Neodymium(III) oxide
Appearance
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Neodymium(III) oxide
| |
| Other names
Neodymium oxide, Neodymium sesquioxide
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.013.832 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
SMILES
| |
| Properties | |
| Nd2O3 | |
| Molar mass | 336.48 g/mol |
| Appearance | light bluish gray hexagonal crystals
|
| Density | 7.24 g/cm3 |
| Melting point | 2,233 °C (4,051 °F; 2,506 K) |
| Boiling point | 3,760 °C (6,800 °F; 4,030 K)[1] |
| .0003 g/100 mL (75 °C) | |
| +10,200.0·10−6 cm3/mol | |
| Structure | |
| Hexagonal, hP5 | |
| P-3m1, No. 164 | |
| Thermochemistry | |
Heat capacity (C)
|
111.3 J·mol−1·K−1[1] |
Std molar
entropy (S⦵298) |
158.6 J·mol−1·K−1 |
Std enthalpy of (ΔfH⦵298)formation |
−1807.9 kJ·mol−1
|
| Related compounds | |
Other anions
|
Neodymium(II) chloride Neodymium(III) chloride |
Other cations
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Neodymium(III) oxide or neodymium sesquioxide is the
rare-earth mixture didymium, previously believed to be an element, partially consists of neodymium(III) oxide.[2]
Uses
Neodymium(III) oxide is used to dope
alexandrite appears blue in sunlight and red in artificial light.[5]
About 7000 catalyst.[4]
Reactions
Neodymium(III) oxide is formed when
neodymium(III) nitride or neodymium(III) hydroxide is roasted in air.[6]
Structure
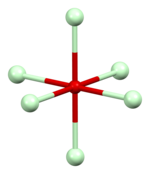
Neodymium(III) oxide has a low-temperature trigonal A
form in space group P3m1.[7] This structure type is favoured by the early lanthanides.[8][9] At higher temperatures it adopts two other forms, the hexagonal H form in space group P63/mmc and the cubic X form in Im3m. The high-temperature forms exhibit crystallographic disorder.[10][11]
| Packing | Neodymium coordination | Oxygen O1 coordination | Oxygen O2 coordination |
|---|---|---|---|

|

|
|

|
| A-M2O3 structure type | approximately capped octahedral | octahedral | approximately tetrahedral |
References
- ^ a b c
Lide, David R. (1998), Handbook of Chemistry and Physics (87 ed.), Boca Raton, FL: CRC Press, pp. 471, 552, ISBN 0-8493-0594-2
- ^
Brady, George Stuart; Clauser, Henry R.; Vaccari, John A. (2002), Materials Handbook (15 ed.), New York: McGraw-Hill Professional, p. 779, ISBN 978-0-07-136076-0, retrieved 2009-03-18
- ^
Eagleson, Mary (1994), Concise Encyclopedia of Chemistry, Springer, p. 680, ISBN 978-3-11-011451-5, retrieved 2009-03-18
- ^ a b
Emsley, John (2003), Nature's Building Blocks, Oxford University Press, pp. 268–9, ISBN 978-0-19-850340-8, retrieved 2009-03-18
- ^
Bray, Charles (2001), Dictionary of Glass (2 ed.), University of Pennsylvania Press, p. 103, ISBN 978-0-8122-3619-4, retrieved 2009-03-18
- ^ Spencer, James Frederick (1919), The Metals of the Rare Earths, London: Longmans, Green, and Co, p. 115, retrieved 2009-03-18
- ^ D. Taylor (1984). "Thermal Expansion Data: III Sesquioxides, U2N3, with the corundum and the A-, B- and C-M2O3 structures". Trans. J. Br. Ceram. Soc. 83: 92–98.
- ISBN 978-0-08-037941-8.
- ^ A. F. Wells (1984). Structural Inorganic Chemistry (5th ed.). Oxford University Press. pp. 544–547.
- .
- .
