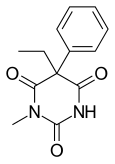
Methylphenobarbital
 | |
| Clinical data | |
|---|---|
| Trade names | Mebaral, generics |
| AHFS/Drugs.com | International Drug Names |
| MedlinePlus | a605022 |
| Routes of administration | By mouth (tablets) |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 70–76% |
| Metabolism | Liver |
| Elimination half-life | 34 hours |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Methylphenobarbital (
Approval history
- 1935 – Mebaral was introduced by Winthrop Pharmaceuticals.
- 2001 – Methylphenobarbital discontinued in the UK.
- 2003 – Mebaral was acquired by Ovation Pharmaceuticals (a specialty pharmaceutical company that acquired under-promoted branded pharmaceutical products).
- 2009 – Ovation was acquired by Lundbeck, which now markets Mebaral.
- 2012 – Lundbeck announced that they were abandoning the product in the US as of January 6, 2012. The stated reason was because "the company thoroughly evaluated all avenues for keeping Mebaral available to patients, but ultimately concluded that no matter what steps they [i.e. Lundbeck] took, patients would be forced to transition to a new therapy."
The company further stated in a letter on its website
Side effects
The use of antiepileptic drugs including methylphenobarbital during pregnancy may be associated with birth defects.[4]
Overdose
Symptoms of overdose of mephobarbital include confusion, decrease in or loss of reflexes, somnolence, fever, irritability, hypothermia, poor judgment, shortness of breath or slow/troubled breathing, slow heartbeat, slurred speech, staggering, trouble in sleeping, unusual movements of the eyes, weakness.
See also
References
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- ISBN 0-632-06046-8.
- ^ "Mebaral® (mephobarbital tablets, USP): Additional Product Discontinuation Information" (PDF). Lundbeck Inc. Archived from the original (PDF) on 10 August 2014.
- PMID 19218807.
