Last universal common ancestor
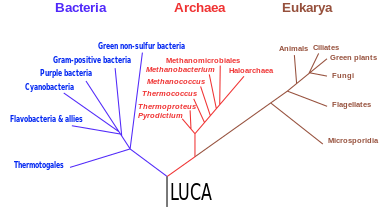
The last universal common ancestor (LUCA) is the hypothesized common ancestral
All earlier forms of life preceding this divergence and all extant organisms are generally thought to share
The genesis of viruses, before or after the LUCA as well as the diversity of extant viruses and their hosts are subjects of investigation.
While no fossil evidence of the LUCA exists, the detailed biochemical similarity of all current life (divided into the three domains) makes it plausible. Its characteristics can be inferred from
Historical background

A phylogenetic tree directly portrays the idea of evolution by descent from a single ancestor.[4] An early tree of life was sketched by Jean-Baptiste Lamarck in his Philosophie zoologique in 1809.[5][6] Charles Darwin more famously proposed the theory of universal common descent through an evolutionary process in his book On the Origin of Species in 1859: "Therefore I should infer from analogy that probably all the organic beings which have ever lived on this earth have descended from some one primordial form, into which life was first breathed."[7] The last sentence of the book begins with a restatement of the hypothesis:
There is grandeur in this view of life, with its several powers, having been originally breathed into a few forms or into one ...
— [7]
The term "last universal common ancestor" or "LUCA" was first used in the 1990s for such a primordial organism.[8][9][10]
Inferring LUCA's features
An anaerobic thermophile
In 2016, Madeline C. Weiss and colleagues genetically analyzed 6.1 million protein-coding genes and 286,514 protein clusters from sequenced
While the gross anatomy of the LUCA can only be reconstructed with much uncertainty, its biochemical mechanisms can be described in some detail, based on the "universal" properties currently shared by all independently living organisms on Earth.[16]
The LUCA certainly had
LUCA was likely capable of sexual interaction in the sense that adaptive gene functions were present that promoted the transfer of DNA between individuals of the population to facilitate genetic recombination. Homologous gene products that promote genetic recombination are present in bacteria, archaea and eukaryota, such as the RecA protein in bacteria, the RadA protein in archaea, and the Rad51 and Dmc1 proteins in eukaryota.[24]
The functionality of LUCA as well as evidence for the early evolution membrane-dependent biological systems together suggest that LUCA had cellularity and cell membranes.
By phylogenetic bracketing, analysis of the presumed LUCA's offspring groups, LUCA appears to have been a small, single-celled organism. It likely had a ring-shaped coil of DNA floating freely within the cell. Morphologically, it would likely not have stood out within a mixed population of small modern-day bacteria. The originator of the three-domain system, Carl Woese, stated that in its genetic machinery, the LUCA would have been a "simpler, more rudimentary entity than the individual ancestors that spawned the three [domains] (and their descendants)".[1]
An alternative to the search for "universal" traits is to use genome analysis to identify phylogenetically ancient genes. This gives a picture of a LUCA that could live in a geochemically harsh environment and is like modern prokaryotes. Analysis of biochemical pathways implies the same sort of chemistry as does phylogenetic analysis. Weiss and colleagues write that "Experiments ... demonstrate that ...
An experiment shows that Zn2+, Cr3+, and Fe can promote 6 of the 11 reactions of an ancient anabolic pathway called the reverse Krebs cycle in acidic conditions which implies that LUCA might have inhabited either hydrothermal vents or acidic metal-rich hydrothermal fields.[30]
Because both bacteria and archaea have differences in the structure of phospholipids and cell wall, ion pumping, most proteins involved in DNA replication, and glycolysis, it is inferred that LUCA had a permeable membrane without an ion pump. The emergence of Na+/H+ antiporters likely lead to the evolution of impermeable membranes present in eukaryotes, archaea, and bacteria. It is stated that "The late and independent evolution of glycolysis but not gluconeogenesis is entirely consistent with LUCA being powered by natural proton gradients across leaky membranes. Several discordant traits are likely to be linked to the late evolution of cell membranes, notably the cell wall, whose synthesis depends on the membrane and DNA replication".
