DNA: Difference between revisions
Extended confirmed users 1,756 edits |
Extended confirmed users 79,276 edits Expanding bare references using ReferenceExpander |
||
| Line 28: | Line 28: | ||
Therefore, any DNA strand normally has one end at which there is a phosphate group attached to the 5′ carbon of a ribose (the 5′ phosphoryl) and another end at which there is a free hydroxyl group attached to the 3′ carbon of a ribose (the 3′ hydroxyl). The orientation of the 3′ and 5′ carbons along the sugar-phosphate backbone confers [[directionality (molecular biology)|directionality]] (sometimes called polarity) to each DNA strand. In a [[nucleic acid double helix]], the direction of the nucleotides in one strand is opposite to their direction in the other strand: the strands are [[Antiparallel (biochemistry)|antiparallel]]. The asymmetric ends of DNA strands are said to have a directionality of five prime end (5′ ), and three prime end (3′), with the 5′ end having a terminal phosphate group and the 3′ end a terminal hydroxyl group. One major difference between DNA and [[RNA]] is the sugar, with the 2-deoxyribose in DNA being replaced by the related pentose sugar [[ribose]] in RNA.<ref name="berg" /> |
Therefore, any DNA strand normally has one end at which there is a phosphate group attached to the 5′ carbon of a ribose (the 5′ phosphoryl) and another end at which there is a free hydroxyl group attached to the 3′ carbon of a ribose (the 3′ hydroxyl). The orientation of the 3′ and 5′ carbons along the sugar-phosphate backbone confers [[directionality (molecular biology)|directionality]] (sometimes called polarity) to each DNA strand. In a [[nucleic acid double helix]], the direction of the nucleotides in one strand is opposite to their direction in the other strand: the strands are [[Antiparallel (biochemistry)|antiparallel]]. The asymmetric ends of DNA strands are said to have a directionality of five prime end (5′ ), and three prime end (3′), with the 5′ end having a terminal phosphate group and the 3′ end a terminal hydroxyl group. One major difference between DNA and [[RNA]] is the sugar, with the 2-deoxyribose in DNA being replaced by the related pentose sugar [[ribose]] in RNA.<ref name="berg" /> |
||
[[File:DNA animation.gif|thumb|upright|A section of DNA. The bases lie horizontally between the two spiraling strands<ref> |
[[File:DNA animation.gif|thumb|upright|A section of DNA. The bases lie horizontally between the two spiraling strands<ref>{{Cite web|last=Bank|first=RCSB Protein Data|title=RCSB PDB - 1D65: MOLECULAR STRUCTURE OF THE B-DNA DODECAMER D(CGCAAATTTGCG)2; AN EXAMINATION OF PROPELLER TWIST AND MINOR-GROOVE WATER STRUCTURE AT 2.2 ANGSTROMS RESOLUTION|url=https://www.rcsb.org/structure/1D65|access-date=2023-03-27|website=www.rcsb.org|language=en-US}}</ref> ([[:File:DNA orbit animated.gif|animated version]]).]] |
||
The DNA double helix is stabilized primarily by two forces: [[hydrogen bond]]s between nucleotides and [[Stacking (chemistry)|base-stacking]] interactions among [[aromatic]] nucleobases.<ref name="Yakovchuk2006">{{cite journal | vauthors = Yakovchuk P, Protozanova E, Frank-Kamenetskii MD | title = Base-stacking and base-pairing contributions into thermal stability of the DNA double helix | journal = Nucleic Acids Research | volume = 34 | issue = 2 | pages = 564–74 | year = 2006 | pmid = 16449200 | pmc = 1360284 | doi = 10.1093/nar/gkj454 |issn=0305-1048 }}</ref> The four bases found in DNA are [[adenine]] ({{mono|A}}), [[cytosine]] ({{mono|C}}), [[guanine]] ({{mono|G}}) and [[thymine]] ({{mono|T}}). These four bases are attached to the sugar-phosphate to form the complete nucleotide, as shown for [[adenosine monophosphate]]. Adenine pairs with thymine and guanine pairs with cytosine, forming {{mono|A-T}} and {{mono|G-C}} [[base pair]]s.<ref>{{cite book | first = Burton E. | last = Tropp | name-list-style = vanc | title = Molecular Biology | edition = 4th | year = 2012 | publisher = Jones and Barlett Learning | location = Sudbury, Mass. | isbn = 978-0-7637-8663-2 }}</ref><ref>{{cite web | url = https://www.mun.ca/biology/scarr/Watson-Crick_Model.html | title = Watson-Crick Structure of DNA | year = 1953 | first = Steven | last = Carr | name-list-style = vanc | publisher = Memorial University of Newfoundland | access-date=13 July 2016 | url-status=live | archive-url = https://web.archive.org/web/20160719095721/http://www.mun.ca/biology/scarr/Watson-Crick_Model.html | archive-date = 19 July 2016 | df = dmy-all }}</ref> |
The DNA double helix is stabilized primarily by two forces: [[hydrogen bond]]s between nucleotides and [[Stacking (chemistry)|base-stacking]] interactions among [[aromatic]] nucleobases.<ref name="Yakovchuk2006">{{cite journal | vauthors = Yakovchuk P, Protozanova E, Frank-Kamenetskii MD | title = Base-stacking and base-pairing contributions into thermal stability of the DNA double helix | journal = Nucleic Acids Research | volume = 34 | issue = 2 | pages = 564–74 | year = 2006 | pmid = 16449200 | pmc = 1360284 | doi = 10.1093/nar/gkj454 |issn=0305-1048 }}</ref> The four bases found in DNA are [[adenine]] ({{mono|A}}), [[cytosine]] ({{mono|C}}), [[guanine]] ({{mono|G}}) and [[thymine]] ({{mono|T}}). These four bases are attached to the sugar-phosphate to form the complete nucleotide, as shown for [[adenosine monophosphate]]. Adenine pairs with thymine and guanine pairs with cytosine, forming {{mono|A-T}} and {{mono|G-C}} [[base pair]]s.<ref>{{cite book | first = Burton E. | last = Tropp | name-list-style = vanc | title = Molecular Biology | edition = 4th | year = 2012 | publisher = Jones and Barlett Learning | location = Sudbury, Mass. | isbn = 978-0-7637-8663-2 }}</ref><ref>{{cite web | url = https://www.mun.ca/biology/scarr/Watson-Crick_Model.html | title = Watson-Crick Structure of DNA | year = 1953 | first = Steven | last = Carr | name-list-style = vanc | publisher = Memorial University of Newfoundland | access-date=13 July 2016 | url-status=live | archive-url = https://web.archive.org/web/20160719095721/http://www.mun.ca/biology/scarr/Watson-Crick_Model.html | archive-date = 19 July 2016 | df = dmy-all }}</ref> |
||
| Line 104: | Line 104: | ||
{{further|Sense (molecular biology)}} |
{{further|Sense (molecular biology)}} |
||
A [[DNA sequencing|DNA sequence]] is called a "sense" sequence if it is the same as that of a [[messenger RNA]] copy that is translated into protein.<ref> |
A [[DNA sequencing|DNA sequence]] is called a "sense" sequence if it is the same as that of a [[messenger RNA]] copy that is translated into protein.<ref>{{Cite web|title=Chemistry - Queen Mary University of London|url=https://www.qmul.ac.uk/undergraduate/coursefinder/courses/2023/chemistry/|access-date=2023-03-27|website=www.qmul.ac.uk}}</ref> The sequence on the opposite strand is called the "antisense" sequence. Both sense and antisense sequences can exist on different parts of the same strand of DNA (i.e. both strands can contain both sense and antisense sequences). In both prokaryotes and eukaryotes, antisense RNA sequences are produced, but the functions of these RNAs are not entirely clear.<ref>{{cite journal | vauthors = Hüttenhofer A, Schattner P, Polacek N | title = Non-coding RNAs: hope or hype? | journal = Trends in Genetics | volume = 21 | issue = 5 | pages = 289–97 | date = May 2005 | pmid = 15851066 | doi = 10.1016/j.tig.2005.03.007 }}</ref> One proposal is that antisense RNAs are involved in regulating [[gene expression]] through RNA-RNA base pairing.<ref>{{cite journal | vauthors = Munroe SH | title = Diversity of antisense regulation in eukaryotes: multiple mechanisms, emerging patterns | journal = Journal of Cellular Biochemistry | volume = 93 | issue = 4 | pages = 664–71 | date = November 2004 | pmid = 15389973 | doi = 10.1002/jcb.20252 | s2cid = 23748148 }}</ref> |
||
A few DNA sequences in prokaryotes and eukaryotes, and more in [[plasmid]]s and [[virus]]es, blur the distinction between sense and antisense strands by having [[overlapping gene]]s.<ref>{{cite journal | vauthors = Makalowska I, Lin CF, Makalowski W | title = Overlapping genes in vertebrate genomes | journal = Computational Biology and Chemistry | volume = 29 | issue = 1 | pages = 1–12 | date = February 2005 | pmid = 15680581 | doi = 10.1016/j.compbiolchem.2004.12.006 }}</ref> In these cases, some DNA sequences do double duty, encoding one protein when read along one strand, and a second protein when read in the opposite direction along the other strand. In [[bacteria]], this overlap may be involved in the regulation of gene transcription,<ref>{{cite journal | vauthors = Johnson ZI, Chisholm SW | title = Properties of overlapping genes are conserved across microbial genomes | journal = Genome Research | volume = 14 | issue = 11 | pages = 2268–72 | date = November 2004 | pmid = 15520290 | pmc = 525685 | doi = 10.1101/gr.2433104 }}</ref> while in viruses, overlapping genes increase the amount of information that can be encoded within the small viral genome.<ref>{{cite journal | vauthors = Lamb RA, Horvath CM | title = Diversity of coding strategies in influenza viruses | journal = Trends in Genetics | volume = 7 | issue = 8 | pages = 261–66 | date = August 1991 | pmid = 1771674 | doi = 10.1016/0168-9525(91)90326-L | pmc = 7173306 }}</ref> |
A few DNA sequences in prokaryotes and eukaryotes, and more in [[plasmid]]s and [[virus]]es, blur the distinction between sense and antisense strands by having [[overlapping gene]]s.<ref>{{cite journal | vauthors = Makalowska I, Lin CF, Makalowski W | title = Overlapping genes in vertebrate genomes | journal = Computational Biology and Chemistry | volume = 29 | issue = 1 | pages = 1–12 | date = February 2005 | pmid = 15680581 | doi = 10.1016/j.compbiolchem.2004.12.006 }}</ref> In these cases, some DNA sequences do double duty, encoding one protein when read along one strand, and a second protein when read in the opposite direction along the other strand. In [[bacteria]], this overlap may be involved in the regulation of gene transcription,<ref>{{cite journal | vauthors = Johnson ZI, Chisholm SW | title = Properties of overlapping genes are conserved across microbial genomes | journal = Genome Research | volume = 14 | issue = 11 | pages = 2268–72 | date = November 2004 | pmid = 15520290 | pmc = 525685 | doi = 10.1101/gr.2433104 }}</ref> while in viruses, overlapping genes increase the amount of information that can be encoded within the small viral genome.<ref>{{cite journal | vauthors = Lamb RA, Horvath CM | title = Diversity of coding strategies in influenza viruses | journal = Trends in Genetics | volume = 7 | issue = 8 | pages = 261–66 | date = August 1991 | pmid = 1771674 | doi = 10.1016/0168-9525(91)90326-L | pmc = 7173306 }}</ref> |
||
| Line 132: | Line 132: | ||
=== Quadruplex structures === |
=== Quadruplex structures === |
||
{{further|G-quadruplex}} |
{{further|G-quadruplex}} |
||
[[File:Parallel telomere quadruple.png|thumb|right|DNA quadruplex formed by [[telomere]] repeats. The looped conformation of the DNA backbone is very different from the typical DNA helix. The green spheres in the center represent potassium ions.<ref> |
[[File:Parallel telomere quadruple.png|thumb|right|DNA quadruplex formed by [[telomere]] repeats. The looped conformation of the DNA backbone is very different from the typical DNA helix. The green spheres in the center represent potassium ions.<ref>{{Cite web|title=Structure Summary for 1KF1|url=http://ndbserver.rutgers.edu/service/ndb/atlas/summary?searchTarget=UD0017|access-date=2023-03-27|website=ndbserver.rutgers.edu}}</ref>]] |
||
