Loxapine
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Loxitane, Adasuve |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682311 |
| License data | |
| Routes of administration | By mouth, inhalation, intramuscular |
| Drug class | Antipsychotic |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 96.8%[3] |
| Metabolism | Extensive Liver; active metabolites include amoxapine and 8-hydroxyloxapine. Inhibits P-gp and is a substrate of CYP1A2, CYP3A4 and CYP2D6[3] |
| Elimination half-life | 4 hours (oral); 7.61 hours (inhalation)[3] |
| Excretion | Majority are excreted within 24 hours, main route through urine (conjugated metabolites), small amounts through the feces (unconjugated metabolites) |
| Identifiers | |
| |
JSmol) | |
| Melting point | 109 to 110 °C (228 to 230 °F) |
| |
| |
| | |

Loxapine, sold under the brand names Loxitane and Adasuve (inhalation only) among others, is a
Loxapine may be metabolized by N-demethylation to amoxapine, a tricyclic antidepressant.[6]
Medical uses
The
A brief review of loxapine found no conclusive evidence that it was particularly effective in patients with schizophrenia.[8] A subsequent systematic review considered that the limited evidence did not indicate a clear difference in its effects from other antipsychotics.[9]
Available forms
Loxapine can be taken by mouth.[10] It is also available as an intramuscular injection and as a powder for inhalation.[7][10]
Side effects
Loxapine can cause side effects that are generally similar to that of other antipsychotic medications. These include, e.g., gastrointestinal problems (like constipation and abdominal pain), cardiovascular problems (like tachycardia), moderate likelihood of drowsiness (relative to other antipsychotics),[11] and movement problems (i.e. extrapyramidal symptoms [EPS]).[12] At lower dosages its propensity for causing EPS appears to be similar to that of atypical antipsychotics.[13] Although it is structurally similar to clozapine, it has much lower risk of agranulocytosis (which, even with clozapine, is 0.8%); however, mild and temporary fluctuations in blood leukocyte levels can occur.[14][15] Abuse of loxapine has been reported.[16]
The inhaled formulation of loxapine carries a low risk for a type of airway adverse reaction called bronchospasm that is not thought to occur when loxapine is taken by mouth.[7]
Pharmacology
Mechanism of action
Some scientists say loxapine is a "mid-potency" typical antipsychotic.[15] However, unlike most other typical antipsychotics, it has significant potency at the 5HT2A receptor (6.6 nM), which is similar to atypical antipsychotics like clozapine (5.35 nM). The higher likelihood of EPS with loxapine, compared to clozapine, may be due to its higher affinity for the D2 receptor compared to clozapine, which has one of the lowest binding affinities at the D2 receptor of any antipsychotic.[15]
| Site | LOX | AMX |
|---|---|---|
| 5-HT1A | 2,460 | ND |
| 5-HT1B | 388 | ND |
| 5-HT1D | 3,470 | ND |
| 5-HT1E | 1,400 | ND |
| 5-HT2A | 6.6 | 0.5 |
| 5-HT2C | 13 | 2 (rat) |
| 5-HT3 | 190 | ND |
| 5-HT5A | 780 | ND |
| 5-HT6 | 31 | 50 |
| 5-HT7 | 88 | 40 (rat) |
| α1A | 31 | ND |
| α1B | 53 | ND |
| α2A | 151 | ND |
| α2B | 108 | ND |
| α2C | 80 | ND |
| β1 | >10,000 | ND |
| β2 | >10,000 | ND |
| M1 | 120 | ND |
| M2 | 445 | ND |
| M3 | 211 | ND |
| M4 | 1,270 | ND |
| M5 | 166 | ND |
D1 |
54 | ND |
D2 |
11 | 21 |
D3 |
19 | 21 |
D4 |
8.4 | 21 |
D5 |
75 | ND |
| H1 | 2.2–4.9 | 7.9–25 |
| H2 | 208 | ND |
| H3 | 55,000 | >100,000 |
| H4 | 5,050–8,710 | 6,310 |
| SERT | >10,000 | 58 |
| NET | 5,700 | 16 |
| DAT | >10,000 | 58 |
| Values are Ki (nM). The smaller the value, the more strongly the drug binds to the site. | ||
Pharmacokinetics
Loxapine is metabolized to
The pharmacokinetics of loxapine change depending on how it is given. Intramuscular injections of loxapine lead to higher blood levels and area under the curve of loxapine than when it is taken by mouth.[19]
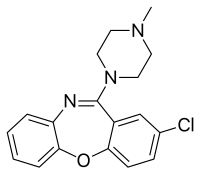
Chemistry
Loxapine is a

References
- FDA. Retrieved 22 Oct 2023.
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- ^ a b c d Truven Health Analytics, Inc. DrugPoint System (Internet) [cited 2013 Sep 21]. Greenwood Village, CO: Thomsen Healthcare; 2013.
- PMID 25859275.
- PMID 10340686.
- PMID 1860915.
- ^ a b c "ADASUVE Package Insert" (PDF). Galen US Inc.
- S2CID 27613339.
- PMID 17943763.
- ^ a b "LOXITANE Package Insert" (PDF). Watson Laboratories, Inc.
- ISBN 978-0-470-97948-8.
- PMID 17943763.
- S2CID 39718567.
- PMID 7041162.
- ^ PMID 8580115.
- PMID 6694719.
- ^ Roth BL, Driscol J. "PDSP Ki Database". Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Retrieved 14 August 2017.
- S2CID 14274150.
- ^ S2CID 21795809.
External links
- "Loxapine". Drug Information Portal. U.S. National Library of Medicine.