UV light between 200-280 nm (at the time, unprotected by the ozone layer) would have been damaging to nucleotides at the surface, as it can cause mutations or transcription errors and ultimately damaging consequences for organisms at the cellular level. However, it is likely that LUCA existed in a UV environment because of the prevalence of photolyase across the tree of life. Photolyase uses UV to drive photoreactivation, a mechanism that works to repair damage from radiation, caused by UV.
Alternative interpretations
Some other researchers have challenged Weiss et al.'s 2016 conclusions. Sarah Berkemer and Shawn McGlynn argue that Weiss et al. undersampled the families of proteins, so that the phylogenetic trees were not complete and failed to describe the evolution of proteins correctly. There are two risks in attempting to attribute LUCA's environment from near-universal gene distribution (as in Weiss et al. 2016). On the one hand, it risks misattributing convergence or horizontal gene transfer events to vertical descent; on the other hand, it risks misattributing potential LUCA gene families as horizontal gene transfer events.
A phylogenomic and geochemical analysis of a set of proteins that probably traced to the LUCA show that it had K+-dependent GTPases and the ionic composition and concentration of its intracellular fluid was seemingly high K+/Na+ ratio, NH+
4, Fe2+, CO2+, Ni2+, Mg2+, Mn2+, Zn2+, pyrophosphate, and PO3−
4 which would imply a terrestrial hot spring habitat. It possibly had a phosphate-based metabolism. Further, these proteins were unrelated to autotrophy (the ability of an organism to create its own organic matter), suggesting that the LUCA had a heterotrophic lifestyle (consuming organic matter) and that its growth was dependent on organic matter produced by the physical environment.[34] Nick Lane argues that Na+/H+ antiporters could readily explain the low concentration of Na+ in the LUCA and its descendants.
The presence of the energy-handling enzymes
Evidence that LUCA was mesophilic
Several lines of evidence now suggest that LUCA was non-thermophilic.
The content of G + C nucleotide pairs (compared to the occurrence of A + T pairs) can indicate an organism's thermal optimum as they are more thermally stable due to an additional hydrogen bond. As a result they occur more frequently in the rRNA of thermophiles; however this is not seen in LUCA's reconstructed rRNA.[37][38][15]
The identification of thermophilic genes in the LUCA has been criticized,[39] as they may instead represent genes that evolved later in archaea or bacteria, then migrated between these via horizontal gene transfer, as in Woese's 1998 hypothesis.[40] LUCA could have been a mesophile that fixed CO2 and relied on H2, and lived close to hydrothermal vents.[41]
Further evidence that LUCA was mesophilic comes from the amino acid composition of its proteins. The abundance of I, V, Y, W, R, E, and L amino acids (denoted IVYWREL) in an organism's proteins is correlated with its optimal growth temperature.[42] According to phylogentic analysis, the IVYWREL content of LUCA's proteins suggests its ideal temperature was below 50°C.[15]
Finally, evidence that bacteria and archaea both independently underwent phases of increased and subsequently decreased thermo-tolerance suggests a dramatic post-LUCA climate shift that affected both populations and would explain the seeming genetic pervasiveness of thermo-tolerant genetics.[43]
Age
Studies from 2000 to 2018 have suggested an increasingly ancient time for the LUCA. In 2000, estimates of the LUCA's age ranged from 3.5 to 3.8 billion years ago in the Paleoarchean,[44] a few hundred million years before the earliest fossil evidence of life, for which candidates range in age from 3.48 to 4.28 billion years ago.[45][46][47][48][49] This placed the origin of the first forms of life shortly after the Late Heavy Bombardment which was thought to have repeatedly sterilized Earth's surface. However, a 2018 study by Holly Betts and colleagues applied a molecular clock model to the genomic and fossil record (102 species, 29 common protein-coding genes, mostly ribosomal), concluding that LUCA preceded the Late Heavy Bombardment. They assumed that there was no sterilizing event after the Moon-forming event, which they dated as 4.520 billion years ago, and concluded that the most likely date for LUCA was within 50 million years of that.[50]
Root of the tree of life

In 1990, a novel concept of the
In the meantime, numerous modifications of this tree, mainly concerning the role and importance of horizontal gene transfer for its rooting and early ramifications have been suggested (e.g.[55][51]). Since heredity occurs both vertically and horizontally, the tree of life may have been more weblike or netlike in its early phase and more treelike when it grew three-stemmed.[51] Presumably horizontal gene transfer has decreased with growing cell stability.[3]