At the ends of the linear chromosomes are specialized regions of DNA called [[telomere]]s. The main function of these regions is to allow the cell to replicate chromosome ends using the enzyme [[telomerase]], as the enzymes that normally replicate DNA cannot copy the extreme 3′ ends of chromosomes.<ref name=Greider>{{cite journal | vauthors = Greider CW, Blackburn EH | title = Identification of a specific telomere terminal transferase activity in Tetrahymena extracts | journal = Cell | volume = 43 | issue = 2 Pt 1 | pages = 405–13 | date = December 1985 | pmid = 3907856 | doi = 10.1016/0092-8674(85)90170-9 | doi-access = free }}</ref> These specialized chromosome caps also help protect the DNA ends, and stop the [[DNA repair]] systems in the cell from treating them as damage to be corrected.<ref name=Nugent>{{cite journal | vauthors = Nugent CI, Lundblad V | title = The telomerase reverse transcriptase: components and regulation | journal = Genes & Development | volume = 12 | issue = 8 | pages = 1073–85 | date = April 1998 | pmid = 9553037 | doi = 10.1101/gad.12.8.1073 | doi-access = free }}</ref> In [[List of distinct cell types in the adult human body|human cells]], telomeres are usually lengths of single-stranded DNA containing several thousand repeats of a simple TTAGGG sequence.<ref>{{cite journal | vauthors = Wright WE, Tesmer VM, Huffman KE, Levene SD, Shay JW | title = Normal human chromosomes have long G-rich telomeric overhangs at one end | journal = Genes & Development | volume = 11 | issue = 21 | pages = 2801–09 | date = November 1997 | pmid = 9353250 | pmc = 316649 | doi = 10.1101/gad.11.21.2801 }}</ref> |
At the ends of the linear chromosomes are specialized regions of DNA called [[telomere]]s. The main function of these regions is to allow the cell to replicate chromosome ends using the enzyme [[telomerase]], as the enzymes that normally replicate DNA cannot copy the extreme 3′ ends of chromosomes.<ref name=Greider>{{cite journal | vauthors = Greider CW, Blackburn EH | title = Identification of a specific telomere terminal transferase activity in Tetrahymena extracts | journal = Cell | volume = 43 | issue = 2 Pt 1 | pages = 405–13 | date = December 1985 | pmid = 3907856 | doi = 10.1016/0092-8674(85)90170-9 | doi-access = free }}</ref> These specialized chromosome caps also help protect the DNA ends, and stop the [[DNA repair]] systems in the cell from treating them as damage to be corrected.<ref name=Nugent>{{cite journal | vauthors = Nugent CI, Lundblad V | title = The telomerase reverse transcriptase: components and regulation | journal = Genes & Development | volume = 12 | issue = 8 | pages = 1073–85 | date = April 1998 | pmid = 9553037 | doi = 10.1101/gad.12.8.1073 | doi-access = free }}</ref> In [[List of distinct cell types in the adult human body|human cells]], telomeres are usually lengths of single-stranded DNA containing several thousand repeats of a simple TTAGGG sequence.<ref>{{cite journal | vauthors = Wright WE, Tesmer VM, Huffman KE, Levene SD, Shay JW | title = Normal human chromosomes have long G-rich telomeric overhangs at one end | journal = Genes & Development | volume = 11 | issue = 21 | pages = 2801–09 | date = November 1997 | pmid = 9353250 | pmc = 316649 | doi = 10.1101/gad.11.21.2801 }}</ref> |
||
| Line 205: | Line 205: | ||
In many [[species]], only a small fraction of the total sequence of the [[genome]] encodes protein. For example, only about 1.5% of the human genome consists of protein-coding [[exon]]s, with over 50% of human DNA consisting of non-coding [[repeated sequence (DNA)|repetitive sequences]].<ref>{{cite journal | vauthors = Wolfsberg TG, McEntyre J, Schuler GD | title = Guide to the draft human genome | journal = Nature | volume = 409 | issue = 6822 | pages = 824–26 | date = February 2001 | pmid = 11236998 | doi = 10.1038/35057000 | bibcode = 2001Natur.409..824W | url = https://zenodo.org/record/1233093 | doi-access = free }}</ref> The reasons for the presence of so much [[noncoding DNA]] in eukaryotic genomes and the extraordinary differences in [[genome size]], or ''[[C-value]]'', among species, represent a long-standing puzzle known as the "[[C-value enigma]]".<ref>{{cite journal | vauthors = Gregory TR | title = The C-value enigma in plants and animals: a review of parallels and an appeal for partnership | journal = Annals of Botany | volume = 95 | issue = 1 | pages = 133–46 | date = January 2005 | pmid = 15596463 | doi = 10.1093/aob/mci009 | pmc = 4246714 }}</ref> However, some DNA sequences that do not code protein may still encode functional [[non-coding RNA]] molecules, which are involved in the [[regulation of gene expression]].<ref name="Birney_2007" /> |
In many [[species]], only a small fraction of the total sequence of the [[genome]] encodes protein. For example, only about 1.5% of the human genome consists of protein-coding [[exon]]s, with over 50% of human DNA consisting of non-coding [[repeated sequence (DNA)|repetitive sequences]].<ref>{{cite journal | vauthors = Wolfsberg TG, McEntyre J, Schuler GD | title = Guide to the draft human genome | journal = Nature | volume = 409 | issue = 6822 | pages = 824–26 | date = February 2001 | pmid = 11236998 | doi = 10.1038/35057000 | bibcode = 2001Natur.409..824W | url = https://zenodo.org/record/1233093 | doi-access = free }}</ref> The reasons for the presence of so much [[noncoding DNA]] in eukaryotic genomes and the extraordinary differences in [[genome size]], or ''[[C-value]]'', among species, represent a long-standing puzzle known as the "[[C-value enigma]]".<ref>{{cite journal | vauthors = Gregory TR | title = The C-value enigma in plants and animals: a review of parallels and an appeal for partnership | journal = Annals of Botany | volume = 95 | issue = 1 | pages = 133–46 | date = January 2005 | pmid = 15596463 | doi = 10.1093/aob/mci009 | pmc = 4246714 }}</ref> However, some DNA sequences that do not code protein may still encode functional [[non-coding RNA]] molecules, which are involved in the [[regulation of gene expression]].<ref name="Birney_2007" /> |
||
[[File:T7 RNA polymerase.jpg|thumb|[[T7 RNA polymerase]] (blue) producing an [[Messenger RNA|mRNA]] (green) from a DNA template (orange)<ref> |
[[File:T7 RNA polymerase.jpg|thumb|[[T7 RNA polymerase]] (blue) producing an [[Messenger RNA|mRNA]] (green) from a DNA template (orange)<ref>{{Cite web|last=Bank|first=RCSB Protein Data|title=RCSB PDB - 1MSW: Structural basis for the transition from initiation to elongation transcription in T7 RNA polymerase|url=https://www.rcsb.org/structure/1MSW|access-date=2023-03-27|website=www.rcsb.org|language=en-US}}</ref>]] |
||
Some noncoding DNA sequences play structural roles in chromosomes. [[Telomere]]s and [[centromere]]s typically contain few genes but are important for the function and stability of chromosomes.<ref name=Nugent /><ref>{{cite journal | vauthors = Pidoux AL, Allshire RC | title = The role of heterochromatin in centromere function | journal = Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences | volume = 360 | issue = 1455 | pages = 569–79 | date = March 2005 | pmid = 15905142 | pmc = 1569473 | doi = 10.1098/rstb.2004.1611 }}</ref> An abundant form of noncoding DNA in humans are [[pseudogene]]s, which are copies of genes that have been disabled by mutation.<ref>{{cite journal | vauthors = Harrison PM, Hegyi H, Balasubramanian S, Luscombe NM, Bertone P, Echols N, Johnson T, Gerstein M | title = Molecular fossils in the human genome: identification and analysis of the pseudogenes in chromosomes 21 and 22 | journal = Genome Research | volume = 12 | issue = 2 | pages = 272–80 | date = February 2002 | pmid = 11827946 | pmc = 155275 | doi = 10.1101/gr.207102 }}</ref> These sequences are usually just molecular [[fossil]]s, although they can occasionally serve as raw [[Genome|genetic material]] for the creation of new genes through the process of [[gene duplication]] and [[divergent evolution|divergence]].<ref>{{cite journal | vauthors = Harrison PM, Gerstein M | title = Studying genomes through the aeons: protein families, pseudogenes and proteome evolution | journal = Journal of Molecular Biology | volume = 318 | issue = 5 | pages = 1155–74 | date = May 2002 | pmid = 12083509 | doi = 10.1016/S0022-2836(02)00109-2 }}</ref> |
Some noncoding DNA sequences play structural roles in chromosomes. [[Telomere]]s and [[centromere]]s typically contain few genes but are important for the function and stability of chromosomes.<ref name=Nugent /><ref>{{cite journal | vauthors = Pidoux AL, Allshire RC | title = The role of heterochromatin in centromere function | journal = Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences | volume = 360 | issue = 1455 | pages = 569–79 | date = March 2005 | pmid = 15905142 | pmc = 1569473 | doi = 10.1098/rstb.2004.1611 }}</ref> An abundant form of noncoding DNA in humans are [[pseudogene]]s, which are copies of genes that have been disabled by mutation.<ref>{{cite journal | vauthors = Harrison PM, Hegyi H, Balasubramanian S, Luscombe NM, Bertone P, Echols N, Johnson T, Gerstein M | title = Molecular fossils in the human genome: identification and analysis of the pseudogenes in chromosomes 21 and 22 | journal = Genome Research | volume = 12 | issue = 2 | pages = 272–80 | date = February 2002 | pmid = 11827946 | pmc = 155275 | doi = 10.1101/gr.207102 }}</ref> These sequences are usually just molecular [[fossil]]s, although they can occasionally serve as raw [[Genome|genetic material]] for the creation of new genes through the process of [[gene duplication]] and [[divergent evolution|divergence]].<ref>{{cite journal | vauthors = Harrison PM, Gerstein M | title = Studying genomes through the aeons: protein families, pseudogenes and proteome evolution | journal = Journal of Molecular Biology | volume = 318 | issue = 5 | pages = 1155–74 | date = May 2002 | pmid = 12083509 | doi = 10.1016/S0022-2836(02)00109-2 }}</ref> |
||
| Line 244: | Line 244: | ||
A distinct group of DNA-binding proteins is the DNA-binding proteins that specifically bind single-stranded DNA. In humans, replication [[protein A]] is the best-understood member of this family and is used in processes where the double helix is separated, including DNA replication, recombination, and DNA repair.<ref>{{cite journal | vauthors = Iftode C, Daniely Y, Borowiec JA | title = Replication protein A (RPA): the eukaryotic SSB | journal = Critical Reviews in Biochemistry and Molecular Biology | volume = 34 | issue = 3 | pages = 141–80 | year = 1999 | pmid = 10473346 | doi = 10.1080/10409239991209255 }}</ref> These binding proteins seem to stabilize single-stranded DNA and protect it from forming [[stem-loop]]s or being degraded by [[nuclease]]s. |
A distinct group of DNA-binding proteins is the DNA-binding proteins that specifically bind single-stranded DNA. In humans, replication [[protein A]] is the best-understood member of this family and is used in processes where the double helix is separated, including DNA replication, recombination, and DNA repair.<ref>{{cite journal | vauthors = Iftode C, Daniely Y, Borowiec JA | title = Replication protein A (RPA): the eukaryotic SSB | journal = Critical Reviews in Biochemistry and Molecular Biology | volume = 34 | issue = 3 | pages = 141–80 | year = 1999 | pmid = 10473346 | doi = 10.1080/10409239991209255 }}</ref> These binding proteins seem to stabilize single-stranded DNA and protect it from forming [[stem-loop]]s or being degraded by [[nuclease]]s. |
||
[[File:Lambda repressor 1LMB.png|thumb|upright=1.1|The lambda repressor [[helix-turn-helix]] transcription factor bound to its DNA target<ref> |
[[File:Lambda repressor 1LMB.png|thumb|upright=1.1|The lambda repressor [[helix-turn-helix]] transcription factor bound to its DNA target<ref>{{Cite web|last=Bank|first=RCSB Protein Data|title=RCSB PDB - 1LMB: REFINED 1.8 ANGSTROM CRYSTAL STRUCTURE OF THE LAMBDA REPRESSOR-OPERATOR COMPLEX|url=https://www.rcsb.org/structure/1LMB|access-date=2023-03-27|website=www.rcsb.org|language=en-US}}</ref>]] |
||
