Safranal
| |
| Names | |
|---|---|
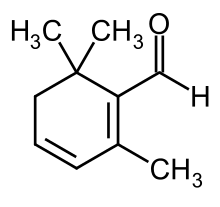
| Preferred IUPAC name
2,6,6-Trimethylcyclohexa-1,3-diene-1-carbaldehyde | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.003.758 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C10H14O | |
| Molar mass | 150.21 g/mol |
| Density | 0.9734 g/cm3 |
| Boiling point | 70 °C (158 °F; 343 K) at 1 mmHg |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Safranal is an
stigmas of crocus
flowers (Crocus sativus). It is the constituent primarily responsible for the aroma of saffron.
It is believed that safranal is a degradation product of the carotenoid zeaxanthin via the intermediate picrocrocin.
Pharmacology
Safranal is an effective anticonvulsant in animal models, shown to act as an agonist at GABAA receptors.[1][2] Safranal also exhibits high antioxidant and free radical scavenging activity,[3][4] along with cytotoxicity towards cancer cells in vitro.[5] One of its anticancer mechanisms of action involves disruption of the normal assembly dynamics of cellular microtubules.[6] It has also been shown to have antidepressant properties in animals and pilot studies in humans.[7][8]
Natural sources
Natural sources of safranal include:[9][unreliable source?]
- Microcystis (cyanobacterium)
- Aspalathus linearis(Rooibos)
- Camellia sinensis (Tea leaf)
- Crocus sativus (Saffron)
- Ficus carica(Fig leaf)
- Lycium chinense (Wolfberry)
- Cuminum cyminum (Cumin Seed)[10]
- Centaurea sibthorpii [11]
- Centaurea amanicola[11]
- Centaurea consanguinea[11]
- Erodium cicutarium (common stork's-bill or pinweed)[11]
- Calycopteris floribunda (Ukshi)[11]
- Sambucus nigra (elderberry)[11]
- Citrus limon (lemon)[11]
- Achillea distans [11]
