Embutramide
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | intravenous |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
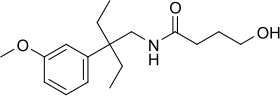
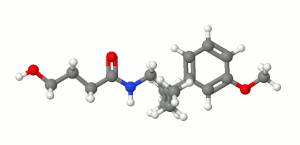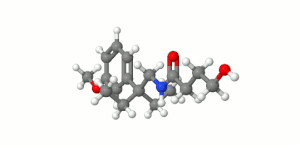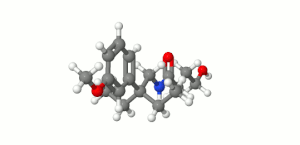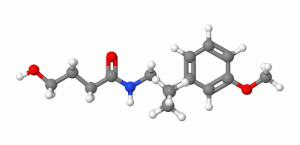
Embutramide (
Embutramide is formulated as a
Embutramide is used for euthanasia of a range of different animals, mainly small animals kept as pets rather than large farm animals. It may cause significant pain to the animal being euthanized,
Chemistry
Embutramide is considered an analog of gamma-hydroxybutyrate (GHB) due to its structural similarity to this naturally occurring neurotransmitter. GHB is known for its medical applications, such as treating narcolepsy and alcohol withdrawal symptoms. However, its recreational use has led to its classification as a controlled substance in many countries. The analog status of embutramide is significant in terms of its regulation and controlled use to prevent any potential misuse or abuse.
Synthesis

Alkylation of (3-Methoxyphenyl)acetonitrile [19924-43-7] (1) with
References
- ISBN 978-1-4757-2085-3.
- ISBN 978-0-7514-0499-9.
- ISBN 978-0-323-24198-4.
- ^ US 3045043, Schmitt K, Henning I, Lindner E, Ott H, "New gamma-hydroxy-carboxylic acid amides and process for their manufacture", issued 17 July 1962, assigned to Hoechst AG
- ^ "TRIBUTAME Euthanasia Solution" (PDF). Freedom of Information Summary NADA 141-245. U.S. Food and Drug Administration. Archived from the original (PDF) on 4 March 2017.
- PMID 2371711.
- ^ "DEA lists embutramide as schedule III controlled substance". Archived from the original on 8 March 2012.
- PMID 2815545.
- PMID 12423012.
- from the original on 29 November 2021. Retrieved 7 August 2020.
- ^ Schmitt Karl, et al. U.S. patent 3,045,043 (1962 to Hoechst Ag).
- ^ Karl Schmitt, et al. GB1002436 (1965 to Hoechst AG).
- ^ "science24.com - Synthesis of embutramide in mild conditions". science24.com. Archived from the original on 19 May 2023. Retrieved 19 May 2023.