In contrast, other proteins have evolved to bind to particular DNA sequences. The most intensively studied of these are the various [[transcription factor]]s, which are proteins that regulate transcription. Each transcription factor binds to one particular set of DNA sequences and activates or inhibits the transcription of genes that have these sequences close to their promoters. The transcription factors do this in two ways. Firstly, they can bind the RNA polymerase responsible for transcription, either directly or through other mediator proteins; this locates the polymerase at the promoter and allows it to begin transcription.<ref>{{cite journal | vauthors = Myers LC, Kornberg RD | title = Mediator of transcriptional regulation | journal = Annual Review of Biochemistry | volume = 69 | pages = 729–49 | year = 2000 | pmid = 10966474 | doi = 10.1146/annurev.biochem.69.1.729 }}</ref> Alternatively, transcription factors can bind [[enzyme]]s that modify the histones at the promoter. This changes the accessibility of the DNA template to the polymerase.<ref>{{cite journal | vauthors = Spiegelman BM, Heinrich R | title = Biological control through regulated transcriptional coactivators | journal = Cell | volume = 119 | issue = 2 | pages = 157–67 | date = October 2004 | pmid = 15479634 | doi = 10.1016/j.cell.2004.09.037 | doi-access = free }}</ref> |
In contrast, other proteins have evolved to bind to particular DNA sequences. The most intensively studied of these are the various [[transcription factor]]s, which are proteins that regulate transcription. Each transcription factor binds to one particular set of DNA sequences and activates or inhibits the transcription of genes that have these sequences close to their promoters. The transcription factors do this in two ways. Firstly, they can bind the RNA polymerase responsible for transcription, either directly or through other mediator proteins; this locates the polymerase at the promoter and allows it to begin transcription.<ref>{{cite journal | vauthors = Myers LC, Kornberg RD | title = Mediator of transcriptional regulation | journal = Annual Review of Biochemistry | volume = 69 | pages = 729–49 | year = 2000 | pmid = 10966474 | doi = 10.1146/annurev.biochem.69.1.729 }}</ref> Alternatively, transcription factors can bind [[enzyme]]s that modify the histones at the promoter. This changes the accessibility of the DNA template to the polymerase.<ref>{{cite journal | vauthors = Spiegelman BM, Heinrich R | title = Biological control through regulated transcriptional coactivators | journal = Cell | volume = 119 | issue = 2 | pages = 157–67 | date = October 2004 | pmid = 15479634 | doi = 10.1016/j.cell.2004.09.037 | doi-access = free }}</ref> |
||
As these DNA targets can occur throughout an organism's genome, changes in the activity of one type of transcription factor can affect thousands of genes.<ref>{{cite journal | vauthors = Li Z, Van Calcar S, Qu C, Cavenee WK, Zhang MQ, Ren B | title = A global transcriptional regulatory role for c-Myc in Burkitt's lymphoma cells | journal = Proceedings of the National Academy of Sciences of the United States of America | volume = 100 | issue = 14 | pages = 8164–69 | date = July 2003 | pmid = 12808131 | pmc = 166200 | doi = 10.1073/pnas.1332764100 | bibcode = 2003PNAS..100.8164L | doi-access = free }}</ref> Consequently, these proteins are often the targets of the [[signal transduction]] processes that control responses to environmental changes or [[cellular differentiation]] and development. The specificity of these transcription factors' interactions with DNA come from the proteins making multiple contacts to the edges of the DNA bases, allowing them to "read" the DNA sequence. Most of these base-interactions are made in the major groove, where the bases are most accessible.<ref name="Pabo1984" /> |
As these DNA targets can occur throughout an organism's genome, changes in the activity of one type of transcription factor can affect thousands of genes.<ref>{{cite journal | vauthors = Li Z, Van Calcar S, Qu C, Cavenee WK, Zhang MQ, Ren B | title = A global transcriptional regulatory role for c-Myc in Burkitt's lymphoma cells | journal = Proceedings of the National Academy of Sciences of the United States of America | volume = 100 | issue = 14 | pages = 8164–69 | date = July 2003 | pmid = 12808131 | pmc = 166200 | doi = 10.1073/pnas.1332764100 | bibcode = 2003PNAS..100.8164L | doi-access = free }}</ref> Consequently, these proteins are often the targets of the [[signal transduction]] processes that control responses to environmental changes or [[cellular differentiation]] and development. The specificity of these transcription factors' interactions with DNA come from the proteins making multiple contacts to the edges of the DNA bases, allowing them to "read" the DNA sequence. Most of these base-interactions are made in the major groove, where the bases are most accessible.<ref name="Pabo1984" /> |
||
[[File:EcoRV 1RVA.png|thumb|left|upright=1.1|The [[restriction enzyme]] [[EcoRV]] (green) in a complex with its substrate DNA<ref> |
[[File:EcoRV 1RVA.png|thumb|left|upright=1.1|The [[restriction enzyme]] [[EcoRV]] (green) in a complex with its substrate DNA<ref>{{Cite web|last=Bank|first=RCSB Protein Data|title=RCSB PDB - 1RVA: MG2+ BINDING TO THE ACTIVE SITE OF ECO RV ENDONUCLEASE: A CRYSTALLOGRAPHIC STUDY OF COMPLEXES WITH SUBSTRATE AND PRODUCT DNA AT 2 ANGSTROMS RESOLUTION|url=https://www.rcsb.org/structure/1RVA|access-date=2023-03-27|website=www.rcsb.org|language=en-US}}</ref>]] |
||
=== DNA-modifying enzymes === |
=== DNA-modifying enzymes === |
||
| Line 280: | Line 280: | ||
|[[File:Holliday junction coloured.png|250px]] |
|[[File:Holliday junction coloured.png|250px]] |
||
|} |
|} |
||
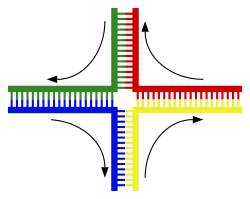
<div style="border: none; width:250px;"><div class="thumbcaption">Structure of the [[Holliday junction]] intermediate in [[genetic recombination]]. The four separate DNA strands are coloured red, blue, green and yellow.<ref> |
<div style="border: none; width:250px;"><div class="thumbcaption">Structure of the [[Holliday junction]] intermediate in [[genetic recombination]]. The four separate DNA strands are coloured red, blue, green and yellow.<ref>{{Cite web|last=Bank|first=RCSB Protein Data|title=RCSB PDB - 1M6G: Structural Characterisation of the Holliday Junction TCGGTACCGA|url=https://www.rcsb.org/structure/1M6G|access-date=2023-03-27|website=www.rcsb.org|language=en-US}}</ref></div></div></div> |
||
{{further|Genetic recombination}} |
{{further|Genetic recombination}} |
||
[[File:Homologous Recombination.jpg|thumb|left|300px| A current model of meiotic recombination, initiated by a double-strand break or gap, followed by pairing with an homologous chromosome and strand invasion to initiate the recombinational repair process. Repair of the gap can lead to crossover (CO) or non-crossover (NCO) of the flanking regions. CO recombination is thought to occur by the Double Holliday Junction (DHJ) model, illustrated on the right, above. NCO recombinants are thought to occur primarily by the Synthesis Dependent Strand Annealing (SDSA) model, illustrated on the left, above. Most recombination events appear to be the SDSA type.]] |
[[File:Homologous Recombination.jpg|thumb|left|300px| A current model of meiotic recombination, initiated by a double-strand break or gap, followed by pairing with an homologous chromosome and strand invasion to initiate the recombinational repair process. Repair of the gap can lead to crossover (CO) or non-crossover (NCO) of the flanking regions. CO recombination is thought to occur by the Double Holliday Junction (DHJ) model, illustrated on the right, above. NCO recombinants are thought to occur primarily by the Synthesis Dependent Strand Annealing (SDSA) model, illustrated on the left, above. Most recombination events appear to be the SDSA type.]] |
||
| Line 307: | Line 307: | ||
{{further|DNA profiling}} |
{{further|DNA profiling}} |
||
[[Forensic science|Forensic scientists]] can use DNA in [[blood]], [[semen]], [[skin]], [[saliva]] or [[hair]] found at a [[crime scene]] to identify a matching DNA of an individual, such as a perpetrator.<ref>{{Cite news|url=https://theconversation.com/from-the-crime-scene-to-the-courtroom-the-journey-of-a-dna-sample-82250|title=From the crime scene to the courtroom: the journey of a DNA sample|last1=Curtis|first1=Caitlin |last2=Hereward |first2=James | name-list-style = vanc |date=29 August 2017 |work=The Conversation |access-date=22 October 2017 |archive-url=https://web.archive.org/web/20171022033110/http://theconversation.com/from-the-crime-scene-to-the-courtroom-the-journey-of-a-dna-sample-82250 |archive-date=22 October 2017 |url-status=live }}</ref> This process is formally termed [[DNA profiling]], also called ''DNA fingerprinting''. In DNA profiling, the lengths of variable sections of repetitive DNA, such as [[short tandem repeat]]s and [[minisatellite]]s, are compared between people. This method is usually an extremely reliable technique for identifying a matching DNA.<ref>{{cite journal | vauthors = Collins A, Morton NE | title = Likelihood ratios for DNA identification | journal = Proceedings of the National Academy of Sciences of the United States of America | volume = 91 | issue = 13 | pages = 6007–11 | date = June 1994 | pmid = 8016106 | pmc = 44126 | doi = 10.1073/pnas.91.13.6007 | bibcode = 1994PNAS...91.6007C | doi-access = free }}</ref> However, identification can be complicated if the scene is contaminated with DNA from several people.<ref>{{cite journal | vauthors = Weir BS, Triggs CM, Starling L, Stowell LI, Walsh KA, Buckleton J | title = Interpreting DNA mixtures | journal = Journal of Forensic Sciences | volume = 42 | issue = 2 | pages = 213–22 | date = March 1997 | doi = 10.1520/JFS14100J | pmid = 9068179 | s2cid = 14511630 | url = http://pdfs.semanticscholar.org/f47f/2c895d0b06b3dc72a4707b464126e6c820aa.pdf }}</ref> DNA profiling was developed in 1984 by British geneticist Sir [[Alec Jeffreys]],<ref>{{cite journal | vauthors = Jeffreys AJ, Wilson V, Thein SL | title = Individual-specific 'fingerprints' of human DNA | journal = Nature | volume = 316 | issue = 6023 | pages = 76–79 | year = 1985 | pmid = 2989708 | doi = 10.1038/316076a0 | bibcode = 1985Natur.316...76J | s2cid = 4229883 | doi-access = free }}</ref> and first used in forensic science to convict Colin Pitchfork in the 1988 [[Colin Pitchfork|Enderby murders]] case.<ref> |
[[Forensic science|Forensic scientists]] can use DNA in [[blood]], [[semen]], [[skin]], [[saliva]] or [[hair]] found at a [[crime scene]] to identify a matching DNA of an individual, such as a perpetrator.<ref>{{Cite news|url=https://theconversation.com/from-the-crime-scene-to-the-courtroom-the-journey-of-a-dna-sample-82250|title=From the crime scene to the courtroom: the journey of a DNA sample|last1=Curtis|first1=Caitlin |last2=Hereward |first2=James | name-list-style = vanc |date=29 August 2017 |work=The Conversation |access-date=22 October 2017 |archive-url=https://web.archive.org/web/20171022033110/http://theconversation.com/from-the-crime-scene-to-the-courtroom-the-journey-of-a-dna-sample-82250 |archive-date=22 October 2017 |url-status=live }}</ref> This process is formally termed [[DNA profiling]], also called ''DNA fingerprinting''. In DNA profiling, the lengths of variable sections of repetitive DNA, such as [[short tandem repeat]]s and [[minisatellite]]s, are compared between people. This method is usually an extremely reliable technique for identifying a matching DNA.<ref>{{cite journal | vauthors = Collins A, Morton NE | title = Likelihood ratios for DNA identification | journal = Proceedings of the National Academy of Sciences of the United States of America | volume = 91 | issue = 13 | pages = 6007–11 | date = June 1994 | pmid = 8016106 | pmc = 44126 | doi = 10.1073/pnas.91.13.6007 | bibcode = 1994PNAS...91.6007C | doi-access = free }}</ref> However, identification can be complicated if the scene is contaminated with DNA from several people.<ref>{{cite journal | vauthors = Weir BS, Triggs CM, Starling L, Stowell LI, Walsh KA, Buckleton J | title = Interpreting DNA mixtures | journal = Journal of Forensic Sciences | volume = 42 | issue = 2 | pages = 213–22 | date = March 1997 | doi = 10.1520/JFS14100J | pmid = 9068179 | s2cid = 14511630 | url = http://pdfs.semanticscholar.org/f47f/2c895d0b06b3dc72a4707b464126e6c820aa.pdf }}</ref> DNA profiling was developed in 1984 by British geneticist Sir [[Alec Jeffreys]],<ref>{{cite journal | vauthors = Jeffreys AJ, Wilson V, Thein SL | title = Individual-specific 'fingerprints' of human DNA | journal = Nature | volume = 316 | issue = 6023 | pages = 76–79 | year = 1985 | pmid = 2989708 | doi = 10.1038/316076a0 | bibcode = 1985Natur.316...76J | s2cid = 4229883 | doi-access = free }}</ref> and first used in forensic science to convict Colin Pitchfork in the 1988 [[Colin Pitchfork|Enderby murders]] case.<ref>{{Cite web|date=2006-12-14|title=Colin Pitchfork|url=https://web.archive.org/web/20061214004903/http://www.forensic.gov.uk/forensic_t/inside/news/list_casefiles.php?case=1|access-date=2023-03-27|website=web.archive.org}}</ref> |
