Gallium(III) oxide
 β-Ga2O3 crystal
| |
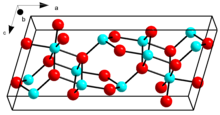 Crystal structure of β-Ga2O3
| |
| Names | |
|---|---|
| Other names
gallium trioxide, gallium sesquioxide
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.031.525 |
| EC Number |
|
PubChem CID
|
|
RTECS number
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Ga2O3 | |
| Molar mass | 187.444 g/mol |
| Appearance | white crystalline powder |
| Melting point | 1,725 °C (3,137 °F; 1,998 K)[1] |
| insoluble | |
| Solubility | soluble in most acids |
| Structure[2][3] | |
Monoclinic, mS20, space group = C2/m, No. 12
| |
a = 1.2232 nm, b = 0.3041 nm, c = 0.5801 nm α = 90°, β = 103.73°, γ = 90° β-phase
| |
Formula units (Z)
|
4 |
| Thermochemistry[4] | |
Heat capacity (C)
|
92.1 J/(mol·K) |
Std molar
entropy (S⦵298) |
85.0 J/(mol·K) |
Std enthalpy of (ΔfH⦵298)formation |
−1089.1 kJ/mol |
Gibbs free energy (ΔfG⦵)
|
−998.3 kJ/mol |
Enthalpy of fusion (ΔfH⦵fus)
|
100 kJ/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Gallium(III) oxide is an
Preparation
Gallium trioxide is precipitated in hydrated form upon neutralization of acidic or basic solution of gallium salt. Also, it is formed on heating gallium in air or by thermally decomposing gallium nitrate at 200–250 °C.
Crystalline Ga2O3 can occur in five polymorphs, α, β, γ, δ, and ε. Of these polymorphs β-Ga2O3 is the most thermodynamically stable phase at standard temperature and pressure[14] while α-Ga2O3 is the most stable polymorph under high pressures.[15]
- β-Ga2O3 HVPE, and MOVPE.[17] HVPE is preferred for vertical power semiconductor devices due to its fast growth rate.[18] β-Ga2O3 epitaxial films grown by MOVPE exhibit higher electron mobilities and lower background carrier concentrations than those grown by other thin-film growth techniques.[19][20]
Bulk substrates of β-Ga2O3 can be produced, which is one of the major advantages of this material system. Bulk substrates can be produced in multiple orientations and by multiple techniques.[21][22]

- α-Ga2O3 can be obtained by heating β-Ga2O3 at 65 kbar and 1100 °C. It has a Epitaxial thin films of α-Ga2O3 deposited on c-plane (0001), m-plane (1010), or a-plane (1120) sapphiresubstrates have been demonstrated.
- γ-Ga2O3 is prepared by rapidly heating the hydroxide gel at 400–500 °C. A more crystalline form of this polymorph can be prepared directly from gallium metal by a solvothermal synthesis.[23]
- δ-Ga2O3 is obtained by heating Ga(NO3)3 at 250 °C.[24]
- ε-Ga2O3 is prepared by heating δ-Ga2O3 at 550 °C.[14] Thin films of ε-Ga2O3 are deposited by means of metalorganic vapour-phase epitaxy using trimethylgallium and water on sapphire substrates at temperatures between 550 and 650 °C[25]
Reactions
Gallium(III) trioxide is
4.
