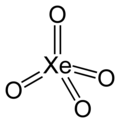
Xenon tetroxide
| |||
| Names | |||
|---|---|---|---|
| IUPAC names
Xenon tetraoxide
Xenon(VIII) oxide | |||
| Other names
Xenon tetroxide
Perxenic anhydride | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChemSpider | |||
CompTox Dashboard (EPA)
|
|||
| |||
SMILES
| |||
| Properties | |||
| XeO4 | |||
| Molar mass | 195.29 g mol−1 | ||
| Appearance | Yellow solid below −36 °C | ||
| Melting point | −35.9 °C (−32.6 °F; 237.2 K) | ||
| Boiling point | 0 °C (32 °F; 273 K)[1] | ||
| reacts | |||
| Structure | |||
| Tetrahedral[2] | |||
| 0 D | |||
| Thermochemistry | |||
Std enthalpy of (ΔfH⦵298)formation |
+153.5 kcal mol−1 [3] | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
powerful explosive | ||
| Related compounds | |||
Related compounds
|
Perxenic acid
Xenon trioxide | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Xenon tetroxide is a
All eight valence electrons of xenon are involved in the bonds with the oxygen, and the oxidation state of the xenon atom is +8. Oxygen is the only element that can bring xenon up to its highest oxidation state; even fluorine can only give XeF6 (+6).
Two other short-lived xenon compounds with an oxidation state of +8, XeO3F2 and XeO2F4, are accessible by the reaction of xenon tetroxide with xenon hexafluoride. XeO3F2 and XeO2F4 can be detected with mass spectrometry. The perxenates are also compounds where xenon has the +8 oxidation state.
Reactions
At temperatures above −35.9 °C, xenon tetroxide is very prone to explosion, decomposing into xenon and oxygen gases with ΔH = −643 kJ/mol:
- XeO4 → Xe + 2 O2
Xenon tetroxide dissolves in water to form perxenic acid and in alkalis to form perxenate salts:
- XeO4 + 2 H2O → H4XeO6
- XeO4 + 4 NaOH → Na4XeO6 + 2 H2O
Xenon tetroxide can also react with xenon hexafluoride to give xenon oxyfluorides:
- XeO4 + XeF6 → XeOF4 + XeO3F2
- XeO4 + 2XeF6 → XeO2F4 + 2 XeOF4
Synthesis
All syntheses start from the perxenates, which are accessible from the xenates through two methods. One is the disproportionation of xenates to perxenates and xenon:
- 2 HXeO−
4 + 2 OH− → XeO4−
6 + Xe + O2 + 2 H2O
The other is oxidation of the xenates with ozone in basic solution:
- HXeO−
4 + O3 + 3 OH− → XeO4−
6 + O2 + 2 H2O
Barium perxenate is reacted with sulfuric acid and the unstable perxenic acid is dehydrated to give xenon tetroxide:[6]
- Ba
2XeO
6 + 2 H
2SO
4 → 2 BaSO
4 + H
4XeO
6 - H
4XeO
6 → 2 H
2O + XeO
4
Any excess perxenic acid slowly undergoes a decomposition reaction to xenic acid and oxygen:
- 2 H
4XeO
6 → O
2 + 2 H
2XeO
4 + 2 H
2O
References
- ^
Lide, David R. (1998). Handbook of Chemistry and Physics (87 ed.). Boca Raton, Florida: CRC Press. p. 494. ISBN 0-8493-0594-2.
- .
- .
- ^
H.Selig, J. G. Malm, H. H. Claassen, C. L. Chernick, J. L. Huston (1964). "Xenon tetroxide – Preparation & Some Properties". Science. 143 (3612): 1322–3. S2CID 29205117.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^
J. L. Huston; M. H. Studier; E. N. Sloth (1964). "Xenon tetroxide — Mass Spectrum". Science. 143 (3611): 1162–3. S2CID 28547895.
- ISBN 9780080501093.
- Lide, D. R., ed. (2002). CRC Handbook of Chemistry and Physics (83rd ed.). Boca Raton, Florida: CRC Press. ISBN 0-8493-0483-0.