||
The development of forensic science and the ability to now obtain genetic matching on minute samples of blood, skin, saliva, or hair has led to re-examining many cases. Evidence can now be uncovered that was scientifically impossible at the time of the original examination. Combined with the removal of the [[double jeopardy]] law in some places, this can allow cases to be reopened where prior trials have failed to produce sufficient evidence to convince a jury. People charged with serious crimes may be required to provide a sample of DNA for matching purposes. The most obvious defense to DNA matches obtained forensically is to claim that cross-contamination of evidence has occurred. This has resulted in meticulous strict handling procedures with new cases of serious crime. |
The development of forensic science and the ability to now obtain genetic matching on minute samples of blood, skin, saliva, or hair has led to re-examining many cases. Evidence can now be uncovered that was scientifically impossible at the time of the original examination. Combined with the removal of the [[double jeopardy]] law in some places, this can allow cases to be reopened where prior trials have failed to produce sufficient evidence to convince a jury. People charged with serious crimes may be required to provide a sample of DNA for matching purposes. The most obvious defense to DNA matches obtained forensically is to claim that cross-contamination of evidence has occurred. This has resulted in meticulous strict handling procedures with new cases of serious crime. |
||
| Line 313: | Line 313: | ||
DNA profiling is also used successfully to positively identify victims of mass casualty incidents,<ref>{{cite web|url=http://massfatality.dna.gov/Introduction/ |title=DNA Identification in Mass Fatality Incidents |date=September 2006 |publisher=National Institute of Justice |url-status=dead |archive-url=https://web.archive.org/web/20061112000837/http://massfatality.dna.gov/Introduction/ |archive-date=12 November 2006 }}</ref> bodies or body parts in serious accidents, and individual victims in mass war graves, via matching to family members. |
DNA profiling is also used successfully to positively identify victims of mass casualty incidents,<ref>{{cite web|url=http://massfatality.dna.gov/Introduction/ |title=DNA Identification in Mass Fatality Incidents |date=September 2006 |publisher=National Institute of Justice |url-status=dead |archive-url=https://web.archive.org/web/20061112000837/http://massfatality.dna.gov/Introduction/ |archive-date=12 November 2006 }}</ref> bodies or body parts in serious accidents, and individual victims in mass war graves, via matching to family members. |
||
DNA profiling is also used in [[DNA paternity testing]] to determine if someone is the biological parent or grandparent of a child with the probability of parentage is typically 99.99% when the alleged parent is biologically related to the child. Normal [[DNA sequencing]] methods happen after birth, but there are new methods to test paternity while a mother is still pregnant.<ref> |
DNA profiling is also used in [[DNA paternity testing]] to determine if someone is the biological parent or grandparent of a child with the probability of parentage is typically 99.99% when the alleged parent is biologically related to the child. Normal [[DNA sequencing]] methods happen after birth, but there are new methods to test paternity while a mother is still pregnant.<ref>{{Cite news|last=Pollack|first=Andrew|date=2012-06-19|title=Before Birth, Dad’s ID|language=en-US|work=The New York Times|url=https://www.nytimes.com/2012/06/20/health/paternity-blood-tests-that-work-early-in-a-pregnancy.html|access-date=2023-03-27|issn=0362-4331}}</ref> |
||
=== DNA enzymes or catalytic DNA === |
=== DNA enzymes or catalytic DNA === |
||
| Line 369: | Line 369: | ||
[[File:TheEaglePub-Cambridge-BluePlaque.jpg|thumb|left|A [[blue plaque]] outside [[The Eagle, Cambridge|The Eagle]] [[pub]] commemorating Crick and Watson]] |
[[File:TheEaglePub-Cambridge-BluePlaque.jpg|thumb|left|A [[blue plaque]] outside [[The Eagle, Cambridge|The Eagle]] [[pub]] commemorating Crick and Watson]] |
||
In May 1952, [[Raymond Gosling]], a graduate student working under the supervision of [[Rosalind Franklin]], took an [[X-ray diffraction]] image, labeled as "[[Photo 51]]",<ref> |
In May 1952, [[Raymond Gosling]], a graduate student working under the supervision of [[Rosalind Franklin]], took an [[X-ray diffraction]] image, labeled as "[[Photo 51]]",<ref>{{Cite web|title=Linus Pauling and the Race for DNA: A Documentary History - Special Collections & Archives Research Center - Oregon State University|url=http://scarc.library.oregonstate.edu/coll/pauling/dna/index.html|access-date=2023-03-27|website=scarc.library.oregonstate.edu}}</ref> at high hydration levels of DNA. This photo was given to Watson and Crick by [[Maurice Wilkins]] and was critical to their obtaining the correct structure of DNA. Franklin told Crick and Watson that the backbones had to be on the outside. Before then, Linus Pauling, and Watson and Crick, had erroneous models with the chains inside and the bases pointing outwards. Franklin's identification of the [[space group]] for DNA crystals revealed to Crick that the two DNA strands were [[Antiparallel (biochemistry)|antiparallel]].<ref name="ReferenceA">{{cite book|last=Schwartz|first=James | name-list-style = vanc |title=In pursuit of the gene : from Darwin to DNA|url=https://archive.org/details/inpursuitofgenef00schw|url-access=registration|year=2008|publisher=Harvard University Press|location=Cambridge, Mass.|isbn=978-0-674-02670-4 }}</ref> In February 1953, [[Linus Pauling]] and [[Robert Corey]] proposed a model for nucleic acids containing three intertwined chains, with the phosphates near the axis, and the bases on the outside.<ref name="pmid16578429">{{cite journal | vauthors = Pauling L, Corey RB | title = A Proposed Structure For The Nucleic Acids | journal = Proceedings of the National Academy of Sciences of the United States of America | volume = 39 | issue = 2 | pages = 84–97 | date = February 1953 | pmid = 16578429 | pmc = 1063734 | doi = 10.1073/pnas.39.2.84| url = http://scarc.library.oregonstate.edu/coll/pauling/dna/papers/1953p.9-084.html | bibcode = 1953PNAS...39...84P | doi-access = free }}</ref> Watson and Crick completed their model, which is now accepted as the first correct model of the double helix of [[Molecular structure of Nucleic Acids|DNA]]. On 28 February 1953 Crick interrupted patrons' lunchtime at [[The Eagle, Cambridge|The Eagle]] [[pub]] in Cambridge to announce that he and Watson had "discovered the secret of life".<ref>{{cite book | last = Regis | first = Ed | name-list-style = vanc | date = 2009 | title = What Is Life?: investigating the nature of life in the age of synthetic biology | location = Oxford | publisher = [[Oxford University Press]] | isbn = 978-0-19-538341-6 | page = 52 }}</ref> |
||
The 25 April 1953 issue of the journal ''[[Nature (journal)|Nature]]'' published a series of five articles giving the Watson and Crick double-helix structure DNA and evidence supporting it.<ref name=NatureDNA50>{{cite web | work = Nature Archives | url = http://www.nature.com/nature/dna50/archive.html | title = Double Helix of DNA: 50 Years | archive-url = https://web.archive.org/web/20150405140401/http://www.nature.com/nature/dna50/archive.html | archive-date=5 April 2015 | url-status=dead }}</ref> The structure was reported in a letter titled "''MOLECULAR STRUCTURE OF NUCLEIC ACIDS A Structure for Deoxyribose Nucleic Acid''{{-"}}, in which they said, "It has not escaped our notice that the specific pairing we have postulated immediately suggests a possible copying mechanism for the genetic material."<ref name="autogenerated2" /> This letter was followed by a letter from Franklin and Gosling, which was the first publication of their own X-ray diffraction data and of their original analysis method.<ref name=NatFranGos /><ref>{{cite web | url = http://osulibrary.oregonstate.edu/specialcollections/coll/pauling/dna/pictures/franklin-typeBphoto.html | title = Original X-ray diffraction image | publisher = Oregon State Library | access-date = 6 February 2011 | url-status=live | archive-url = https://web.archive.org/web/20090130111849/http://osulibrary.oregonstate.edu/specialcollections/coll/pauling/dna/pictures/franklin-typeBphoto.html | archive-date=30 January 2009 }}</ref> Then followed a letter by Wilkins and two of his colleagues, which contained an analysis of ''in vivo'' B-DNA X-ray patterns, and which supported the presence ''in vivo'' of the Watson and Crick structure.<ref name="NatWilk" /> |
The 25 April 1953 issue of the journal ''[[Nature (journal)|Nature]]'' published a series of five articles giving the Watson and Crick double-helix structure DNA and evidence supporting it.<ref name=NatureDNA50>{{cite web | work = Nature Archives | url = http://www.nature.com/nature/dna50/archive.html | title = Double Helix of DNA: 50 Years | archive-url = https://web.archive.org/web/20150405140401/http://www.nature.com/nature/dna50/archive.html | archive-date=5 April 2015 | url-status=dead }}</ref> The structure was reported in a letter titled "''MOLECULAR STRUCTURE OF NUCLEIC ACIDS A Structure for Deoxyribose Nucleic Acid''{{-"}}, in which they said, "It has not escaped our notice that the specific pairing we have postulated immediately suggests a possible copying mechanism for the genetic material."<ref name="autogenerated2" /> This letter was followed by a letter from Franklin and Gosling, which was the first publication of their own X-ray diffraction data and of their original analysis method.<ref name=NatFranGos /><ref>{{cite web | url = http://osulibrary.oregonstate.edu/specialcollections/coll/pauling/dna/pictures/franklin-typeBphoto.html | title = Original X-ray diffraction image | publisher = Oregon State Library | access-date = 6 February 2011 | url-status=live | archive-url = https://web.archive.org/web/20090130111849/http://osulibrary.oregonstate.edu/specialcollections/coll/pauling/dna/pictures/franklin-typeBphoto.html | archive-date=30 January 2009 }}</ref> Then followed a letter by Wilkins and two of his colleagues, which contained an analysis of ''in vivo'' B-DNA X-ray patterns, and which supported the presence ''in vivo'' of the Watson and Crick structure.<ref name="NatWilk" /> |
||
Revision as of 02:25, 27 March 2023
| Part of a series on |
| Genetics |
|---|
 |
Deoxyribonucleic acid (
The two DNA strands are known as polynucleotides as they are composed of simpler
Both strands of double-stranded DNA store the same
Within eukaryotic cells, DNA is organized into long structures called chromosomes. Before typical cell division, these chromosomes are duplicated in the process of DNA replication, providing a complete set of chromosomes for each daughter cell. Eukaryotic organisms (animals, plants, fungi and protists) store most of their DNA inside the cell nucleus as nuclear DNA, and some in the mitochondria as mitochondrial DNA or in chloroplasts as chloroplast DNA.[5] In contrast, prokaryotes (bacteria and archaea) store their DNA only in the cytoplasm, in circular chromosomes. Within eukaryotic chromosomes, chromatin proteins, such as histones, compact and organize DNA. These compacting structures guide the interactions between DNA and other proteins, helping control which parts of the DNA are transcribed.