A modified version of the tree, based on several molecular studies, has its root between a
The nature of LUCA remains disputed. In 1994, on the basis of primordial metabolism (sensu Wächtershäuser), Otto Kandler proposed a successive divergence of the three domains of life[1] from a multiphenotypical population of pre-cells, reached by gradual evolutionary improvements (cellularization).[62][63][64] These phenotypically diverse pre-cells were metabolising, self-reproducing entities exhibiting frequent mutual exchange of genetic information. Thus, in this scenario there was no "first cell". It may explain the unity and, at the same time, the partition into three lines (the three domains) of life. Kandler's pre-cell theory is supported by Wächtershäuser.[65][66] In 1998, Carl Woese, based on the RNA world concept, proposed that no individual organism could be considered a LUCA, and that the genetic heritage of all modern organisms derived through horizontal gene transfer among an ancient community of organisms.[67] Other authors concur that there was a "complex collective genome"[68] at the time of the LUCA, and that horizontal gene transfer was important in the evolution of later groups;[68] Nicolas Glansdorff states that LUCA "was in a metabolically and morphologically heterogeneous community, constantly shuffling around genetic material" and "remained an evolutionary entity, though loosely defined and constantly changing, as long as this promiscuity lasted."[69]
The theory of a universal common ancestry of life is widely accepted. In 2010, based on "the vast array of molecular sequences now available from all domains of life,"[70] D. L. Theobald published a "formal test" of universal common ancestry (UCA). This deals with the common descent of all extant terrestrial organisms, each being a genealogical descendant of a single species from the distant past. His formal test favoured the existence of a universal common ancestry over a wide class of alternative hypotheses that included horizontal gene transfer. Basic biochemical principles imply that all organisms do have a common ancestry.[71]
A proposed, earlier, non-cellular ancestor to LUCA is the First universal common ancestor (FUCA).[72][73] FUCA would therefore be the ancestor to every modern cell as well as ancient, now-extinct cellular lineages not descendant of LUCA. FUCA is assumed to have had other descendants than LUCA, none of which have modern descendants. Some genes of these ancient now-extinct cell lineages are thought to have been horizontally transferred into the genome of early descendants of LUCA.[61]
LUCA and viruses
The
Based on how viruses are currently distributed across the
LUCA might have been the ancestor to some viruses, as it might have had at least two descendants: LUCELLA, the Last Universal Cellular Ancestor, the ancestor to all cells, and the archaic virocell ancestor, the ancestor to large-to-medium-sized DNA viruses.[77] Viruses might have evolved before LUCA but after the First universal common ancestor (FUCA), according to the reduction hypothesis, where giant viruses evolved from primordial cells that became parasitic.[61]
See also
- Abiogenesis – Life arising from non-living matter
- Cellularization – Scientific theory to explain the origin and formation of cells
- Chemoton – Abstract model for the fundamental unit of life
- Darwinian threshold – Period during the evolution of the first cells
- Last eukaryotic common ancestor– Process of forming the first eukaryotic cell
- Mitochondrial Eve – Matrilineal most recent common ancestor of all living humans
- Pre-cell – Hypothetical life before complete cells
- Proto-metabolism
- Timeline of the evolutionary history of life
- Urmetazoan – Hypothetical last common ancestor of all animals
- Y-chromosomal Adam – Patrilineal most recent common ancestor of all living humans
Notes
- ^ Other studies propose that LUCA may have been defined wholly through RNA,[18] consisted of a RNA-DNA hybrid genome, or possessed a retrovirus-like genetic cycle with DNA serving as a stable genetic repository.[19]
- ^ One debate dealt with a former cladistic hypothesis: The tree could not be ascribed a root in the usual algorithmic way, because that would require an outgroup for reference. In the case of the universal tree, no outgroup would exist. The cladistic method was used "to root the purple bacteria, for example. But establishing a root for the universal tree of life, the branching order among the primary urkingdoms, was another matter entirely."[54]
References
- ^ PMID 2112744.