With HCl, it forms gallium trichloride GaCl3.[28]
- Ga2O3 + 6 HCl → 2 GaCl3 + 3 H2O
It can be reduced to
- Ga2O3 + 2 H2 → Ga2O + 2 H2O
- Ga2O3 + 4 Ga → 3 Ga2O
Structure
β-Ga2O3, with a melting point of 1900 °C, is the most stable crystalline modification. The oxide ions are in a distorted cubic closest packing arrangement, and the gallium (III) ions occupy distorted tetrahedral and octahedral sites, with Ga–O bond distances of 1.83 and 2.00 Å respectively.[31]
α-Ga2O3 has the same structure (corundum) as α-Al2O3, wherein Ga ions are 6-coordinate.[32][33]
γ-Ga2O3 has a defect spinel structure similar to that of γ-Al2O3.[34]
ε-Ga2O3 films deposited by metalorganic vapour-phase epitaxy show a columnar structure with orthorhombic crystal symmetry. Macroscopically, this structure is seen by X-ray crystallography as hexagonal close packed.[35]
κ-Ga2O3 has an orthorhombic structure and forms with 120° twin domains, resulting in hexagonal symmetry which is often identified as ε-Ga2O3.[36]
| Phase of Ga2O3 | Figure | Crystal structure name |
|---|---|---|
| α |  |
Rhombohedral
(Corundum) |
| β |  |
Monoclinic |
| γ |  |
Cubic defect spinel |
| δ | 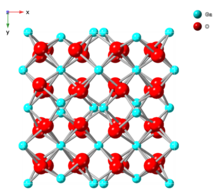 |
Body-centered cubic bixbyite |
| ε | 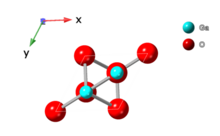 |
Hexagonal |
| κ (subgroup of ε phase)[41] |  |
Orthorhombic |
Applications
Gallium(III) oxide has been studied for usage as passive components in lasers,[43] phosphors,[5] and luminescent materials[44] as well as active components for gas sensors,[6] power diodes,[45] and power transistors.[46][47] Since the first publication in January 2012 by the National Institute of Information and Communications Technology, in collaboration with Tamura Co., Ltd. and Koha Co., Ltd. of the world's first single-crystal gallium oxide (Ga2O3) field-effect transistors, the predominant interest in gallium oxide is in the β-polymorph for power electronics.[48][7]
Monoclinic β-Ga2O3 has shown increasing performance since 2012 approaching state of the art GaN and SiC power devices.[7] β-Ga2O3 Schottky diodes have exceeded breakdown voltages of 2400 V.[45] β-Ga2O3/NiOx p–n diodes have exhibited breakdown voltages over 1200 V.[49] β-Ga2O3 MOSFETs have individually achieved figures of merits of fT of 27 GHz,[46] fMAX of 48 GHz,[47] and 5.4 MV/cm average breakdown field.[47] This field exceeds that which is possible in SiC or GaN.
ε-Ga2O3 thin films deposited on sapphire show potential applications as solar-blind UV photodetector.[8]
References
- ISBN 0-07-049439-8.
- .
- .
- ISBN 1-4398-5511-0.
- ^ S2CID 254561209.
- ^ .
- ^ S2CID 246660179.
- ^ .
- PMID 37733937.
- S2CID 127789383.
- .
- S2CID 230578016.
- ^ Pearton, Stephen J.; Ren, Fan; Law, Mark; Mastro, Michael (2021). Fundamental Studies and Modeling of Radiation Effects in beta-Gallium Oxide. Defense Threat Reduction Agency, U.S.
- ^ a b Bailar, J; Emeléus, H; Nyholm, R; Trotman-Dickenson, A. F. (1973). Comprehensive Inorganic Chemistry. Vol. 1, p. 1091.
- S2CID 250882992.)
{{cite journal}}: CS1 maint: numeric names: authors list (link - S2CID 221403770.
- S2CID 246660179.
- S2CID 245773856.
- S2CID 38628999.
- S2CID 104453229.
- S2CID 246074647.
- ^ "Substrates". Novel Crystal Technology, Inc.
- PMID 23307528.
- .
- .
- ISBN 0538497521
- ISBN 075140103X
- ISBN 9780470145395
- ^ Koch, H. F.; Girard, L. A.; Roundhill, D. M. (1999). "Determination of Gallium in a Cerium Surrogate and in Drops from a Copper Collector by ICP as Model Studies for the Removal of Gallium from Plutonium". Atomic Spectroscopy. 20 (1): 30.
- ^ Greenwood, N.N.; Emeleus, H. J. and Sharpe, A. G. (1963) "The chemistry of Gallium" in Advances in Inorganic Chemistry and Radiochemistry, Vol. 5, Elsevier, Academic Press
- ISBN 978-0-470-86078-6.
- .
- ^ .
- ISBN 978-0-08-037941-8.
- hdl:10831/67366.
- .
- hdl:10023/6874.
- .
- .
- PMID 23307528.
- S2CID 247259950.
- S2CID 102474875.
- S2CID 216243413.
- S2CID 23058882.
- ^ S2CID 209696027.
- ^ S2CID 195439906.
- ^ arXiv:2211.01088 [cond-mat.mtrl-sci].
- .
- S2CID 232372680.