Properties
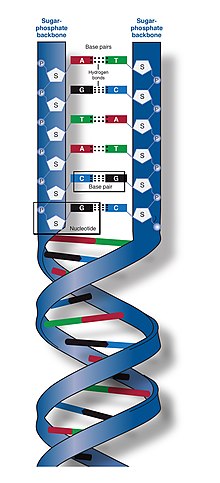
DNA is a long
DNA does not usually exist as a single strand, but instead as a pair of strands that are held tightly together.
The backbone of the DNA strand is made from alternating phosphate and sugar groups.[13] The sugar in DNA is 2-deoxyribose, which is a pentose (five-carbon) sugar. The sugars are joined by phosphate groups that form phosphodiester bonds between the third and fifth carbon atoms of adjacent sugar rings. These are known as the 3′-end (three prime end), and 5′-end (five prime end) carbons, the prime symbol being used to distinguish these carbon atoms from those of the base to which the deoxyribose forms a glycosidic bond.[11]
Therefore, any DNA strand normally has one end at which there is a phosphate group attached to the 5′ carbon of a ribose (the 5′ phosphoryl) and another end at which there is a free hydroxyl group attached to the 3′ carbon of a ribose (the 3′ hydroxyl). The orientation of the 3′ and 5′ carbons along the sugar-phosphate backbone confers directionality (sometimes called polarity) to each DNA strand. In a nucleic acid double helix, the direction of the nucleotides in one strand is opposite to their direction in the other strand: the strands are antiparallel. The asymmetric ends of DNA strands are said to have a directionality of five prime end (5′ ), and three prime end (3′), with the 5′ end having a terminal phosphate group and the 3′ end a terminal hydroxyl group. One major difference between DNA and RNA is the sugar, with the 2-deoxyribose in DNA being replaced by the related pentose sugar ribose in RNA.[11]
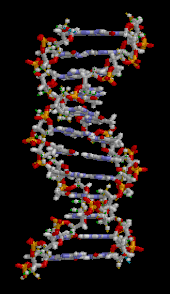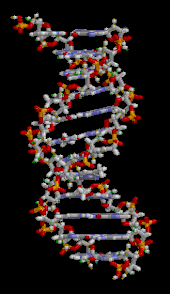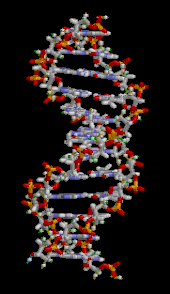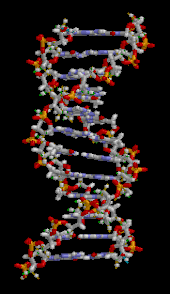
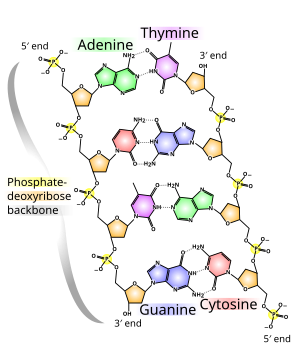
The DNA double helix is stabilized primarily by two forces:
Nucleobase classification
The nucleobases are classified into two types: the purines, A and G, which are fused five- and six-membered heterocyclic compounds, and the pyrimidines, the six-membered rings C and T.[11] A fifth pyrimidine nucleobase, uracil (U), usually takes the place of thymine in RNA and differs from thymine by lacking a methyl group on its ring. In addition to RNA and DNA, many artificial nucleic acid analogues have been created to study the properties of nucleic acids, or for use in biotechnology.[18]
Non-canonical bases
Modified bases occur in DNA. The first of these recognized was
A number of noncanonical bases are known to occur in DNA.[22] Most of these are modifications of the canonical bases plus uracil.
- Modified Adenine
- N6-carbamoyl-methyladenine
- N6-methyadenine
- Modified Guanine
- 7-Deazaguanine
- 7-Methylguanine
- Modified Cytosine
- N4-Methylcytosine
- 5-Carboxylcytosine
- 5-Formylcytosine
- 5-Glycosylhydroxymethylcytosine
- 5-Hydroxycytosine
- 5-Methylcytosine
- Modified Thymidine
- α-Glutamythymidine
- α-Putrescinylthymine
- Uracil and modifications
- Base J
- Uracil
- 5-Dihydroxypentauracil
- 5-Hydroxymethyldeoxyuracil
- Others
- Deoxyarchaeosine
- 2,6-Diaminopurine (2-Aminoadenine)
Grooves
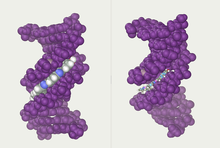
Twin helical strands form the DNA backbone. Another double helix may be found tracing the spaces, or grooves, between the strands. These voids are adjacent to the base pairs and may provide a
Base pairing

|
|
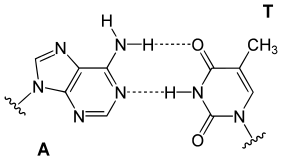
In a DNA double helix, each type of nucleobase on one strand bonds with just one type of nucleobase on the other strand. This is called complementary base pairing. Purines form hydrogen bonds to pyrimidines, with adenine bonding only to thymine in two hydrogen bonds, and cytosine bonding only to guanine in three hydrogen bonds. This arrangement of two nucleotides binding together across the double helix (from six-carbon ring to six-carbon ring) is called a Watson-Crick base pair. DNA with high GC-content is more stable than DNA with low GC-content. A Hoogsteen base pair (hydrogen bonding the 6-carbon ring to the 5-carbon ring) is a rare variation of base-pairing.[25] As hydrogen bonds are not covalent, they can be broken and rejoined relatively easily. The two strands of DNA in a double helix can thus be pulled apart like a zipper, either by a mechanical force or high temperature.[26] As a result of this base pair complementarity, all the information in the double-stranded sequence of a DNA helix is duplicated on each strand, which is vital in DNA replication. This reversible and specific interaction between complementary base pairs is critical for all the functions of DNA in organisms.[7]
ssDNA vs. dsDNA
As noted above, most DNA molecules are actually two polymer strands, bound together in a helical fashion by noncovalent bonds; this double-stranded (dsDNA) structure is maintained largely by the intrastrand base stacking interactions, which are strongest for G,C stacks. The two strands can come apart—a process known as melting—to form two single-stranded DNA (ssDNA) molecules. Melting occurs at high temperatures, low salt and high pH (low pH also melts DNA, but since DNA is unstable due to acid depurination, low pH is rarely used).
The stability of the dsDNA form depends not only on the GC-content (% G,C basepairs) but also on sequence (since stacking is sequence specific) and also length (longer molecules are more stable). The stability can be measured in various ways; a common way is the
In the laboratory, the strength of this interaction can be measured by finding the melting temperature Tm necessary to break half of the hydrogen bonds. When all the base pairs in a DNA double helix melt, the strands separate and exist in solution as two entirely independent molecules. These single-stranded DNA molecules have no single common shape, but some conformations are more stable than others.[29]
Amount

In humans, the total female
In eukaryotes, in addition to nuclear DNA, there is also mitochondrial DNA (mtDNA) which encodes certain proteins used by the mitochondria. The mtDNA is usually relatively small in comparison to the nuclear DNA. For example, the human mitochondrial DNA forms closed circular molecules, each of which contains 16,569[32][33] DNA base pairs,[34] with each such molecule normally containing a full set of the mitochondrial genes. Each human mitochondrion contains, on average, approximately 5 such mtDNA molecules.[34] Each human cell contains approximately 100 mitochondria, giving a total number of mtDNA molecules per human cell of approximately 500.[34] However, the amount of mitochondria per cell also varies by cell type, and an egg cell can contain 100,000 mitochondria, corresponding to up to 1,500,000 copies of the mitochondrial genome (constituting up to 90% of the DNA of the cell).[35]
Sense and antisense
A DNA sequence is called a "sense" sequence if it is the same as that of a messenger RNA copy that is translated into protein.[36] The sequence on the opposite strand is called the "antisense" sequence. Both sense and antisense sequences can exist on different parts of the same strand of DNA (i.e. both strands can contain both sense and antisense sequences). In both prokaryotes and eukaryotes, antisense RNA sequences are produced, but the functions of these RNAs are not entirely clear.[37] One proposal is that antisense RNAs are involved in regulating gene expression through RNA-RNA base pairing.[38]
A few DNA sequences in prokaryotes and eukaryotes, and more in plasmids and viruses, blur the distinction between sense and antisense strands by having overlapping genes.[39] In these cases, some DNA sequences do double duty, encoding one protein when read along one strand, and a second protein when read in the opposite direction along the other strand. In bacteria, this overlap may be involved in the regulation of gene transcription,[40] while in viruses, overlapping genes increase the amount of information that can be encoded within the small viral genome.[41]
Supercoiling
DNA can be twisted like a rope in a process called
Alternative DNA structures
DNA exists in many possible
The first published reports of A-DNA X-ray diffraction patterns—and also B-DNA—used analyses based on Patterson functions that provided only a limited amount of structural information for oriented fibers of DNA.[46][47] An alternative analysis was proposed by Wilkins et al. in 1953 for the in vivo B-DNA X-ray diffraction-scattering patterns of highly hydrated DNA fibers in terms of squares of Bessel functions.[48] In the same journal, James Watson and Francis Crick presented their molecular modeling analysis of the DNA X-ray diffraction patterns to suggest that the structure was a double helix.[9]
Although the B-DNA form is most common under the conditions found in cells,
Compared to B-DNA, the A-DNA form is a wider right-handed spiral, with a shallow, wide minor groove and a narrower, deeper major groove. The A form occurs under non-physiological conditions in partly dehydrated samples of DNA, while in the cell it may be produced in hybrid pairings of DNA and RNA strands, and in enzyme-DNA complexes.[53][54] Segments of DNA where the bases have been chemically modified by methylation may undergo a larger change in conformation and adopt the Z form. Here, the strands turn about the helical axis in a left-handed spiral, the opposite of the more common B form.[55] These unusual structures can be recognized by specific Z-DNA binding proteins and may be involved in the regulation of transcription.[56]
Alternative DNA chemistry
For many years,
Quadruplex structures

At the ends of the linear chromosomes are specialized regions of DNA called telomeres. The main function of these regions is to allow the cell to replicate chromosome ends using the enzyme telomerase, as the enzymes that normally replicate DNA cannot copy the extreme 3′ ends of chromosomes.[62] These specialized chromosome caps also help protect the DNA ends, and stop the DNA repair systems in the cell from treating them as damage to be corrected.[63] In human cells, telomeres are usually lengths of single-stranded DNA containing several thousand repeats of a simple TTAGGG sequence.[64]
These guanine-rich sequences may stabilize chromosome ends by forming structures of stacked sets of four-base units, rather than the usual base pairs found in other DNA molecules. Here, four guanine bases, known as a guanine tetrad, form a flat plate. These flat four-base units then stack on top of each other to form a stable G-quadruplex structure.[65] These structures are stabilized by hydrogen bonding between the edges of the bases and chelation of a metal ion in the centre of each four-base unit.[66] Other structures can also be formed, with the central set of four bases coming from either a single strand folded around the bases, or several different parallel strands, each contributing one base to the central structure.
