Uranium dioxide
| |
| Names | |
|---|---|
| IUPAC names
Uranium dioxide
Uranium(IV) oxide | |
| Other names
Urania
Uranous oxide | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.014.273 |
| EC Number |
|
PubChem CID
|
|
RTECS number
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| UO2 | |
| Molar mass | 270.03 g/mol |
| Appearance | black powder |
| Density | 10.97 g/cm3 |
| Melting point | 2,865 °C (5,189 °F; 3,138 K) |
| insoluble | |
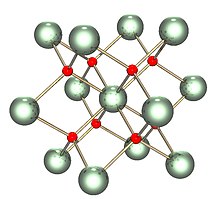
| Structure | |
| Fluorite (cubic), cF12 | |
| Fm3m, No. 225 | |
a = 547.1 pm [1]
| |
| Tetrahedral (O2−); cubic (UIV) | |
| Thermochemistry | |
Std molar
entropy (S⦵298) |
78 J·mol−1·K−1[2] |
Std enthalpy of (ΔfH⦵298)formation |
−1084 kJ·mol−1[2] |
| Hazards | |
| GHS labelling: | |
  
| |
| Danger | |
| H300, H330, H373, H410 | |
| P260, P264, P270, P271, P273, P284, P301+P310, P304+P340, P310, P314, P320, P321, P330, P391, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | N/A |
| Safety data sheet (SDS) | ICSC 1251 |
| Related compounds | |
Other anions
|
Uranium(IV) selenide
|
Other cations
|
Protactinium(IV) oxide Neptunium(IV) oxide |
| Triuranium octoxide Uranium trioxide | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Uranium dioxide or uranium(IV) oxide (UO2), also known as urania or uranous oxide, is an
Production
Uranium dioxide is produced by reducing uranium trioxide with hydrogen.
- UO3 + H2 → UO2 + H2O at 700 °C (973 K)
This reaction plays an important part in the creation of
Chemistry
Structure
The solid is isostructural with (has the same structure as) fluorite (calcium fluoride), where each U is surrounded by eight O nearest neighbors in a cubic arrangement. In addition, the dioxides of cerium, thorium, and the transuranic elements from neptunium through californium have the same structures.[3] No other elemental dioxides have the fluorite structure. Upon melting, the measured average U-O coordination reduces from 8 in the crystalline solid (UO8 cubes), down to 6.7±0.5 (at 3270 K) in the melt.[4] Models consistent with these measurements show the melt to consist mainly of UO6 and UO7 polyhedral units, where roughly 2⁄3 of the connections between polyhedra are corner sharing and 1⁄3 are edge sharing.[4]
-
Uranium dioxide
-
Sintered uranium dioxide pellet
Oxidation
Uranium dioxide is
- 3 UO2 + O2 → U3O8 at 700 °C (970 K)
The electrochemistry of uranium dioxide has been investigated in detail as the galvanic corrosion of uranium dioxide controls the rate at which used nuclear fuel dissolves. See spent nuclear fuel for further details. Water increases the oxidation rate of plutonium and uranium metals.[5][6]
Carbonization
Uranium dioxide is carbonized in contact with carbon, forming uranium carbide and carbon monoxide.
- .
This process must be done under an inert gas as uranium carbide is easily oxidized back into uranium oxide.
Uses
Nuclear fuel
UO2 is used mainly as
The
-
Uranium oxide fuel pellet
-
Starting material containers for uranium dioxide fuel pellet production at a plant in Russia
Color for glass ceramic glaze
Uranium oxide (urania) was used to color glass and ceramics prior to World War II, and until the applications of radioactivity were discovered this was its main use. In 1958 the military in both the US and Europe allowed its commercial use again as depleted uranium, and its use began again on a more limited scale. Urania-based ceramic glazes are dark green or black when fired in a reduction or when UO2 is used; more commonly it is used in oxidation to produce bright yellow, orange and red glazes.
Other uses
Prior to the realisation of the harmfulness of radiation, uranium was included in false teeth and dentures, as its slight fluorescence made the dentures appear more like real teeth in a variety of lighting conditions.[citation needed]
Depleted uranium dioxide can be also used as a
The use of uranium dioxide as a material for
Uranium dioxide displays strong
Semiconductor properties
The band gap of uranium dioxide is comparable to those of silicon and gallium arsenide, near the optimum for efficiency vs band gap curve for absorption of solar radiation, suggesting its possible use for very efficient solar cells based on Schottky diode structure; it also absorbs at five different wavelengths, including infrared, further enhancing its efficiency. Its intrinsic conductivity at room temperature is about the same as of single crystal silicon.[11]
The
The
The
The stoichiometry of the material dramatically influences its electrical properties. For example, the electrical conductivity of UO1.994 is orders of magnitude lower at higher temperatures than the conductivity of UO2.001[citation needed].
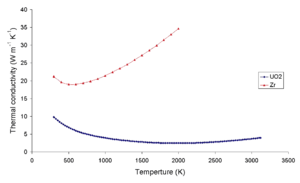
Uranium dioxide, like U3O8, is a ceramic material capable of withstanding high temperatures (about 2300 °C, in comparison with at most 200 °C for silicon or GaAs), making it suitable for high-temperature applications like thermophotovoltaic devices.
Uranium dioxide is also resistant to radiation damage, making it useful for rad-hard devices for special military and aerospace applications.
A
Toxicity
Uranium dioxide is known to be absorbed by phagocytosis in the lungs.[13]
See also
References
- S2CID 97183844.
- ^ ISBN 978-0-618-94690-7.
- S2CID 118365366.
- ^ S2CID 206561628.
- )
- .
- ^ Örtel, Stefan. Uran in der Keramik. Geschichte - Technik - Hersteller.
- S2CID 4299921.
- PMID 28740123.
- S2CID 231812027.
- PMID 21668262.
- .
- ISBN 0-8493-7302-6[page needed]
Further reading
- Barrett, S. A.; Jacobson, A. J.; Tofield, B. C.; Fender, B. E. F. (1982). "The preparation and structure of barium uranium oxide BaUO3+x". Acta Crystallographica Section B. 38 (11): 2775. .
External links
- Semiconducting properties of uranium oxides Archived 2012-09-01 at the Wayback Machine
- Free Dictionary Listing for Uranium Dioxide
- The Uranium dioxide Archived 2013-09-16 at the Wayback Machine International Bio-Analytical Industries, Inc.






