Sponge
| Sponges | |
|---|---|

| |
| A stove-pipe sponge | |
| Scientific classification | |
| Domain: | Eukaryota |
| Kingdom: | Animalia |
| Phylum: | Porifera Grant , 1836
|
| Classes | |
| |
| Synonyms | |
Sponges (also known as sea sponges), the members of the
Sponges have unspecialized cells that can
Etymology
The term sponge derives from the Ancient Greek word σπόγγος (spóngos 'sponge').[9]
Overview
Sponges are similar to other animals in that they are
Although most of the approximately 5,000–10,000 known species of sponges feed on bacteria and other microscopic food in the water, some host photosynthesizing microorganisms as endosymbionts, and these alliances often produce more food and oxygen than they consume. A few species of sponges that live in food-poor environments have evolved as carnivores that prey mainly on small crustaceans.[12]
Sponges reproduce both asexually and sexually. Most species that use
In most sponges, an internal gelatinous matrix called mesohyl functions as an
The
The few species of demosponge that have entirely soft fibrous skeletons with no hard elements have been used by humans over thousands of years for several purposes, including as padding and as cleaning tools. By the 1950s, though, these had been overfished so heavily that the industry almost collapsed, and most sponge-like materials are now synthetic. Sponges and their microscopic endosymbionts are now being researched as possible sources of medicines for treating a wide range of diseases. Dolphins have been observed using sponges as tools while foraging.[16]
Distinguishing features
Sponges constitute the
Even if a few sponges are able to produce mucus – which acts as a microbial barrier in all other animals – no sponge with the ability to secrete a functional mucus layer has been recorded. Without such a mucus layer their living tissue is covered by a layer of microbial symbionts, which can contribute up to 40–50% of the sponge wet mass. This inability to prevent microbes from penetrating their porous tissue could be a major reason why they have never evolved a more complex anatomy.[19]
Like cnidarians (jellyfish, etc.) and ctenophores (comb jellies), and unlike all other known metazoans, sponges' bodies consist of a non-living jelly-like mass (mesohyl) sandwiched between two main layers of cells.[20][21] Cnidarians and ctenophores have simple nervous systems, and their cell layers are bound by internal connections and by being mounted on a basement membrane (thin fibrous mat, also known as "basal lamina").[21] Sponges do not have a nervous system similar to that of vertebrates but may have one that is quite different.[8] Their middle jelly-like layers have large and varied populations of cells, and some types of cells in their outer layers may move into the middle layer and change their functions.[18]
| Sponges[18][20] | ctenophores[21]
| |
|---|---|---|
| Nervous system | No/Yes | Yes, simple |
| Cells in each layer bound together | No, except that Homoscleromorpha have basement membranes.[22] |
Yes: inter-cell connections; basement membranes |
| Number of cells in middle "jelly" layer | Many | Few |
| Cells in outer layers can move inwards and change functions | Yes | No |
Basic structure
Cell types
A sponge's body is hollow and is held in shape by the
Other types of cells live and move within the mesohyl:[18][20]
- Lophocytes are amoeba-like cells that move slowly through the mesohyl and secrete collagen fibres.
- Collencytes are another type of collagen-producing cell.
- Rhabdiferous cells secrete polysaccharides that also form part of the mesohyl.
- Oocytes and spermatocytes are reproductive cells.
- Sclerocytes secrete the mineralized spicules ("little spines") that form the skeletons of many sponges and in some species provide some defense against predators.
- In addition to or instead of sclerocytes, demosponges have spongocytes that secrete a form of collagen that polymerizes into spongin, a thick fibrous material that stiffens the mesohyl.
- Myocytes("muscle cells") conduct signals and cause parts of the animal to contract.
- "Grey cells" act as sponges' equivalent of an immune system.
- totipotent, in other words, each is capable of transformation into any other type of cell. They also have important roles in feeding and in clearing debris that block the ostia.