In addition to these stacked structures, telomeres also form large loop structures called telomere loops, or T-loops. Here, the single-stranded DNA curls around in a long circle stabilized by telomere-binding proteins.[67] At the very end of the T-loop, the single-stranded telomere DNA is held onto a region of double-stranded DNA by the telomere strand disrupting the double-helical DNA and base pairing to one of the two strands. This triple-stranded structure is called a displacement loop or D-loop.[65]
Branched DNA
In DNA,
Artificial bases
Several artificial nucleobases have been synthesized, and successfully incorporated in the eight-base DNA analogue named Hachimoji DNA. Dubbed S, B, P, and Z, these artificial bases are capable of bonding with each other in a predictable way (S–B and P–Z), maintain the double helix structure of DNA, and be transcribed to RNA. Their existence could be seen as an indication that there is nothing special about the four natural nucleobases that evolved on Earth.[69][70] On the other hand, DNA is tightly related to RNA which does not only act as a transcript of DNA but also performs as molecular machines many tasks in cells. For this purpose it has to fold into a structure. It has been shown that to allow to create all possible structures at least four bases are required for the corresponding RNA,[71] while a higher number is also possible but this would be against the natural principle of least effort.
Acidity
The phosphate groups of DNA give it similar acidic properties to phosphoric acid and it can be considered as a strong acid. It will be fully ionized at a normal cellular pH, releasing protons which leave behind negative charges on the phosphate groups. These negative charges protect DNA from breakdown by hydrolysis by repelling nucleophiles which could hydrolyze it.[72]
Macroscopic appearance

Pure DNA extracted from cells forms white, stringy clumps.[73]
Chemical modifications and altered DNA packaging
|

|

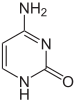
|
| cytosine | 5-methylcytosine | thymine |
Base modifications and DNA packaging
The expression of genes is influenced by how the DNA is packaged in chromosomes, in a structure called chromatin. Base modifications can be involved in packaging, with regions that have low or no gene expression usually containing high levels of methylation of cytosine bases. DNA packaging and its influence on gene expression can also occur by covalent modifications of the histone protein core around which DNA is wrapped in the chromatin structure or else by remodeling carried out by chromatin remodeling complexes (see Chromatin remodeling). There is, further, crosstalk between DNA methylation and histone modification, so they can coordinately affect chromatin and gene expression.[74]
For one example, cytosine methylation produces
Damage
DNA can be damaged by many sorts of
Many mutagens fit into the space between two adjacent base pairs, this is called
Biological functions

DNA usually occurs as linear
Genes and genomes
Genomic DNA is tightly and orderly packed in the process called
In many

Some noncoding DNA sequences play structural roles in chromosomes. Telomeres and centromeres typically contain few genes but are important for the function and stability of chromosomes.[63][101] An abundant form of noncoding DNA in humans are pseudogenes, which are copies of genes that have been disabled by mutation.[102] These sequences are usually just molecular fossils, although they can occasionally serve as raw genetic material for the creation of new genes through the process of gene duplication and divergence.[103]
Transcription and translation
A gene is a sequence of DNA that contains genetic information and can influence the phenotype of an organism. Within a gene, the sequence of bases along a DNA strand defines a messenger RNA sequence, which then defines one or more protein sequences. The relationship between the nucleotide sequences of genes and the amino-acid sequences of proteins is determined by the rules of translation, known collectively as the genetic code. The genetic code consists of three-letter 'words' called codons formed from a sequence of three nucleotides (e.g. ACT, CAG, TTT).
In transcription, the codons of a gene are copied into messenger RNA by
Replication
Cell division is essential for an organism to grow, but, when a cell divides, it must replicate the DNA in its genome so that the two daughter cells have the same genetic information as their parent. The double-stranded structure of DNA provides a simple mechanism for DNA replication. Here, the two strands are separated and then each strand's complementary DNA sequence is recreated by an enzyme called DNA polymerase. This enzyme makes the complementary strand by finding the correct base through complementary base pairing and bonding it onto the original strand. As DNA polymerases can only extend a DNA strand in a 5′ to 3′ direction, different mechanisms are used to copy the antiparallel strands of the double helix.[104] In this way, the base on the old strand dictates which base appears on the new strand, and the cell ends up with a perfect copy of its DNA.
Extracellular nucleic acids
Naked extracellular DNA (eDNA), most of it released by cell death, is nearly ubiquitous in the environment. Its concentration in soil may be as high as 2 μg/L, and its concentration in natural aquatic environments may be as high at 88 μg/L.[105] Various possible functions have been proposed for eDNA: it may be involved in horizontal gene transfer;[106] it may provide nutrients;[107] and it may act as a buffer to recruit or titrate ions or antibiotics.[108] Extracellular DNA acts as a functional extracellular matrix component in the biofilms of several bacterial species. It may act as a recognition factor to regulate the attachment and dispersal of specific cell types in the biofilm;[109] it may contribute to biofilm formation;[110] and it may contribute to the biofilm's physical strength and resistance to biological stress.[111]
Cell-free fetal DNA is found in the blood of the mother, and can be sequenced to determine a great deal of information about the developing fetus.[112]
Under the name of environmental DNA eDNA has seen increased use in the natural sciences as a survey tool for ecology, monitoring the movements and presence of species in water, air, or on land, and assessing an area's biodiversity.[113][114]
Neutrophil extracellular traps
Neutrophil extracellular traps (NETs) are networks of extracellular fibers, primarily composed of DNA, which allow
Interactions with proteins
All the functions of DNA depend on interactions with proteins. These
DNA-binding proteins

Structural proteins that bind DNA are well-understood examples of non-specific DNA-protein interactions. Within chromosomes, DNA is held in complexes with structural proteins. These proteins organize the DNA into a compact structure called
A distinct group of DNA-binding proteins is the DNA-binding proteins that specifically bind single-stranded DNA. In humans, replication protein A is the best-understood member of this family and is used in processes where the double helix is separated, including DNA replication, recombination, and DNA repair.[122] These binding proteins seem to stabilize single-stranded DNA and protect it from forming stem-loops or being degraded by nucleases.

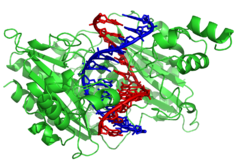
In contrast, other proteins have evolved to bind to particular DNA sequences. The most intensively studied of these are the various transcription factors, which are proteins that regulate transcription. Each transcription factor binds to one particular set of DNA sequences and activates or inhibits the transcription of genes that have these sequences close to their promoters. The transcription factors do this in two ways. Firstly, they can bind the RNA polymerase responsible for transcription, either directly or through other mediator proteins; this locates the polymerase at the promoter and allows it to begin transcription.[124] Alternatively, transcription factors can bind enzymes that modify the histones at the promoter. This changes the accessibility of the DNA template to the polymerase.[125]
As these DNA targets can occur throughout an organism's genome, changes in the activity of one type of transcription factor can affect thousands of genes.[126] Consequently, these proteins are often the targets of the signal transduction processes that control responses to environmental changes or cellular differentiation and development. The specificity of these transcription factors' interactions with DNA come from the proteins making multiple contacts to the edges of the DNA bases, allowing them to "read" the DNA sequence. Most of these base-interactions are made in the major groove, where the bases are most accessible.[24]
DNA-modifying enzymes
Nucleases and ligases
Enzymes called
Topoisomerases and helicases
Topoisomerases are enzymes with both nuclease and ligase activity. These proteins change the amount of supercoiling in DNA. Some of these enzymes work by cutting the DNA helix and allowing one section to rotate, thereby reducing its level of supercoiling; the enzyme then seals the DNA break.[43] Other types of these enzymes are capable of cutting one DNA helix and then passing a second strand of DNA through this break, before rejoining the helix.[130] Topoisomerases are required for many processes involving DNA, such as DNA replication and transcription.[44]
Helicases are proteins that are a type of molecular motor. They use the chemical energy in nucleoside triphosphates, predominantly adenosine triphosphate (ATP), to break hydrogen bonds between bases and unwind the DNA double helix into single strands.[131] These enzymes are essential for most processes where enzymes need to access the DNA bases.
Polymerases
In DNA replication, DNA-dependent DNA polymerases make copies of DNA polynucleotide chains. To preserve biological information, it is essential that the sequence of bases in each copy are precisely complementary to the sequence of bases in the template strand. Many DNA polymerases have a proofreading activity. Here, the polymerase recognizes the occasional mistakes in the synthesis reaction by the lack of base pairing between the mismatched nucleotides. If a mismatch is detected, a 3′ to 5′ exonuclease activity is activated and the incorrect base removed.[133] In most organisms, DNA polymerases function in a large complex called the replisome that contains multiple accessory subunits, such as the DNA clamp or helicases.[134]
RNA-dependent DNA polymerases are a specialized class of polymerases that copy the sequence of an RNA strand into DNA. They include
Transcription is carried out by a DNA-dependent RNA polymerase that copies the sequence of a DNA strand into RNA. To begin transcribing a gene, the RNA polymerase binds to a sequence of DNA called a promoter and separates the DNA strands. It then copies the gene sequence into a messenger RNA transcript until it reaches a region of DNA called the terminator, where it halts and detaches from the DNA. As with human DNA-dependent DNA polymerases, RNA polymerase II, the enzyme that transcribes most of the genes in the human genome, operates as part of a large protein complex with multiple regulatory and accessory subunits.[136]
Genetic recombination
|

|
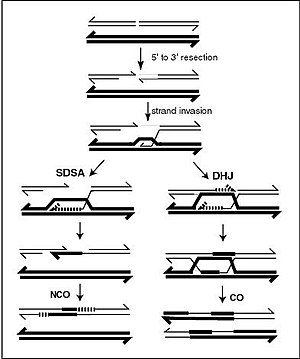
A DNA helix usually does not interact with other segments of DNA, and in human cells, the different chromosomes even occupy separate areas in the nucleus called "chromosome territories".[138] This physical separation of different chromosomes is important for the ability of DNA to function as a stable repository for information, as one of the few times chromosomes interact is in chromosomal crossover which occurs during sexual reproduction, when genetic recombination occurs. Chromosomal crossover is when two DNA helices break, swap a section and then rejoin.
Recombination allows chromosomes to exchange genetic information and produces new combinations of genes, which increases the efficiency of natural selection and can be important in the rapid evolution of new proteins.[139] Genetic recombination can also be involved in DNA repair, particularly in the cell's response to double-strand breaks.[140]
The most common form of chromosomal crossover is
Evolution
DNA contains the genetic information that allows all forms of life to function, grow and reproduce. However, it is unclear how long in the 4-billion-year
Building blocks of DNA (
In February 2021, scientists reported, for the first time, the sequencing of DNA from animal remains, a mammoth in this instance over a million years old, the oldest DNA sequenced to date.[156][157]
Uses in technology
Genetic engineering
Methods have been developed to purify DNA from organisms, such as
DNA profiling
The development of forensic science and the ability to now obtain genetic matching on minute samples of blood, skin, saliva, or hair has led to re-examining many cases. Evidence can now be uncovered that was scientifically impossible at the time of the original examination. Combined with the removal of the double jeopardy law in some places, this can allow cases to be reopened where prior trials have failed to produce sufficient evidence to convince a jury. People charged with serious crimes may be required to provide a sample of DNA for matching purposes. The most obvious defense to DNA matches obtained forensically is to claim that cross-contamination of evidence has occurred. This has resulted in meticulous strict handling procedures with new cases of serious crime.
DNA profiling is also used successfully to positively identify victims of mass casualty incidents,[167] bodies or body parts in serious accidents, and individual victims in mass war graves, via matching to family members.
DNA profiling is also used in DNA paternity testing to determine if someone is the biological parent or grandparent of a child with the probability of parentage is typically 99.99% when the alleged parent is biologically related to the child. Normal DNA sequencing methods happen after birth, but there are new methods to test paternity while a mother is still pregnant.[168]
DNA enzymes or catalytic DNA
Deoxyribozymes, also called DNAzymes or catalytic DNA, were first discovered in 1994.[169] They are mostly single stranded DNA sequences isolated from a large pool of random DNA sequences through a combinatorial approach called in vitro selection or systematic evolution of ligands by exponential enrichment (SELEX). DNAzymes catalyze variety of chemical reactions including RNA-DNA cleavage, RNA-DNA ligation, amino acids phosphorylation-dephosphorylation, carbon-carbon bond formation, etc. DNAzymes can enhance catalytic rate of chemical reactions up to 100,000,000,000-fold over the uncatalyzed reaction.[170] The most extensively studied class of DNAzymes is RNA-cleaving types which have been used to detect different metal ions and designing therapeutic agents. Several metal-specific DNAzymes have been reported including the GR-5 DNAzyme (lead-specific),[169] the CA1-3 DNAzymes (copper-specific),[171] the 39E DNAzyme (uranyl-specific) and the NaA43 DNAzyme (sodium-specific).[172] The NaA43 DNAzyme, which is reported to be more than 10,000-fold selective for sodium over other metal ions, was used to make a real-time sodium sensor in cells.