- ^ ISBN 978-1-292-40479-0.
- ^ ISBN 978-0-226-17428-0. Archivedfrom the original on 31 October 2023. Retrieved 12 October 2023.
- S2CID 15488906.
- ^ Lamarck, Jean Baptiste Pierre Antoine de Monet de (1994) [1809]. Philosophie zoologique (PDF). Paris. p. 737.
{{cite book}}: CS1 maint: location missing publisher (link) - from the original on 1 March 2022. Retrieved 23 December 2022.
- ^ a b Darwin, Charles (1859). The Origin of Species by Means of Natural Selection. John Murray. pp. 484, 490. Archived from the original on 8 October 2022. Retrieved 8 October 2022.
- ProQuest 304192982
- PMID 9468785.
- OCLC 55642057.
- ^ S2CID 52019935.
- ^ (PDF) from the original on 18 April 2022. Retrieved 10 October 2022.
- PMID 35868450.
- PMID 30538225.
- ^ from the original on 23 February 2024. Retrieved 4 December 2023.
- .
- ^ .
- ^ Marshall, Michael. "Life began with a planetary mega-organism". New Scientist. Archived from the original on 25 July 2016. Retrieved 31 July 2016.
- PMID 16223546.
- ^ Garwood, Russell J. (2012). "Patterns In Palaeontology: The first 3 billion years of evolution". Palaeontology Online. 2 (11): 1–14. Archived from the original on 26 June 2015. Retrieved 25 June 2015.
- PMID 32517760.
- PMID 28534707.
- PMID 30520969.
- ^ Bernstein, H., Bernstein, C. (2017). Sexual Communication in Archaea, the Precursor to Eukaryotic Meiosis. In: Witzany, G. (eds) Biocommunication of Archaea. Springer, Cham. https://doi.org/10.1007/978-3-319-65536-9_7 Archived 23 February 2024 at the Wayback Machine
- from the original on 23 February 2024. Retrieved 4 December 2023.
- .
- PMID 17255002.
- PMID 20108228.
- S2CID 52019935.
- PMID 28970480.
- PMID 25116890.
- S2CID 52019935.
- license.
- PMID 22331915.
- PMID 29358391.
- S2CID 52019935.
- from the original on 23 February 2024. Retrieved 4 December 2023.
- PMID 24046876.
- from the original on 3 April 2020. Retrieved 25 June 2019.
- PMID 9618502.
- ISSN 1572-9672.
- PMID 17222055.
- from the original on 23 February 2024. Retrieved 4 December 2023.
- PMID 10710791.
- PMID 24205812.
- doi:10.1038/ngeo2025.
- S2CID 205257931.
- PMID 26483481.
- (PDF) from the original on 23 July 2018. Retrieved 25 June 2019.
- PMID 30127539.
- ^ S2CID 2265315.
- ^ ISBN 978-0-199-73438-2. Archivedfrom the original on 6 November 2023. Retrieved 21 November 2023.
- ISBN 978-1-292-01831-7.
- ^ Sapp 2009, p. 255.
- ^ PMID 7708661.
- PMID 21356104.
- PMID 16834776.
- PMID 25964353.
- S2CID 3833474.
- from the original on 1 October 2022. Retrieved 23 September 2022.
- ^ PMID 33519747.
- ^ Kandler, Otto (1994). "The early diversification of life". In Stefan Bengtson (ed.). Early Life on Earth. Nobel Symposium 84. New York: Columbia University Press. pp. 152–160.
- S2CID 83906865.
- ISBN 978-0-203-48420-3. Archivedfrom the original on 25 February 2023. Retrieved 21 June 2023.
- S2CID 37944519.
- PMID 17008219.
- PMID 9618502.
- ^ PMID 25382122.
- S2CID 18250196.
- S2CID 205055573.
- S2CID 4422345.
- S2CID 199534387. Retrieved 2 November 2023.
- from the original on 23 February 2024, retrieved 2 November 2023
- ^ (PDF) from the original on 21 October 2022. Retrieved 15 August 2021.
- S2CID 2767388.
- PMID 26475454.
- PMID 23550145.
Further reading
- ISBN 978-1781250372.


