000868.
- ISBN 978-1-4613-7214-1.
- PMID 8169189.
- S2CID 207580823.
- ^ "Highlights of Prescribing Information: Kapvay" (PDF). US FDA. Retrieved 31 March 2024.
- PMID 24151426.
- ^ Giannini AJ (1997). Drugs of Abuse (2nd ed.). Los Angeles: Practice Management Information.
- PMID 15266422.
- S2CID 40652097.
- PMID 30667476.
- S2CID 31570614.
- PMID 25750484.
- PMID 18443642.
- ^ PMID 11723871.
- ^ PMID 12788870.
- PMID 2960712.
- PMID 7937367.
- S2CID 32148292.
- S2CID 33940152.
- S2CID 1607064.
- PMID 17506224.
- S2CID 23494905.
- S2CID 31292297.
- S2CID 18885497.
- S2CID 85093127.
- S2CID 72544106.
- ^ "Treatment and Management of RLS". www.medscape.org. WebMD LLC. Archived from the original on 29 September 2017. Retrieved 3 October 2018.
- from the original on 26 July 2011. Retrieved 12 February 2012.
- PMID 22683276.
- ^ "Clonidine Oral Uses". WebMD. Archived from the original on 25 October 2007. Retrieved 30 May 2007.
- ^ "Clonidine". Drugs.com. Archived from the original on 14 April 2017. Retrieved 25 May 2017.
- PMID 27134659.
- PMID 24619591.
- PMID 29547576.
- S2CID 3861878.
- PMID 33103317.
- ^ a b "CATAPRES® 150 TABLETS CATAPRES® AMPOULES" (PDF). TGA eBusiness Services. Boehringer Ingelheim Pty Limited. 28 February 2013. Archived from the original on 16 January 2017. Retrieved 27 November 2013.
- from the original on 5 December 2020. Retrieved 5 January 2019.
- ^ "Clonidine". Prescription Marketed Drugs. www.drugsdb.eu. Archived from the original on 28 March 2012. Retrieved 2 August 2011.
- ^ "Clonidine 25 mcg Tablets BP - Summary of Product Characteristics (SPC)". electronic Medicines Compendium. Sandoz Limited. 2 August 2012. Archived from the original on 2 December 2013. Retrieved 27 November 2013.
- ^ Brayfield, A, ed. (13 January 2014). "Clonidine". Martindale: The Complete Drug Reference. London, UK: Pharmaceutical Press. Archived from the original on 28 August 2021. Retrieved 28 June 2014.
- ISBN 978-0-07-142280-2.
- PMID 18295156.
- ^ a b Roth BL, Driscol J. "PDSP Ki Database". Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Archived from the original on 14 November 2022. Retrieved 14 August 2017.
- ^ PMID 11082457.
- PMID 1738111.
- ^ PMID 9605427.
- PMID 2138666.
- PMID 2877462.
- ISBN 9780123695215.
- PMID 9536007.
- PMID 9374795.
- PMID 25849473.
- ^ "CATAPRES- clonidine hydrochloride tablet". DailyMed. 6 September 2016. Archived from the original on 4 August 2020. Retrieved 21 December 2019.
The antihypertensive effect is reached at plasma concentrations between about 0.2 and 2.0 ng/mL in patients with normal excretory function. A further rise in the plasma levels will not enhance the antihypertensive effect.
- ^ PMID 20652773.
- PMID 1901390.
- ^ "Growth Hormone Test". www.cincinnatichildrens.org. Cincinnati Children's Hospital Medical Center. Archived from the original on 14 October 2018. Retrieved 13 October 2018.
- ^ S2CID 28405271.
- ISBN 9780781768795.
- PMID 16489364.
- PMID 6128647.
- S2CID 41743285.
- PMID 7332729.
- S2CID 39473828.
- S2CID 24702183.
- .
- ^ "Clonidine: Drug Uses, Dosage & Side Effects - Drugs.com". Drugs.com. Archived from the original on 14 April 2017. Retrieved 10 December 2017.
- ^ a b "Clonidine brand names". Drugs.com. Archived from the original on 6 August 2017. Retrieved 16 June 2017.
External links
- Alpha-2 agonists in ADHD