Bioinformatics
DNA nanotechnology
DNA nanotechnology uses the unique
History and anthropology
Because DNA collects mutations over time, which are then inherited, it contains historical information, and, by comparing DNA sequences, geneticists can infer the evolutionary history of organisms, their phylogeny.[183] This field of phylogenetics is a powerful tool in evolutionary biology. If DNA sequences within a species are compared, population geneticists can learn the history of particular populations. This can be used in studies ranging from ecological genetics to anthropology.
Information storage
DNA as a
History

DNA was first isolated by the Swiss physician Friedrich Miescher who, in 1869, discovered a microscopic substance in the pus of discarded surgical bandages. As it resided in the nuclei of cells, he called it "nuclein".[186][187] In 1878, Albrecht Kossel isolated the non-protein component of "nuclein", nucleic acid, and later isolated its five primary nucleobases.[188][189]
In 1909,
In 1933, while studying virgin sea urchin eggs, Jean Brachet suggested that DNA is found in the cell nucleus and that RNA is present exclusively in the cytoplasm. At the time, "yeast nucleic acid" (RNA) was thought to occur only in plants, while "thymus nucleic acid" (DNA) only in animals. The latter was thought to be a tetramer, with the function of buffering cellular pH.[198][199]
In 1937, William Astbury produced the first X-ray diffraction patterns that showed that DNA had a regular structure.[200]
In 1943,

In May 1952,
The 25 April 1953 issue of the journal Nature published a series of five articles giving the Watson and Crick double-helix structure DNA and evidence supporting it.[209] The structure was reported in a letter titled "MOLECULAR STRUCTURE OF NUCLEIC ACIDS A Structure for Deoxyribose Nucleic Acid", in which they said, "It has not escaped our notice that the specific pairing we have postulated immediately suggests a possible copying mechanism for the genetic material."[9] This letter was followed by a letter from Franklin and Gosling, which was the first publication of their own X-ray diffraction data and of their original analysis method.[47][210] Then followed a letter by Wilkins and two of his colleagues, which contained an analysis of in vivo B-DNA X-ray patterns, and which supported the presence in vivo of the Watson and Crick structure.[48]
In 1962, after Franklin's death, Watson, Crick, and Wilkins jointly received the Nobel Prize in Physiology or Medicine.[211] Nobel Prizes are awarded only to living recipients. A debate continues about who should receive credit for the discovery.[212]
In an influential presentation in 1957, Crick laid out the central dogma of molecular biology, which foretold the relationship between DNA, RNA, and proteins, and articulated the "adaptor hypothesis".[213] Final confirmation of the replication mechanism that was implied by the double-helical structure followed in 1958 through the Meselson–Stahl experiment.[214] Further work by Crick and co-workers showed that the genetic code was based on non-overlapping triplets of bases, called codons, allowing Har Gobind Khorana, Robert W. Holley, and Marshall Warren Nirenberg to decipher the genetic code.[215] These findings represent the birth of molecular biology.[216]
See also
- Autosome – Any chromosome other than a sex chromosome
- Crystallography – Scientific study of crystal structures
- DNA Day – Holiday celebrated on April 25
- DNA microarray – Collection of microscopic DNA spots attached to a solid surface
- DNA sequencing – Process of determining the nucleic acid sequence
- Genetic disorder – Health problem caused by one or more abnormalities in the genome
- Genetic genealogy – DNA testing to infer relationships
- Haplotype – Group of genes from one parent
- Meiosis – Cell division producing haploid gametes
- Nucleic acid notation – Universal notation using the Roman characters A, C, G, and T to call the four DNA nucleotides
- Nucleic acid sequence – Succession of nucleotides in a nucleic acid
- Ribosomal DNA – specific region of DNA that that codes for ribosomal RNA
- Southern blot – DNA analysis technique
- X-ray scattering techniques – family of non-destructive analytical techniques
- Xeno nucleic acid – Synthetic nucleic acid analogues
References
- ^ "deoxyribonucleic acid". Merriam-Webster.com Dictionary.
- ISBN 978-0-8153-4432-2. Archivedfrom the original on 14 July 2014.
- ^ Purcell A. "DNA". Basic Biology. Archived from the original on 5 January 2017.
- ^ "Uracil". Genome.gov. Retrieved 21 November 2019.
- ISBN 0-8053-4553-1.
- ISBN 0-387-90762-9.
- ^ from the original on 1 November 2016.
- PMID 26455586.
- ^ (PDF) from the original on 4 February 2007.
- PMID 7338906.
- ^ ISBN 0-7167-4955-6.
- PMID 5499957. Archived from the originalon 5 February 2007.
- ^ PMID 12657780.
- ^ Bank, RCSB Protein Data. "RCSB PDB - 1D65: MOLECULAR STRUCTURE OF THE B-DNA DODECAMER D(CGCAAATTTGCG)2; AN EXAMINATION OF PROPELLER TWIST AND MINOR-GROOVE WATER STRUCTURE AT 2.2 ANGSTROMS RESOLUTION". www.rcsb.org. Retrieved 27 March 2023.
- PMID 16449200.
- ISBN 978-0-7637-8663-2.
- ^ Carr S (1953). "Watson-Crick Structure of DNA". Memorial University of Newfoundland. Archived from the original on 19 July 2016. Retrieved 13 July 2016.
- PMID 9759484.
- ISSN 0002-7863.
- PMID 27319741.
- PMID 30619465.
- PMID 28941008.
- S2CID 4315465.
- ^ PMID 6236744.
- PMID 23818176.
- PMID 10733978.
- PMID 10393911.
- S2CID 24479358.
- (PDF) from the original on 10 June 2007.
- ^ PMID 30813969.)
{{cite journal}}: CS1 maint: unflagged free DOI (link - PMID 16710414.
- S2CID 4355527.
- ^ "Untitled". Archived from the original on 13 August 2011. Retrieved 13 June 2012.
- ^ PMID 1715276.
- PMID 28721182.
- ^ "Chemistry - Queen Mary University of London". www.qmul.ac.uk. Retrieved 27 March 2023.
- PMID 15851066.
- S2CID 23748148.
- PMID 15680581.
- PMID 15520290.
- PMID 1771674.
- S2CID 1427671. Archived from the original(PDF) on 1 March 2019.
- ^ S2CID 18144189.
- ^ S2CID 205496065.
- PMID 2482766.
- ^
- Franklin RE, Gosling RG (6 March 1953). "The Structure of Sodium Thymonucleate Fibres I. The Influence of Water Content" (PDF). Acta Crystallogr. 6 (8–9): 673–77. (PDF) from the original on 9 January 2016.
- Franklin RE, Gosling RG (1953). "The structure of sodium thymonucleate fibres. II. The cylindrically symmetrical Patterson function" (PDF). Acta Crystallogr. 6 (8–9): 678–85. (PDF) from the original on 29 June 2017.
- ^ (PDF) from the original on 3 January 2011.
- ^ (PDF) from the original on 13 May 2011.
- PMID 7441761.
- S2CID 189888972.
- ^ Hosemann R, Bagchi RN (1962). Direct analysis of diffraction by matter. Amsterdam – New York: North-Holland Publishers.
- doi:10.1107/S0567739478001540. Archived from the original(PDF) on 14 March 2020. Retrieved 29 August 2019.
- PMID 9097733.
- PMID 10891271.
- S2CID 20589136.
- PMID 12486233.
- ^ Palmer J (2 December 2010). "Arsenic-loving bacteria may help in hunt for alien life". BBC News. Archived from the original on 3 December 2010. Retrieved 2 December 2010.
- ^ a b Bortman H (2 December 2010). "Arsenic-Eating Bacteria Opens New Possibilities for Alien Life". Space.com. Archived from the original on 4 December 2010. Retrieved 2 December 2010.
- from the original on 12 February 2012.
- S2CID 87341731.
- ^ "Structure Summary for 1KF1". ndbserver.rutgers.edu. Retrieved 27 March 2023.
- ^ PMID 3907856.
- ^ PMID 9553037.
- PMID 9353250.
- ^ PMID 17012276.
- S2CID 4422211.
- S2CID 721901.
- PMID 16515737.
- PMID 30809059.
- PMID 30792304.
- S2CID 17574854.
- ^ Reusch, William. "Nucleic Acids". Michigan State University. Retrieved 30 June 2022.
- ^ "How To Extract DNA From Anything Living". University of Utah. Retrieved 30 June 2022.
- PMID 23133442.
- PMID 16403636.
- PMID 11782440.
- PMID 16570853.
- PMID 19372393.
- PMID 16479578.
- S2CID 24801094.
- ^ Created from PDB 1JDG Archived 22 September 2008 at the Wayback Machine
- PMID 12885257.
- PMID 10064846.
- PMID 9289489.
- PMID 12947387.
- ^ Johnson G (28 December 2010). "Unearthing Prehistoric Tumors, and Debate". The New York Times. Archived from the original on 24 June 2017.
If we lived long enough, sooner or later we all would get cancer.
- ISBN 0-8153-4072-9. Archivedfrom the original on 2 January 2016.
A certain irreducible background incidence of cancer is to be expected regardless of circumstances: mutations can never be absolutely avoided, because they are an inescapable consequence of fundamental limitations on the accuracy of DNA replication, as discussed in Chapter 5. If a human could live long enough, it is inevitable that at least one of his or her cells would eventually accumulate a set of mutations sufficient for cancer to develop.
- ISBN 978-1-60456-581-2. Archivedfrom the original on 25 October 2014.
- PMID 19812404.
- PMID 21600302.
- PMID 1881402.
- PMID 10799645.
- PMID 3936066.
- PMID 11562309.
- PMID 11181995.
- PMID 15988757.
- PMID 11236998.
- PMID 15596463.
- PMID 17571346.
- ^ Bank, RCSB Protein Data. "RCSB PDB - 1MSW: Structural basis for the transition from initiation to elongation transcription in T7 RNA polymerase". www.rcsb.org. Retrieved 27 March 2023.
- PMID 15905142.
- PMID 11827946.
- PMID 12083509.
- PMID 11178285.)
{{cite journal}}: CS1 maint: unflagged free DOI (link - ISBN 978-3-642-12616-1.
- S2CID 32463239.
- PMID 11591672.
- PMID 19023416.)
{{cite journal}}: CS1 maint: unflagged free DOI (link - PMID 20598083.
- PMID 11859186.
- PMID 23300576.
- PMID 23158400.
- PMID 22952587.
- ^ "Researchers Detect Land Animals Using DNA in Nearby Water Bodies".
- S2CID 21101836.
- S2CID 26965112.
- S2CID 4328827.
- (PDF) from the original on 8 August 2017.
- PMID 12596902.
- PMID 11497996.
- PMID 8178371.
- PMID 10473346.
- ^ Bank, RCSB Protein Data. "RCSB PDB - 1LMB: REFINED 1.8 ANGSTROM CRYSTAL STRUCTURE OF THE LAMBDA REPRESSOR-OPERATOR COMPLEX". www.rcsb.org. Retrieved 27 March 2023.
- PMID 10966474.
- PMID 15479634.
- PMID 12808131.
- ^ Bank, RCSB Protein Data. "RCSB PDB - 1RVA: MG2+ BINDING TO THE ACTIVE SITE OF ECO RV ENDONUCLEASE: A CRYSTALLOGRAPHIC STUDY OF COMPLEXES WITH SUBSTRATE AND PRODUCT DNA AT 2 ANGSTROMS RESOLUTION". www.rcsb.org. Retrieved 27 March 2023.
- PMID 8336674.
- ^ PMID 11058099.
