6-Methylapigenin
| |
| Names | |
|---|---|
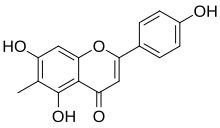
| IUPAC name
5,7-Dihydroxy-2-(4-hydroxyphenyl)-6-methylchromen-4-one
| |
| Identifiers | |
3D model (
JSmol ) |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C16H12O5 | |
| Molar mass | 284.267 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
6-Methylapigenin is a naturally occurring flavonoid and a derivative of apigenin. It has activity at GABAA receptors as a positive modulator.
Natural occurrence
6-Methylapigenin can be found in multiple plants, such as
Biological activity
6-Methylapigenin binds to the GABAA receptor on the benzodiazepine binding site. This compound possesses anxiolytic effects. In a mouse model, it is also able to potentiate sleep induced by hesperidin, another flavonoid.[2][3] However, since it does not have the chemical structure of benzodiazepines, it can therefore be classed as a nonbenzodiazepine.
References
- ^ PubChem. "6-Methylapigenin". pubchem.ncbi.nlm.nih.gov. Retrieved 2024-02-04.
- PMID 15840404.
- S2CID 37559366.