Many larval sponges possess neuron-less eyes that are based on cryptochromes. They mediate phototaxic behavior.[25]
Glass sponges' syncytia
Some types of cells have a single nucleus and membrane each but are connected to other single-nucleus cells and to the main syncytium by "bridges" made of cytoplasm. The sclerocytes that build spicules have multiple nuclei, and in glass sponge larvae they are connected to other tissues by cytoplasm bridges; such connections between sclerocytes have not so far been found in adults, but this may simply reflect the difficulty of investigating such small-scale features. The bridges are controlled by "plugged junctions" that apparently permit some substances to pass while blocking others.[26]
Water flow and body structures
Most sponges work rather like chimneys: they take in water at the bottom and eject it from the osculum ("little mouth") at the top. Since ambient currents are faster at the top, the suction effect that they produce by Bernoulli's principle does some of the work for free. Sponges can control the water flow by various combinations of wholly or partially closing the osculum and ostia (the intake pores) and varying the beat of the flagella, and may shut it down if there is a lot of sand or silt in the water.[18]
Although the layers of
The simplest body structure in sponges is a tube or vase shape known as "asconoid", but this severely limits the size of the animal. The body structure is characterized by a stalk-like spongocoel surrounded by a single layer of choanocytes. If it is simply scaled up, the ratio of its volume to surface area increases, because surface increases as the square of length or width while volume increases proportionally to the cube. The amount of tissue that needs food and oxygen is determined by the volume, but the pumping capacity that supplies food and oxygen depends on the area covered by choanocytes. Asconoid sponges seldom exceed 1 mm (0.039 in) in diameter.[18]
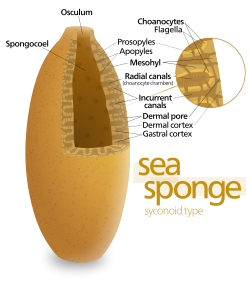
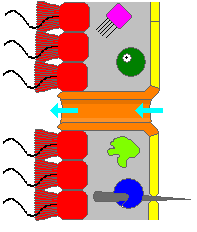
Some sponges overcome this limitation by adopting the "syconoid" structure, in which the body wall is pleated. The inner pockets of the pleats are lined with choanocytes, which connect to the outer pockets of the pleats by ostia. This increase in the number of choanocytes and hence in pumping capacity enables syconoid sponges to grow up to a few centimeters in diameter.
The "leuconoid" pattern boosts pumping capacity further by filling the interior almost completely with mesohyl that contains a network of chambers lined with choanocytes and connected to each other and to the water intakes and outlet by tubes. Leuconid sponges grow to over 1 m (3.3 ft) in diameter, and the fact that growth in any direction increases the number of choanocyte chambers enables them to take a wider range of forms, for example, "encrusting" sponges whose shapes follow those of the surfaces to which they attach. All freshwater and most shallow-water marine sponges have leuconid bodies. The networks of water passages in
Skeleton
In zoology a
Some sponges also secrete
Vital functions

Movement
Although adult sponges are fundamentally
Respiration, feeding and excretion
Sponges do not have distinct
It used to be claimed that
Sponges' cells absorb oxygen by diffusion from water into cells as water flows through body, into which carbon dioxide and other soluble waste products such as ammonia also diffuse. Archeocytes remove mineral particles that threaten to block the ostia, transport them through the mesohyl and generally dump them into the outgoing water current, although some species incorporate them into their skeletons.[18]
Carnivorous sponges

In waters where the supply of food particles is very poor, some species prey on
Most known carnivorous sponges have completely lost the water flow system and choanocytes. However, the genus Chondrocladia uses a highly modified water flow system to inflate balloon-like structures that are used for capturing prey.[35][38]
Endosymbionts
Freshwater sponges often host
A recently discovered carnivorous sponge that lives near
"Immune" system
Sponges do not have the complex
Reproduction
Asexual

Sponges have three
: 90–94Gemmules are "survival pods" which a few marine sponges and many freshwater species produce by the thousands when dying and which some, mainly freshwater species, regularly produce in autumn.
