Peripherally acting μ-opioid receptor antagonist
Peripherally acting
Discovery and development
Opioid drugs are known to cause
OIC is one of the most common
In the late 1970s, Dennis M. Zimmerman and his co-workers from
Mechanism of action
PAMORAs act by inhibiting the binding of opioids
PAMORAs effect on gut motility is that it can increase the resting tone in the circular muscle layer. The antagonist enhances the effect on tonic inhibition of the
PAMORAs effect on gut secretion will help reverse the decreased
PAMORAs effect on the function of the
Structure–activity relationship
Even though μ-opioid receptor (MOR) targeting drugs have been used for a long time, not much is known about the
Structure

Methylnaltrexone bromide, naloxegol, and naldemedine all have similar structures, which is not far away from the chemical structure of
Binding site
Agonists and antagonists form certain
It can be concluded that interactions with the amino acid residues, Asp147 and Tyr148 are essential for the ligand to bind to the receptor and the molecules that form additional polar interactions with other residues are more often antagonists than agonists.[17]
The N-substituent group can form
Methylnaltrexone bromide
Methylnaltrexone bromide is the

Alvimopan

Peripherally selective trans-3,4-dimethyl-4-(3-hydroxylphenyl)piperidine opioid antagonists were developed for the treatment of
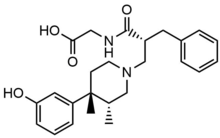
Naloxegol
Naloxegol is a
Naldemedine
Naldemedine has a similar chemical structure as naltrexone but with an additional side chain that increases the
Pharmacokinetics
The
| Chemical name | Chemical structure | Molecular weight (g/mol) | Bioavailability (%) | Plasma protein binding (%) | t1/2 (h) | tmax | Ki μ (nM) | Ki κ (nM) | Ki δ (nM) |
|---|---|---|---|---|---|---|---|---|---|
| Methylnaltrexone bromide | 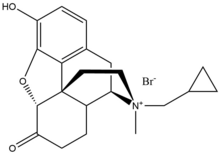 |
436,3 | Low | 11-15 | 8 | 30 min | 5.50 | 32.1 | 3453.8 |
| Alvimopan | 424,53 | 6 | 80-90 | 10-17 | 2 h | 0.77 | 40 | 4.4 | |
| Naloxegol | 651,798 | NA | 4,2 | 6-11 | 2 h | 7.42 | 8.65 | 203.0 | |
| Naldemedine | 570,6 | 29 | 93-94 | 11 | 45 min | 0.34 | 0.94 | 0.43 |
- t1/2: Biological half-life
- tmax: Time to achieve maximum plasma concentration
- pKi: the measurement of ligand bindingaffinity
Methylnaltrexone bromide has poor oral bioavailability, and for that reason, every other day it is administered
Alvimopan has considerable low bioavailability (6%) due to its high
When naloxegol is given with a fatty meal,
Naldemedine metabolites mainly via
PAMORAs in development

There is a demand for optimization of the receptor selectivity and affinity accompanied by an exploration of candidate compounds regarding their route of administration. These are the main objectives and future strategies for drug discovery and the development of PAMORAs. Predominantly, the MORs exhibit functionally selective agonism. Therefore, future possible candidate compounds that target OIC are PAMORAs with optimized selectivity and affinity.[27]
References
- PMID 28336575.
- PMID 29630236. Retrieved 4 June 2019.
- S2CID 3458268.
- ^ "Drug developed at the University of Chicago wins FDA approval". University of Chicago News. 30 April 2008. Retrieved 30 April 2008.
- ^ PMID 24981721.
- S2CID 4149532.
- ^ "Drug Approval Package: Entereg (Alvinopan) Capsules 21775". www.accessdata.fda.gov. FDA. Retrieved 18 July 2008.
- PMID 30340754.
- ^ "Drug Approval Package: MOVANTIC (naloxegol) Tablets". www.accessdata.fda.gov. FDA.
- ^ "Symproic (naldemedine) Tablets". www.accessdata.fda.gov. FDA. Retrieved 4 May 2017.
- PMID 28946783.
- S2CID 173168.
- ^ PMID 17981003.
- PMID 26811699.
- PMID 19829093.
- S2CID 173168.
- ^ PMID 26888328.
- ^ DeRuiter, Jack (2000). Principles of Drug Action (PDF). Auburn Education.
- PMID 24919067.
- PMID 28314512.
- ISBN 9780444804020.
- ^ PMID 22437502.
- ^ a b "Methylnaltrexone bromide". pubchem.ncbi.nlm.nih.gov.
- ^ ISBN 9780123809025.
- ^ S2CID 81387334.
- ^ a b "Naloxegol". pubchem.ncbi.nlm.nih.gov.
- ^ PMID 28946783.
- PMID 30271103.
- PMID 30446313.
- ^ a b Turan, Alparslan; Saasouh, Wael; Hovsepyan, Karen; You, Jing. "Ancillary Effects of Oral Naloxegol (Movantik)" (PDF). Clinicaltrials.gov.
- ^ "Alvimopan (ADL 8-2698) | Opioid Receptor Antagonist | MedChemExpress". MedchemExpress.com.
- ^ "Methylnaltrexone bromide". pubchem.ncbi.nlm.nih.gov.
- PMID 30821019.
- ^ "Opioid Receptor | kappa, mu Opioid Receptor". www.selleckchem.com. Retrieved 2019-10-10.
- S2CID 19271743.
- PMID 30288274.
- ^ "Theravance Biopharma: Programs | Gastrointestinal Motility Dysfunction". SITE.