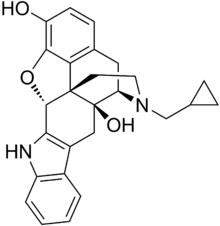
Naltrindole
Source: Wikipedia, the free encyclopedia.
Chemical compound
 | |
| Clinical data | |
|---|---|
| Routes of administration | IV |
| ATC code |
|
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Naltrindole is a highly potent, highly selective
antagonist used in biomedical research. In May 2012 a paper was published in Nature with the structure of naltrindole in complex with the mouse δ-opioid G-protein coupled receptor, solved by X-ray crystallography.[1]
Drug design
Since
phenyl group on its Phe4 residue, which was hypothesized to be the "address" sequence responsible for the opiate's delta opioid receptor affinity.[2] Thus, attachment of a phenyl-containing indole molecule to the C-ring of naltrexone's morphinan base successfully produced a drug with the high receptor affinity of naltrexone, but which binds almost exclusively to the delta opioid receptor.[3]