Sexual
Most sponges are
A few species release fertilized eggs into the water, but most retain the eggs until they hatch. By retaining the eggs, the parents can transfer symbiotic microorganisms directly to their offspring through
Meiosis
The cytological progression of porifera oogenesis and spermatogenesis (gametogenesis) is very similar to that of other metazoa.[45] Most of the genes from the classic set of meiotic genes, including genes for DNA recombination and double-strand break repair, that are conserved in eukaryotes are expressed in the sponges (e.g. Geodia hentscheli and Geodia phlegraei).[45] Since porifera are considered to be the earliest divergent animals, these findings indicate that the basic toolkit of meiosis including capabilities for recombination and DNA repair were present early in eukaryote evolution.[45]
Life cycle
Sponges in
Coordination of activities
Adult sponges lack
Sponges contain genes very similar to those that contain the "recipe" for the post-synaptic density, an important signal-receiving structure in the neurons of all other animals. However, in sponges these genes are only activated in "flask cells" that appear only in larvae and may provide some sensory capability while the larvae are swimming. This raises questions about whether flask cells represent the predecessors of true neurons or are evidence that sponges' ancestors had true neurons but lost them as they adapted to a sessile lifestyle.[47]
Ecology
Habitats
Sponges are worldwide in their distribution, living in a wide range of ocean habitats, from the polar regions to the tropics.[30] Most live in quiet, clear waters, because sediment stirred up by waves or currents would block their pores, making it difficult for them to feed and breathe.[31] The greatest numbers of sponges are usually found on firm surfaces such as rocks, but some sponges can attach themselves to soft sediment by means of a root-like base.[48]
Sponges are more abundant but less diverse in temperate waters than in tropical waters, possibly because organisms that prey on sponges are more abundant in tropical waters.
The different classes of sponge live in different ranges of habitat:
Class Water type[20] Depth[20] Type of surface[20] CalcareaMarine less than 100 m (330 ft) Hard Glass spongesMarine Deep Soft or firm sediment Demosponges Marine, brackish; and about 150 freshwater species[18] Inter-tidal to abyssal;[20] a carnivorous demosponge has been found at 8,840 m (5.49 mi)[37] Any
As primary producers
Sponges with photosynthesizing endosymbionts produce up to three times more oxygen than they consume, as well as more organic matter than they consume. Such contributions to their habitats' resources are significant along Australia's Great Barrier Reef but relatively minor in the Caribbean.[30]
Defenses

Many sponges shed
A few species, the Caribbean fire sponge Tedania ignis, cause a severe rash in humans who handle them.[18] Turtles and some fish feed mainly on sponges. It is often said that sponges produce chemical defenses against such predators.[18] However, experiments have been unable to establish a relationship between the toxicity of chemicals produced by sponges and how they taste to fish, which would diminish the usefulness of chemical defenses as deterrents. Predation by fish may even help to spread sponges by detaching fragments.[20] However, some studies have shown fish showing a preference for non chemically defended sponges,[51] and another study found that high levels of coral predation did predict the presence of chemically defended species.[52]
Predation
Spongeflies, also known as spongillaflies (Neuroptera, Sisyridae), are specialist predators of freshwater sponges. The female lays her eggs on vegetation overhanging water. The larvae hatch and drop into the water where they seek out sponges to feed on. They use their elongated mouthparts to pierce the sponge and suck the fluids within. The larvae of some species cling to the surface of the sponge while others take refuge in the sponge's internal cavities. The fully grown larvae leave the water and spin a cocoon in which to pupate.[53]
Bioerosion
The Caribbean chicken-liver sponge
Diseases
Caribbean sponges of the genus
Collaboration with other organisms
In addition to hosting photosynthesizing endosymbionts,[18] sponges are noted for their wide range of collaborations with other organisms. The relatively large encrusting sponge Lissodendoryx colombiensis is most common on rocky surfaces, but has extended its range into seagrass meadows by letting itself be surrounded or overgrown by seagrass sponges, which are distasteful to the local starfish and therefore protect Lissodendoryx against them; in return, the seagrass sponges get higher positions away from the sea-floor sediment.[56]

