Verapamil
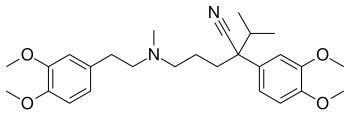 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /vɛˈræpəmɪl/ ve-RAP-ə-mil |
| Trade names | Isoptin, Calan, others[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a684030 |
| License data | |
| Pregnancy category |
|
intravenous | |
| Drug class | Calcium channel blocker |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 35.1% |
| Metabolism | Liver |
| Elimination half-life | 2.8–7.4 hours[6] |
| Excretion | Kidney: 11% |
| Identifiers | |
| |
JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| (verify) | |
Verapamil, sold under various trade names,
Common side effects include headache,
Verapamil was approved for medical use in the United States in 1981.
Medical uses
Verapamil is used for controlling
Verapamil is also used for the treatment of
Verapamil is a
Verapamil is also used intra-arterially to treat cerebral vasospasm.[22] Verapamil is used to treat the condition cluster headache.[23] Tentative evidence supports the use of verapamil topically to treat plantar fibromatosis.[24]
Verapamil use in people with recent onset type-1 diabetes may improve pancreatic beta cell function. In a meta-analysis involving data from 2 randomized controlled trials (113 patients with recent onset type-1 diabetes), Dutta et al. demonstrated use of verapamil over 1 year was associated with significantly higher C-peptide area under the curve levels at 1-year [MD 0.27 nmol/L (95%CI: 0.19-0.35); P < 0.01]. Higher C-peptide levels means better pancreatic insulin production and beta cell function.[25]
Verapamil has been reported to be effective in both short-term[26] and long-term treatment of mania and hypomania.[27] Addition of magnesium oxide to the verapamil treatment protocol enhances the antimanic effect.[28]
Contraindications
Use of verapamil is generally avoided in people with severe left ventricular dysfunction,
Side effects
The most common side effect of verapamil is constipation (7.3%). While the definite mechanism by which Verapamil causes constipation has not been studied, studies have been conducted to rule out mechanisms of actions that might yield this adverse effect. In a study conducted by The National Library of Medicine titled, "Effect of Verapamil on the Human Intestinal Transit", the study found that verapamil does not have an effect on upper GI transit but rather in the colon.[30]
Other side effects include
Overdose
Acute overdose is often manifested by nausea,
Mechanism of action
Verapamil's
Calcium channels are also present in the
Calcium channel blockers like verapamil dilate the coronary blood vessels, which increases the supply of blood and oxygen to the heart. They also cause dilatation of systemic peripheral vessels as well, causing a reduction in the workload of the heart. Thereby reducing myocardial oxygen consumption.[4]
Cluster headaches
Preventive therapy with verapamil is believed to work because it has an effect on the circadian rhythm and on CGRPs. As CGRP-release is controlled by voltage-gated calcium channels.[35]
Pharmacokinetic details
More than 90% of verapamil is absorbed when given orally,
Veterinary use
Intra-abdominal adhesions are common in rabbits following surgery. Verapamil can be given postoperatively in rabbits which have suffered trauma to abdominal organs to prevent formation of these adhesions.[36][37][38] Such effect was not documented in another study with ponies.[39]
Uses in cell biology
Verapamil inhibits the ATP-binding cassette (ABC) transporter family of proteins found in stem cells and has been used to study cancer stem cells (CSC) within head and neck squamous cell carcinomas.[40]
Verapamil is also used in
References
- ^ a b "Verapamil". www.drugs.com. Archived from the original on 1 August 2017. Retrieved 14 December 2016.
- ^ a b "Verapamil Use During Pregnancy". Drugs.com. 18 November 2019. Retrieved 26 March 2020.
- ^ "Securon SR - Summary of Product Characteristics (SmPC)". (emc). 17 May 2017. Retrieved 26 March 2020.
- ^ a b c d e f g h "Calan- verapamil hydrochloride tablet, film coated". DailyMed. 17 December 2019. Retrieved 26 March 2020.
- ^ Human Medicines Evaluation Division (14 October 2020). "Active substance(s): verapamil" (PDF). List of nationally authorised medicinal products. European Medicines Agency.
- ISBN 978-1-4557-0101-8.
The elimination half-life of standard verapamil tablets is usually 3 to 7 hours,...
- ^ a b c d e f "Verapamil Hydrochloride". The American Society of Health-System Pharmacists. Archived from the original on 21 December 2016. Retrieved 8 December 2016.
- S2CID 22522914.
- S2CID 28302667.
- ^ ISBN 9789241547659.
- ^ "Isoptin: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Retrieved 26 March 2020.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Verapamil - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ISBN 978-1609137137.
- PMID 30860730.
- PMID 21886903.
- PMID 24352797.
- ISBN 978-1609137137.
- from the original on 13 November 2015. Retrieved 4 November 2015.
- PMID 20616179.
- ISBN 978-0-7817-9685-9. Archivedfrom the original on 8 June 2013. Retrieved 14 November 2010.
- PMID 30774465.
- PMID 37583402.
- PMID 6439057.
- S2CID 34536914.
- S2CID 18216795.
- ^ "Securon 2.5 mg/ml IV Intravenous Injection - Summary of Product Characteristics (SmPC)". (emc). 24 November 2016. Retrieved 26 March 2020.
- S2CID 1007332.
- S2CID 41746099.
- S2CID 10351499.
- ^ Baselt R (2008). Disposition of Toxic Drugs and Chemicals in Man (8th ed.). Foster City, California: Biomedical Publications. pp. 1637–39.
- S2CID 205457755.
- S2CID 198193843.
- PMID 6704142.
- S2CID 10911875.
- PMID 2296179.
- PMID 8116206.
- ^ PMID 20625515.
- PMID 8725386.