- PMID 16246147.
- PMID 15128295.
- PMID 7592405.
- S2CID 26171993. Archived from the original(PDF) on 26 January 2021.
- PMID 15952889.
- ^ S2CID 39614573.)
{{cite journal}}: CS1 maint: unflagged free DOI (link - S2CID 24946189.
- ^ Bank, RCSB Protein Data. "RCSB PDB - 1M6G: Structural Characterisation of the Holliday Junction TCGGTACCGA". www.rcsb.org. Retrieved 27 March 2023.
- S2CID 8547149.
- S2CID 23225873.
- S2CID 7779574.
- PMID 9466696.
- PMID 16838012.
- S2CID 39505263.
- S2CID 4331004.
- PMID 15217990.
- S2CID 85976762.
- PMID 1372984.
- S2CID 4283694.
- S2CID 9879073.
- PMID 15866038.
- S2CID 24740859.
- PMID 21836052.
- ^ Steigerwald J (8 August 2011). "NASA Researchers: DNA Building Blocks Can Be Made in Space". NASA. Archived from the original on 23 June 2015. Retrieved 10 August 2011.
- ^ ScienceDaily Staff (9 August 2011). "DNA Building Blocks Can Be Made in Space, NASA Evidence Suggests". ScienceDaily. Archived from the original on 5 September 2011. Retrieved 9 August 2011.
- ^ Marlaire R (3 March 2015). "NASA Ames Reproduces the Building Blocks of Life in Laboratory". NASA. Archived from the original on 5 March 2015. Retrieved 5 March 2015.
- ^ Hunt, Katie (17 February 2021). "World's oldest DNA sequenced from a mammoth that lived more than a million years ago". CNN News. Retrieved 17 February 2021.
- PMID 33597786.
- S2CID 41788896.
- PMID 17172731.
- PMID 11950565.
- PMID 12595138.
- ^ Curtis C, Hereward J (29 August 2017). "From the crime scene to the courtroom: the journey of a DNA sample". The Conversation. Archived from the original on 22 October 2017. Retrieved 22 October 2017.
- PMID 8016106.
- S2CID 14511630.
- S2CID 4229883.
- ^ "Colin Pitchfork". web.archive.org. 14 December 2006. Retrieved 27 March 2023.
- ^ "DNA Identification in Mass Fatality Incidents". National Institute of Justice. September 2006. Archived from the original on 12 November 2006.
- ISSN 0362-4331. Retrieved 27 March 2023.
- ^ PMID 9383394.
- PMID 19684594.
- PMID 9000012.
- PMID 25918425.
- OCLC 45951728.
- ISBN 978-0-521-58519-4.
- PMID 14734307.
- OCLC 55106399.
- S2CID 13222080.)
{{cite journal}}: CS1 maint: unflagged free DOI (link - S2CID 4316391.
- S2CID 4430815.
- PMID 19421208.
- S2CID 2755388.
- . Retrieved 30 June 2022.
- PMID 11806830.)
{{cite journal}}: CS1 maint: unflagged free DOI (link - PMID 29744271.
- S2CID 51905843.
- ^ Miescher F (1871). "Ueber die chemische Zusammensetzung der Eiterzellen" [On the chemical composition of pus cells]. Medicinisch-chemische Untersuchungen (in German). 4: 441–60.
[p. 456] Ich habe mich daher später mit meinen Versuchen an die ganzen Kerne gehalten, die Trennung der Körper, die ich einstweilen ohne weiteres Präjudiz als lösliches und unlösliches Nuclein bezeichnen will, einem günstigeren Material überlassend. (Therefore, in my experiments I subsequently limited myself to the whole nucleus, leaving to a more favorable material the separation of the substances, that for the present, without further prejudice, I will designate as soluble and insoluble nuclear material ("Nuclein"))
- S2CID 915930.
- ^ See:
- Kossel A (1879). "Ueber Nucleïn der Hefe" [On nuclein in yeast]. Zeitschrift für physiologische Chemie (in German). 3: 284–91.
- Kossel A (1880). "Ueber Nucleïn der Hefe II" [On nuclein in yeast, Part 2]. Zeitschrift für physiologische Chemie (in German). 4: 290–95.
- Kossel A (1881). "Ueber die Verbreitung des Hypoxanthins im Thier- und Pflanzenreich" [On the distribution of hypoxanthins in the animal and plant kingdoms]. Zeitschrift für physiologische Chemie (in German). 5: 267–71.
- Kossel A (1881). Trübne KJ (ed.). Untersuchungen über die Nucleine und ihre Spaltungsprodukte [Investigations into nuclein and its cleavage products] (in German). Strassburg, Germany. p. 19.
{{cite book}}: CS1 maint: location missing publisher (link) - Kossel A (1882). "Ueber Xanthin und Hypoxanthin" [On xanthin and hypoxanthin]. Zeitschrift für physiologische Chemie. 6: 422–31.
- Albrect Kossel (1883) "Zur Chemie des Zellkerns" Archived 17 November 2017 at the Wayback Machine (On the chemistry of the cell nucleus), Zeitschrift für physiologische Chemie, 7 : 7–22.
- Kossel A (1886). "Weitere Beiträge zur Chemie des Zellkerns" [Further contributions to the chemistry of the cell nucleus]. Zeitschrift für Physiologische Chemie (in German). 10: 248–64.
On p. 264, Kossel remarked presciently: Der Erforschung der quantitativen Verhältnisse der vier stickstoffreichen Basen, der Abhängigkeit ihrer Menge von den physiologischen Zuständen der Zelle, verspricht wichtige Aufschlüsse über die elementaren physiologisch-chemischen Vorgänge. (The study of the quantitative relations of the four nitrogenous bases—[and] of the dependence of their quantity on the physiological states of the cell—promises important insights into the fundamental physiological-chemical processes.)
- PMID 13103145.
- .
- .
- .
- PMID 4609088.
- ^ Koltsov proposed that a cell's genetic information was encoded in a long chain of amino acids. See:
- Кольцов, Н. К. (12 December 1927). Физико-химические основы морфологии [The physical-chemical basis of morphology] (Speech). 3rd All-Union Meeting of Zoologist, Anatomists, and Histologists (in Russian). Leningrad, U.S.S.R.
- Reprinted in: Кольцов, Н. К. (1928). "Физико-химические основы морфологии" [The physical-chemical basis of morphology]. Успехи экспериментальной биологии (Advances in Experimental Biology) series B (in Russian). 7 (1): ?.
- Reprinted in German as: Koltzoff NK (1928). "Physikalisch-chemische Grundlagen der Morphologie" [The physical-chemical basis of morphology]. Biologisches Zentralblatt (in German). 48 (6): 345–69.
- In 1934, Koltsov contended that the proteins that contain a cell's genetic information replicate. See: Koltzoff N (October 1934). "The structure of the chromosomes in the salivary glands of Drosophila". Science. 80 (2075): 312–13. PMID 17769043.
From page 313: "I think that the size of the chromosomes in the salivary glands [of Drosophila] is determined through the multiplication of genonemes. By this term I designate the axial thread of the chromosome, in which the geneticists locate the linear combination of genes; … In the normal chromosome there is usually only one genoneme; before cell-division this genoneme has become divided into two strands."
- S2CID 46277758.
- PMID 20474956.
- PMID 7968924.
- ^ Brachet J (1933). "Recherches sur la synthese de l'acide thymonucleique pendant le developpement de l'oeuf d'Oursin". Archives de Biologie (in Italian). 44: 519–76.
- ^ Burian R (1994). "Jean Brachet's Cytochemical Embryology: Connections with the Renovation of Biology in France?" (PDF). In Debru C, Gayon J, Picard JF (eds.). Les sciences biologiques et médicales en France 1920–1950. Cahiers pour I'histoire de la recherche. Vol. 2. Paris: CNRS Editions. pp. 207–20.
- ^ See:
- Astbury WT, Bell FO (1938). "Some recent developments in the X-ray study of proteins and related structures" (PDF). Cold Spring Harbor Symposia on Quantitative Biology. 6: 109–21. doi:10.1101/sqb.1938.006.01.013. Archived from the original(PDF) on 14 July 2014.
- Astbury WT (1947). "X-ray studies of nucleic acids". Symposia of the Society for Experimental Biology (1): 66–76. PMID 20257017. Archived from the originalon 5 July 2014.
- Astbury WT, Bell FO (1938). "Some recent developments in the X-ray study of proteins and related structures" (PDF). Cold Spring Harbor Symposia on Quantitative Biology. 6: 109–21.
- PMID 19871359.
- S2CID 2522535.
- .
- PMID 12981234.
- ^ "Linus Pauling and the Race for DNA: A Documentary History - Special Collections & Archives Research Center - Oregon State University". scarc.library.oregonstate.edu. Retrieved 27 March 2023.
- ISBN 978-0-674-02670-4.
- PMID 16578429.
- ISBN 978-0-19-538341-6.
- ^ "Double Helix of DNA: 50 Years". Nature Archives. Archived from the original on 5 April 2015.
- ^ "Original X-ray diffraction image". Oregon State Library. Archived from the original on 30 January 2009. Retrieved 6 February 2011.
- ^ "The Nobel Prize in Physiology or Medicine 1962". Nobelprize.org.
- (PDF) from the original on 17 October 2016.
- ^ Crick F (1955). A Note for the RNA Tie Club (PDF) (Speech). Cambridge, England. Archived from the original (PDF) on 1 October 2008.
- PMID 16590258.
- ^ "The Nobel Prize in Physiology or Medicine 1968". Nobelprize.org.
- ^ Pray L (2008). "Discovery of DNA structure and function: Watson and Crick". Nature Education. 1 (1): 100.
Further reading
- Berry A, ISBN 0-375-41546-7.
- Calladine CR, Drew HR, Luisi BF, Travers AA (2003). Understanding DNA: the molecule & how it works. Amsterdam: Elsevier Academic Press. ISBN 0-12-155089-3.
- Carina D, Clayton J (2003). 50 years of DNA. Basingstoke: Palgrave Macmillan. ISBN 1-4039-1479-6.
- ISBN 0-671-22540-5.
- ISBN 0-486-68117-3. First published in October 1974 by MacMillan, with foreword by Francis Crick; the definitive DNA textbook, revised in 1994 with a nine-page postscript.
- PMID 12540907.
- Olby RC (2009). Francis Crick: A Biography. Plainview, N.Y: Cold Spring Harbor Laboratory Press. ISBN 978-0-87969-798-3.
- Micklas D (2003). DNA Science: A First Course. Cold Spring Harbor Press. ISBN 978-0-87969-636-8.
- ISBN 0-06-082333-X.
- Rosenfeld I (2010). DNA: A Graphic Guide to the Molecule that Shook the World. Columbia University Press. ISBN 978-0-231-14271-7.
- Schultz M, Cannon Z (2009). The Stuff of Life: A Graphic Guide to Genetics and DNA. Hill and Wang. ISBN 978-0-8090-8947-5.
- ISBN 0-393-95075-1.
- Watson J (2004). DNA: The Secret of Life. Random House. ISBN 978-0-09-945184-6.
- ISBN 0-19-860665-6.
External links
- DNA at Curlie
- DNA binding site prediction on protein
- DNA the Double Helix Game From the official Nobel Prize web site
- DNA under electron microscope
- Dolan DNA Learning Center
- Double Helix: 50 years of DNA, Nature
- Proteopedia DNA
- Proteopedia Forms_of_DNA
- ENCODE threads explorer ENCODE home page at Nature
- Double Helix 1953–2003 National Centre for Biotechnology Education
- Genetic Education Modules for Teachers – DNA from the Beginning Study Guide
- PDB Molecule of the Month DNA
- "Clue to chemistry of heredity found". The New York Times, June 1953. First American newspaper coverage of the discovery of the DNA structure
- DNA from the Beginning Another DNA Learning Center site on DNA, genes, and heredity from Mendel to the human genome project.
- The Register of Francis Crick Personal Papers 1938 – 2007 at Mandeville Special Collections Library, University of California, San Diego
- Seven-page, handwritten letter that Crick sent to his 12-year-old son Michael in 1953 describing the structure of DNA. See Crick's medal goes under the hammer, Nature, 5 April 2013.