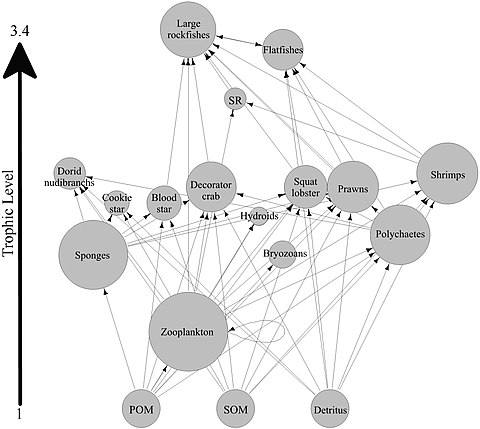
Sponge loop
Most sponges are
The hypothesis has been made that coral reef sponges facilitate the transfer of coral-derived organic matter to their associated detritivores via the production of sponge detritus, as shown in the diagram. Several sponge species are able to convert coral-derived DOM into sponge detritus,[63][64] and transfer organic matter produced by corals further up the reef food web. Corals release organic matter as both dissolved and particulate mucus,[65][66]</ref>[67][68] as well as cellular material such as expelled Symbiodinium.[69][70][62]
Organic matter could be transferred from corals to sponges by all these pathways, but DOM likely makes up the largest fraction, as the majority (56 to 80%) of coral mucus dissolves in the water column,[66] and coral loss of fixed carbon due to expulsion of Symbiodinium is typically negligible (0.01%)[69] compared with mucus release (up to ~40%).[71][72] Coral-derived organic matter could also be indirectly transferred to sponges via bacteria, which can also consume coral mucus.[73][74][75][62]
Sponge holobiont
Besides a one to one
Some of these relationships include endosymbionts within bacteriocyte cells, and cyanobacteria or microalgae found below the pinacoderm cell layer where they are able to receive the highest amount of light, used for phototrophy. They can host over 50 different microbial phyla and candidate phyla, including Alphaprotoebacteria,
Systematics and evolutionary history
Taxonomy
The phylum Porifera is further divided into classes mainly according to the composition of their skeletons:[17][30]
- Calcarea have skeletons made of calcite, a form of calcium carbonate, which may form separate spicules or large masses. All the cells have a single nucleus and membrane.[30]
- Most
In the 1970s, sponges with massive calcium carbonate skeletons were assigned to a separate class,
So far scientific publications have identified about 9,000 poriferan species,[30] of which: about 400 are glass sponges; about 500 are calcareous species; and the rest are demosponges.[18] However, some types of habitat, vertical rock and cave walls and galleries in rock and coral boulders, have been investigated very little, even in shallow seas.[30]
Classes
Sponges were traditionally distributed in three classes: calcareous sponges (Calcarea), glass sponges (Hexactinellida) and demosponges (Demospongiae). However, studies have shown that the
Sponges are divided into classes mainly according to the composition of their skeletons:[20] These are arranged in evolutionary order as shown below in ascending order of their evolution from top to bottom:
Class Type of cells[20] Spicules[20] Spongin fibers[20] Massive exoskeleton[30] Body form[20] HexactinellidaMostly syncytiain all speciesSilica
May be individual or fusedNever Never Leuconoid DemospongiaeSingle nucleus, single external membrane Silica In many species In some species.
Made of aragonite if present.[17][30]Leuconoid CalcareaSingle nucleus, single external membrane Calcite
May be individual or large massesNever Common.
Made of calcite if present.Asconoid, syconoid, leuconoid or solenoid[88] HomoscleromorphaSingle nucleus, single external membrane Silica In many species Never Sylleibid or leuconoid
Fossil record

Although
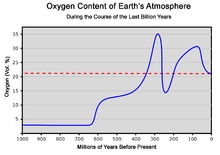
Freshwater sponges appear to be much younger, as the earliest known fossils date from the Mid-
A chemical tracer is 24-isopropylcholestane, which is a stable derivative of 24-isopropylcholesterol, which is said to be produced by demosponges but not by eumetazoans ("true animals", i.e. cnidarians and bilaterians). Since choanoflagellates are thought to be animals' closest single-celled relatives, a team of scientists examined the biochemistry and genes of one choanoflagellate species. They concluded that this species could not produce 24-isopropylcholesterol but that investigation of a wider range of choanoflagellates would be necessary in order to prove that the fossil 24-isopropylcholestane could only have been produced by demosponges.[97] Although a previous publication reported traces of the chemical 24-isopropylcholestane in ancient rocks dating to 1,800 million years ago,[98] recent research using a much more accurately dated rock series has revealed that these biomarkers only appear before the end of the Marinoan glaciation approximately 635 million years ago,[99] and that "Biomarker analysis has yet to reveal any convincing evidence for ancient sponges pre-dating the first globally extensive Neoproterozoic glacial episode (the Sturtian, ~713 million years ago in Oman)". While it has been argued that this 'sponge biomarker' could have originated from marine algae, recent research suggests that the algae's ability to produce this biomarker evolved only in the Carboniferous; as such, the biomarker remains strongly supportive of the presence of demosponges in the Cryogenian.[100][101][102]
