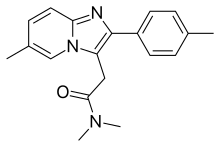
Zolpidem
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Ambien and Ambien CR, others[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a693025 |
| License data | |
| Pregnancy category |
|
| Dependence liability | Physical: High
sedative-hypnotic |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 70% (by mouth) |
| Protein binding | 92%[9] |
| Metabolism | Liver through CYP3A4 (~60%), CYP2C9 (~20%), and CYP1A2 (~14%)[10] |
| Metabolites | (ZCA) zolpidem 6-carboxylic acid; (ZPCA) zolpidem phenyl-4-carboxylic acid |
| Onset of action | ≤ 30 Minutes |
| Elimination half-life | 2.0 - 3 hours[4][9] |
| Duration of action | 3 hours |
| Excretion | Kidney (56%) fecal (34%) |
| Identifiers | |
| |
JSmol) | |
| Melting point | 193–197 °C (379–387 °F) [9] |
| |
| |
| | |
Zolpidem, sold under the brand name Ambien among others, is a medication primarily used for the short-term treatment of
Common side effects include daytime
Zolpidem is a
Zolpidem was approved for medical use in the United States in 1992.
Medical uses

Zolpidem is labeled for short-term (usually about two to six weeks) treatment of insomnia at the lowest possible dose.[4][11] It may be used for both improving sleep onset, sleep onset latency, and staying asleep.[7]
Guidelines from
Contraindications
Zolpidem should not be taken by people with
Use of zolpidem may impair driving skills with a resultant increased risk of
Zolpidem should not be prescribed to older people, who are more sensitive to the effects of hypnotics including zolpidem and are at an increased risk of falls and adverse cognitive effects, such as
Zolpidem has not been assigned to a
Adverse effects
The most common adverse effects of short-term use include headache (reported by 7% of people in clinical trials), drowsiness (2%), dizziness (1%), and diarrhea (1%); the most common side effects of long-term use included drowsiness (8%), dizziness (5%), allergy (4%), sinusitis (4%), back pain (3%), diarrhea (3%), drugged feeling (3%), dry mouth (3%), lethargy (3%), sore throat (3%), abdominal pain (2%), constipation (2%), heart palpitations (2%), lightheadedness (2%), rash (2%), abnormal dreams (1%), amnesia (1%), chest pain (1%), depression (1%), flu-like symptoms (1%), and sleep disorder (1%).[8]
Zolpidem increases risk of depression, falls and bone fracture, poor driving, suppressed respiration, and has been associated with an increased risk of death.[28] Upper and lower respiratory infections are also common (experienced by 1–10% of people).[11]
Residual 'hangover' effects, such as sleepiness and
Some users have reported unexplained
Tolerance, dependence and withdrawal

As zolpidem is associated with
Rodent studies of the tolerance-inducing properties have shown that zolpidem has less tolerance-producing potential than benzodiazepines, but in primates, the tolerance-producing potential of zolpidem was the same as seen with benzodiazepines.[38]
Overdose
Overdose can lead to coma or death.[11] When overdose occurs, there are often other drugs in the person's system.[11][15]
Zolpidem overdose can be treated with the GABAA receptor antagonist flumazenil, which displaces zolpidem from its binding site on the GABAA receptor to rapidly reverse the effects of the zolpidem.[11]
Detection in body fluids
Zolpidem may be quantitated in blood or plasma to confirm a diagnosis of poisoning in people who are hospitalized, to provide evidence in an impaired driving arrest, or to assist in a medicolegal death investigation. Blood or plasma zolpidem concentrations are usually in a range of 30–300 μg/L in persons receiving the drug therapeutically, 100–700 μg/L in those arrested for impaired driving, and 1000–7000 μg/L in victims of acute overdosage. Analytical techniques, in general, involve gas or liquid chromatography.[39][40][41]
Pharmacology
Mechanism of action