It is difficult to fit
Relationships to other animal groups

| Simplified family tree showing calcareous sponges as closest to more complex animals[106]
| |||||||||||||||||||||||||||||||||||||||
|
| Simplified family tree showing Homoscleromorpha as closest to more complex animals[107]
| ||||||||||||||||||
|
In the 1990s, sponges were widely regarded as a
Analyses since 2001 have concluded that
Other anatomical and biochemical evidence links the Eumetazoa with
- Homoscleromorphaare most closely related to Eumetazoa;
- calcareous sponges are the next closest;
- the other demosponges are evolutionary "aunts" of these groups; and
- the chancelloriids, bag-like animals whose fossils are found in Cambrian rocks, may be sponges.[107]
The sperm of Homoscleromorpha share features with the sperm of Eumetazoa, that sperm of other sponges lack. In both Homoscleromorpha and Eumetazoa layers of cells are bound together by attachment to a carpet-like basal membrane composed mainly of "typ IV" collagen, a form of collagen not found in other sponges – although the spongin fibers that reinforce the mesohyl of all demosponges is similar to "type IV" collagen.[22]

The analyses described above concluded that sponges are closest to the ancestors of all Metazoa, of all multi-celled animals including both sponges and more complex groups. However, another comparison in 2008 of 150 genes in each of 21 genera, ranging from fungi to humans but including only two species of sponge, suggested that comb jellies (ctenophora) are the most basal lineage of the Metazoa included in the sample.[109][110][111][112] If this is correct, either modern comb jellies developed their complex structures independently of other Metazoa, or sponges' ancestors were more complex and all known sponges are drastically simplified forms. The study recommended further analyses using a wider range of sponges and other simple Metazoa such as Placozoa.[109]
However, reanalysis of the data showed that the computer algorithms used for analysis were misled by the presence of specific ctenophore genes that were markedly different from those of other species, leaving sponges as either the sister group to all other animals, or an ancestral paraphyletic grade.
A very large and internally consistent alignment of 1,719 proteins at the metazoan scale, published in 2017, showed that (i) sponges – represented by Homoscleromorpha, Calcarea, Hexactinellida, and Demospongiae – are monophyletic, (ii) sponges are sister-group to all other multicellular animals, (iii) ctenophores emerge as the second-earliest branching animal lineage, and (iv)
In March 2021, scientists from Dublin found additional evidence that sponges are the sister group to all other animals,[117] while in May 2023, Schultz et al. found patterns of irreversible change in genome synteny that provide strong evidence that ctenophores are the sister group to all other animals instead.[118]
Notable spongiologists
- Céline Allewaert
- Patricia Bergquist
- James Scott Bowerbank
- Maurice Burton
- Henry John Carter
- Max Walker de Laubenfels
- Arthur Dendy
- Édouard Placide Duchassaing de Fontbressin
- Randolph Kirkpatrick
- Robert J. Lendlmayer von Lendenfeld
- Edward Alfred Minchin
- Giovanni Domenico Nardo
- Eduard Oscar Schmidt
- Émile Topsent
Use
By dolphins
A report in 1997 described use of sponges
By humans

Skeleton
The
Many objects with sponge-like textures are now made of substances not derived from poriferans. Synthetic sponges include personal and household
The luffa "sponge", also spelled loofah, which is commonly sold for use in the kitchen or the shower, is not derived from an animal but mainly from the fibrous "skeleton" of the sponge gourd (Luffa aegyptiaca, Cucurbitaceae).[123]
Antibiotic compounds
Sponges have
Other biologically active compounds

Lacking any protective shell or means of escape, sponges have evolved to synthesize a variety of unusual compounds. One such class is the oxidized fatty acid derivatives called oxylipins. Members of this family have been found to have anti-cancer, anti-bacterial and anti-fungal properties. One example isolated from the Okinawan plakortis sponges, plakoridine A, has shown potential as a cytotoxin to murine lymphoma cells.[126][127]
See also
- Lists of sponges
- Sponge Reef Project
- 3-Alkylpyridinium, compounds found in marine Haplosclerida sponges
References
- PMID 30323207.
- .
- ^ PMID 29199080.
- PMID 26621703.
- ^ (PDF) from the original on 2020-04-25. Retrieved 2018-11-04.
- ISSN 1463-6409.
- bioRxiv 10.1101/200972.
- ^ PMID 36619868.
- ^ "Henry George Liddell, Robert Scott, A Greek-English Lexicon, Σ ς, , σπλαχρός: , σπόγγος". www.perseus.tufts.edu. Archived from the original on 2021-09-05. Retrieved 2021-09-05.
- ^ a b Hooper J (2018). "Structure of Sponges". Queensland Museum. Archived from the original on 26 September 2019. Retrieved 27 September 2019.
- PMID 25276334.
- S2CID 24484610.
- ^ ISBN 978-0-520-03658-1.
- ISBN 978-1-947172-52-4.
- PMID 9860990.
- ^ PMID 15947077.
- ^ ISBN 978-0-306-47260-2.
- ^ ISBN 978-0-03-025982-1.
- PMID 30002868.
- ^ ISBN 978-0-19-551368-4.
- ^ ISBN 978-0-19-551368-4.
- ^ PMID 12382326.
- ISBN 978-0-03-025982-1.
- PMID 34735222.
- PMID 22442365.
- ^ PMID 21680406.
- ISBN 978-0-03-025982-1.
- ISBN 978-0-07-290961-6.
- ^ "Marine Species Identification Portal: Halisarca dujardini". species-identification.org. Archived from the original on 2020-10-17. Retrieved 2019-08-02.
- ^ ISBN 978-0-470-01617-6.
- ^ a b c Krautter M (1998). "Ecology of siliceous sponges: Application to the environmental interpretation of the Upper Jurassic sponge facies (Oxfordian) from Spain" (PDF). Cuadernos de Geología Ibérica. 24: 223–239. Archived from the original (PDF) on March 19, 2009. Retrieved 2008-10-10.
- S2CID 86297053.
- PMID 22558119.
- ^ "4 new species of 'killer' sponges discovered off Pacific coast". CBC News. April 19, 2014. Archived from the original on April 19, 2014. Retrieved 2014-09-04.
- ^ (PDF) from the original on 2008-09-06. Retrieved 2008-10-31.
- S2CID 86588792.
- ^ S2CID 4320216.
- .
- .
- Matt Walker (10 November 2008). "Nature's 'fibre optics' experts". BBC News. Archived from the original on 17 December 2008. Retrieved 11 November 2008.
- ^ ISBN 978-0-471-35837-4.
- .
- PMID 35524305.
- PMID 25785444.
- PMID 21672727.
- ^ PMID 32929503.
- PMID 15579547.
- PMID 17551586.
- PMID 17175169.
- S2CID 1495896. Archived from the original(PDF) on 2008-10-06.
- ISBN 978-0-521-33665-9.
- S2CID 84799900.
- PMID 24567392.
- ISBN 978-0-313-33922-6.
- ^ a b Gochfeld DJ, Easson CG, Slattery M, Thacker RW, Olson JB (2012). Steller D, Lobel L (eds.). "Population Dynamics of a Sponge Disease on Caribbean Reefs". Diving for Science 2012. Proceedings of the American Academy of Underwater Sciences 31st Symposium. Archived from the original on 2015-09-04. Retrieved 2013-11-17.
{{cite journal}}: CS1 maint: unfit URL (link) - PMID 16956064. Lay summary in: Clarke M (2006-10-17). "New disease threatens sponges". Practical Fishkeeping. Archived from the originalon 2007-09-26.
- .
- .
- ISBN 978-0-87850-138-0.
- (PDF) from the original on 2018-07-19. Retrieved 2022-01-24.
- PMID 33384908.
- ISSN 2296-7745..
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine - ^ doi:10.3354/meps12443.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine - ^ Rix L, de Goeij JM, Mueller CE, Struck U and others (2016) "Coral mucus fuels the sponge loop in warm- and coldwater coral reef ecosystems". Sci Rep, 6: 18715.
- ^ .
- .
- ^ PMID 14999280.
- .
- .
- ^ .
- PMID 11147708.
- .
- PMID 22442377.
- .
- .
- .
- PMID 24092742.
- PMID 29523192..
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 2017-10-16 at the Wayback Machine - PMID 26136729.
- ^ PMID 27103626.
- ^ "Spongia Linnaeus, 1759". World Register of Marine Species. Archived from the original on 2016-03-27. Retrieved 2012-07-18.