Zolpidem is a
Like
Pharmacokinetics
Microsome studies indicate zolpidem is metabolized by CYP3A4 (61%) CYP2C9 (22%), CYP1A2 (14%), CYP2D6 (<3%), and CYP2C19 (<3%).[10] Less than 1% is excreted in urine unchanged.[9] It is principally metabolized into three metabolites, none of which are believed to be pharmacologically active. The absolute bioavailability of zolpidem is about 70%. The drug reaches peak concentration in about 2 hours and has a half life in healthy adults of about 2–3 hours.[4][9] Zolpidem's half life is decreased in children and increased in the elderly and people with liver issues. While some studies show men metabolize zolpidem faster than women (possibly due to testosterone),[50] others do not.[9] A review found only a 33% lower clearance in women compared to men, suggesting the FDA's dosage reduction of 50% for women may have been too large.[51]
Interactions
People should not consume alcohol while taking zolpidem, and should not be prescribed opioid drugs nor take such illicit drugs recreationally.[52] Opioids can also increase the risk of becoming psychologically dependent on zolpidem.[medical citation needed] Use of opioids with zolpidem increases the risk of respiratory depression and death.[11] The US Food and Drug Administration (FDA) is advising that the opioid addiction medications buprenorphine and methadone should not be withheld from patients taking benzodiazepines or other drugs that depress the central nervous system (CNS).[53]
Next day sedation can be worsened if people take zolpidem while they are also taking antipsychotics, other sedatives, anxiolytics, antidepressant agents, antiepileptic drugs, and antihistamines. Some people taking antidepressants have had visual hallucinations when they also took zolpidem.[11]
Chemistry
Three chemical syntheses of zolpidem are common. 4-Methylacetophenone is used as a common precursor. This is brominated and reacted with 2-amino-5-methylpyridine to give the imidazopyridine. From here the reactions use a variety of reagents to complete the synthesis, either involving thionyl chloride or sodium cyanide. These reagents are challenging to handle and require thorough safety assessments.[56][57][58] Though such safety procedures are common in industry, they make clandestine manufacture difficult.
A number of major side-products of the sodium cyanide reaction have been characterised and include dimers and mannich products.[59]
Alpidem is also an imidazopyridine and is an analogue of zolpidem.[60][61][62] Both agents are GABAA receptor positive allosteric modulators.[60][61][62] However, whereas zolpidem is used as a hypnotic and sedative, alpidem was used as an anxiolytic.[60][61][62]
History
Zolpidem was used in Europe starting in 1988, and was brought to market there by
In 2015, the American Geriatrics Society said that zolpidem, eszopiclone and zaleplon met the Beers criteria and should be avoided in individuals 65 and over "because of their association with harms balanced with their minimal efficacy in treating insomnia."[25][26] The AGS stated the strength of the recommendation that older adults avoid zolpidem is "strong" and the quality of evidence supporting it is "moderate."[26]
Society and culture
Prescriptions in the US for all sleeping pills (including zolpidem) steadily declined from around 57 million tablets in 2013, to around 47 million in 2017, possibly in relation to concern about prescribing addictive drugs in the midst of the
Military use
The
Recreational use
Zolpidem has potential for medical misuse when the drug is continued long term without or against medical advice, or for recreational use when the drug is taken to achieve a "high".
Other drugs, including benzodiazepines and
Nonmedical use of zolpidem is increasingly common in the US, Canada, and the UK. Some users have reported decreased anxiety, mild
Regulation
For the stated reason of its potential for recreational use and dependence, zolpidem (along with the other benzodiazepine-like
Use in crime
The
Sleepwalking
Zolpidem received widespread media coverage in Australia after the death of a student who fell 20 metres (66 ft) from the Sydney Harbour Bridge while under the influence of zolpidem.[76]
Brand names
As of September 2018, zolpidem is marketed under many brands.[1]
Research
While cases of zolpidem improving
Animal studies in FDA files for zolpidem showed a dose dependent increase in some types of tumors, although the studies were too small to reach statistical significance.
References
- ^ a b "International brands for zolpidem". Drugs.com. Retrieved 15 March 2018.
- ^ a b "Zolpidem Use During Pregnancy". Drugs.com. 30 June 2020. Retrieved 14 August 2020.
- ISBN 9783319414003.
- ^ a b c d e f g h i j k l m "Zolpidem (Monograph)". The American Society of Health-System Pharmacists. 27 April 2023. Retrieved 10 March 2024.
- ^ "Scheduling of zolpidem (Stilnox)". Therapeutic Goods Administration (TGA). 21 February 2008. Retrieved 15 August 2020.
- ^ Anvisa (31 March 2023). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 4 April 2023). Archived from the original on 3 August 2023. Retrieved 3 August 2023.
- ^ PMID 28671376.
- ^ a b c "Ambien- zolpidem tartrate tablet, film coated". DailyMed. 29 August 2019. Retrieved 15 August 2020.
- ^ S2CID 23391285.
- ^ PMID 10383565.
- ^ a b c d e f g h i j k l m n o "Stilnoct 10mg Film-Coated Tablets - Summary of Product Characteristics (SmPC)". UK Electronic Medicines Compendium. 21 May 2018. Archived from the original on 20 August 2018. Retrieved 19 August 2018.
- ^ a b c "Guidance on the use of zaleplon, zolpidem and zopiclone for the short-term management of insomnia". NICE. 28 April 2004.
- ^ PMID 28875581.

- ^ PMID 27136449.

- ^ PMID 23404347.
- ^ a b "Drug Approval Package: Ambien (Zolpidem Tartrate) NDA 19908". U.S. Food and Drug Administration (FDA). 24 December 1999. Retrieved 15 August 2020.
- ^ a b "FDA Approves First Generic Versions of Ambien (Zolpidem Tartrate) for the Treatment of Insomnia". U.S. Food and Drug Administration (FDA) (Press release). Archived from the original on 6 March 2010. Retrieved 24 January 2010.
- ^ "Zolpidem". LiverTox. Archived from the original on 16 March 2018. Retrieved 15 March 2018.
- ^ "Some Sleep Drugs Can Impair Driving". U.S. Food and Drug Administration (FDA). 13 June 2013. Retrieved 15 March 2018.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Zolpidem - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- PMID 23248080.
- PMID 23348358. Retrieved 14 April 2013.
- ^ "FDA Drug Safety Communication: Risk of next-morning impairment after use of insomnia drugs; FDA requires lower recommended doses for certain drugs containing zolpidem (Ambien, Ambien CR, Edluar, and Zolpimist)". U.S. Food and Drug Administration (FDA). 10 January 2013. Archived from the original on 22 July 2017. Retrieved 14 April 2013.
- ^ S2CID 6621392.

- ^ S2CID 38797655.)
{{cite journal}}: CS1 maint: numeric names: authors list (link - ^ Drugsdb.eu. "Zolpidem Pregnancy Warnings". Archived from the original on 18 August 2018. Retrieved 1 February 2014.
- S2CID 7946506.
- S2CID 25592318.
- S2CID 4701575.
- ^ "Zolpidem and sleep-related behaviours" (PDF). NPS Position Statement. National Prescribing Service Limited. July 2008. Archived from the original (PDF) on 10 April 2012.
- ^ "Zolpidem ("Stilnox") – updated information – February 2008". Therapeutic Goods Administration (TGA). 21 February 2008. Archived from the original on 3 June 2009. Retrieved 22 June 2009.
- PMID 18853708.
- ^ PMID 23456542.
- ^ PMID 25613443.
- PMID 16096519.
- S2CID 30959471.
- S2CID 46510829.
- ^ S2CID 25511804.
- PMID 10517569.
- ^ Baselt R (2011). Disposition of Toxic Drugs and Chemicals in Man (9th ed.). Seal Beach, CA: Biomedical Publications. pp. 1836–1838.
- PMID 27346730.
- S2CID 86674799.
- S2CID 86156878.
- S2CID 25654504. Archived from the originalon 12 January 2002.
- PMID 8794909. Archived from the originalon 8 January 2009. Retrieved 7 October 2007.
- PMID 9880578.
- PMID 15037809.
WARNING: The reference indicates that zaleplon-Sonata, not zolpidem, increases Slow-wave sleep
- S2CID 10888200.
- PMID 12919178.
- S2CID 92998845.
- ^ "FDA Drug Safety Communication: FDA warns about serious risks and death when combining opioid pain or cough medicines with benzodiazepines; requires its strongest warning". U.S. Food and Drug Administration (FDA). 31 August 2016. Retrieved 18 August 2018.
- ^ "FDA urges caution about withholding opioid addiction medications". U.S. Food and Drug Administration. 20 September 2017. Retrieved 15 August 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- S2CID 209164976.
- S2CID 46250744.
- ISBN 9780471752158.
- ^ IN 246080, Rawalnath SR, Crasta Santosh R, Saxena A, "Process for the preparation of zolpidem", published 21 December 2005, issued 14 February 2011
- .
- .
- ^ PMID 1981304.
- ^ S2CID 11612995.
- ^ PMID 22981367.
- ^ a b Morris S (22 December 1992). "Searle Wins Ok To Sell Sleep Aid". Chicago Tribune.
- ^ Crow D (1 June 2018). "Ambien defence: the real side effects of sleeping pills". Financial Times. Archived from the original on 10 December 2022.
- ^ "Air Force Special Operations Command Instruction 48-101" (PDF). e-publishing.af.mil. 30 November 2012. Archived from the original (PDF) on 11 June 2014. Retrieved 8 March 2014.
- ^ PMID 26648651.
- PMID 16336040.
- PMID 12022074.
- ^ "Kennedy To Enter Drug Rehab After Car Crash; Congressman Wrecked Car Near Capitol". Archived from the original on 28 March 2020. Retrieved 23 June 2009.
- ^ Mulvihill K. "Ambien Abuse on Rise Among Teens". KSL. Archived from the original on 20 February 2009. Retrieved 22 June 2009.
- ^ "Swimming Australia's 'Stilnox six' given final warning as AOC decides not to issue any further sanctions". ABC News. www.abc.net.au. 23 August 2013. Retrieved 3 August 2016.
- ^ US 4382938, Kaplan J-P, George P, "Imidazo[1,2-a] pyridine derivatives and their application as pharmaceuticals", published 10 May 1983, issued 17 July 1984, assigned to Synthelabo
- ^ "Zolpidem most frequently used date rape drug in Korea". The Korea Herald. 29 February 2016.
- ^ a b Schrotenboer B (26 March 2014). "Darren Sharper case spotlights sleep drug's dark side". USA Today.
- ^ Red C (17 February 2014). "In the rape case against Darren Sharper, former LAPD detective says Ambien is used often and can be similar to GHB". Daily News. New York.
- ^ "Stilnox blamed for Harbour Bridge death". nineMSN News. 23 February 2007. Archived from the original on 15 June 2007.
- S2CID 38304960.
- PMID 20460949.
- S2CID 10756280.
- PMID 32979847.
- PMID 27303633.
- PMID 29973038.
- PMID 27667780.