- ^ JSTOR 1307076.
- S2CID 34175521. Archived from the original(PDF) on May 9, 2009. Retrieved 2012-08-22.
- PMID 25902535.
- ^ Hartman WD, Goreau TF (1970). "Jamaican coralline sponges: Their morphology, ecology and fossil relatives". Symposium of the Zoological Society of London. 25: 205–243. (cited by MGG.rsmas.miami.edu). Archived 2018-08-18 at the Wayback Machine
- ISBN 978-0-19-857181-0.
- PMID 21179486.
- (PDF) from the original on 2019-08-01. Retrieved 2019-08-01.
- S2CID 21745242.
- S2CID 41195363.
- ^ Reitner J, Wörheide G (2002). "Non-lithistid fossil Demospongiae – origins of their palaeobiodiversity and highlights in history of preservation". In Hooper JN, Van Soest RW (eds.). Systema Porifera: A Guide to the Classification of Sponges (PDF). New York, NY: Kluwer Academic Plenum. Archived (PDF) from the original on 2008-12-16. Retrieved November 4, 2008.
- ^ .
- ^ McMenamin MA (2008). "Early Cambrian sponge spicules from the Cerro Clemente and Cerro Rajón, Sonora, México". Geologica Acta. 6 (4): 363–367.
- ^ S2CID 38837724.
- PMID 34321662.
- ^ "Demospongia". University of California Museum of Paleontology. Berkeley, CA: U.C. Berkeley. Archived from the original on October 18, 2013. Retrieved 2008-11-27.
- from the original on 2024-04-10. Retrieved 2015-06-18.
- PMID 18632573.
- PMID 21676777.
- S2CID 4314662. Archived from the original(PDF) on 2018-07-24. Retrieved 2019-08-01.
- .
- S2CID 90887224.
- PMID 26903629.
- ^ .
- S2CID 133427906.
- S2CID 129127213. free text at Janussen D (2002). "(full text without images)". Journal of Paleontology. Archived from the originalon December 10, 2008. Retrieved 2008-08-04.
- ^ PMID 29280585.
- ^ S2CID 34175521. Archived from the original(PDF) on May 9, 2009. Retrieved 2008-11-04.
- PMID 11504944.
- ^ S2CID 4397099.
- PMID 19759036.
- PMID 24337300.
- PMID 24847885.
- PMID 26621703.
- ^ Berwald J (2017). Spineless: the science of jellyfish and the art of growing a backbone. Riverhead Books.[page needed]
- PMID 19175291.
- PMID 33310849.
- PMID 33741994.
- PMID 37198475.
- hdl:2027.42/71936.
- ISBN 978-1-84407-527-0.
- ISBN 978-0-8135-2715-4.
- ^ "Sponges". Cervical Barrier Advancement Society. 2004. Archived from the original on January 14, 2009. Retrieved 2006-09-17.
- S2CID 27313678.
- ISBN 978-3-540-00968-9.
- S2CID 1593418.
- .
- .
Further reading
- Hickman C, Roberts L, Larson A (2003). Animal Diversity (3rd ed.). New York: McGraw-Hill. ISBN 978-0-07-234903-0.
- Ereskovsky AV (2010). The Comparative Embryology of Sponges. Russia: Springer Science+Business Media. ISBN 978-90-481-8575-7.
- Wörheide G (April 2008). "A hypercalcified sponge with soft relatives: Vaceletia is a keratose demosponge". Molecular Phylogenetics and Evolution. 47 (1): 433–8. PMID 18321733.
External links
- Water flow and feeding in the phylum Porifera (sponges) – Flash animations of sponge body structures, water flow and feeding
- Carsten's Spongepage, Information on the ecology and the biotechnological potential of sponges and their associated bacteria.
- History of Tarpon Springs sponge industry, Tarpon Springs, Florida
- Nature's 'fibre optics' experts
- The Sponge Reef Project
- Queensland Museum information about sponges
- Queensland Museum Sessile marine invertebrates collections
- Queensland Museum Sessile marine invertebrates research
- Sponge Guide for Britain and Ireland Archived 2008-12-08 at the Wayback Machine, Bernard Picton, Christine Morrow & Rob van Soest
- World Porifera database, the world list of extant sponges, includes a searchable database.
- Sponges: World production and markets // Food and Agriculture Organisation