Lithium: Difference between revisions
25,040 edits m Undid revision 817334447 by 188.121.220.222 (talk) rV test |
Rescuing 1 sources and tagging 0 as dead. #IABot (v1.6.1) |
||
| Line 225: | Line 225: | ||
According to a 2011 study by [[Lawrence Berkeley National Laboratory]] and the [[University of California, Berkeley]], the currently estimated reserve base of lithium should not be a limiting factor for large-scale battery production for electric vehicles because an estimated 1 billion 40 [[kWh]] Li-based batteries could be built with current reserves<ref>{{cite journal |last1=Wadia |first1=Cyrus |last2=Albertus |first2=Paul |last3=Srinivasan |first3=Venkat |title=Resource constraints on the battery energy storage potential for grid and transportation applications |journal=Journal of Power Sources |volume=196 |issue=3 |year=2011 |pages=1593–8 |doi=10.1016/j.jpowsour.2010.08.056 |bibcode=2011JPS...196.1593W }}</ref> - about 10 kg of lithium per car.<ref>{{cite web|url=http://www.anl.gov/energy-systems/publication/lithium-ion-batteries-examining-material-demand-and-recycling-issues|title=Lithium-Ion Batteries: Examining Material Demand and Recycling Issues|author=Gaines, LL. Nelson, P.|publisher=[[Argonne National Laboratory]]|date=2010|accessdate=11 June 2016|deadurl=no|archiveurl=https://web.archive.org/web/20160803104250/http://www.anl.gov/energy-systems/publication/lithium-ion-batteries-examining-material-demand-and-recycling-issues|archivedate=3 August 2016|df=dmy-all}}</ref> Another 2011 study at the [[University of Michigan]] and [[Ford Motor Company]] found enough resources to support global demand until 2100, including the lithium required for the potential widespread transportation use. The study estimated global reserves at 39 million tons, and total demand for lithium during the 90-year period analyzed at 12–20 million tons, depending on the scenarios regarding economic growth and recycling rates.<ref>{{cite web|url=http://www.greencarcongress.com/2011/08/lithium-20110803.html|title=University of Michigan and Ford researchers see plentiful lithium resources for electric vehicles|publisher=[[Green Car Congress]]|date=3 August 2011|accessdate=11 August 2011|deadurl=no|archiveurl=https://web.archive.org/web/20110916075249/http://www.greencarcongress.com/2011/08/lithium-20110803.html|archivedate=16 September 2011|df=dmy-all}}</ref> |
According to a 2011 study by [[Lawrence Berkeley National Laboratory]] and the [[University of California, Berkeley]], the currently estimated reserve base of lithium should not be a limiting factor for large-scale battery production for electric vehicles because an estimated 1 billion 40 [[kWh]] Li-based batteries could be built with current reserves<ref>{{cite journal |last1=Wadia |first1=Cyrus |last2=Albertus |first2=Paul |last3=Srinivasan |first3=Venkat |title=Resource constraints on the battery energy storage potential for grid and transportation applications |journal=Journal of Power Sources |volume=196 |issue=3 |year=2011 |pages=1593–8 |doi=10.1016/j.jpowsour.2010.08.056 |bibcode=2011JPS...196.1593W }}</ref> - about 10 kg of lithium per car.<ref>{{cite web|url=http://www.anl.gov/energy-systems/publication/lithium-ion-batteries-examining-material-demand-and-recycling-issues|title=Lithium-Ion Batteries: Examining Material Demand and Recycling Issues|author=Gaines, LL. Nelson, P.|publisher=[[Argonne National Laboratory]]|date=2010|accessdate=11 June 2016|deadurl=no|archiveurl=https://web.archive.org/web/20160803104250/http://www.anl.gov/energy-systems/publication/lithium-ion-batteries-examining-material-demand-and-recycling-issues|archivedate=3 August 2016|df=dmy-all}}</ref> Another 2011 study at the [[University of Michigan]] and [[Ford Motor Company]] found enough resources to support global demand until 2100, including the lithium required for the potential widespread transportation use. The study estimated global reserves at 39 million tons, and total demand for lithium during the 90-year period analyzed at 12–20 million tons, depending on the scenarios regarding economic growth and recycling rates.<ref>{{cite web|url=http://www.greencarcongress.com/2011/08/lithium-20110803.html|title=University of Michigan and Ford researchers see plentiful lithium resources for electric vehicles|publisher=[[Green Car Congress]]|date=3 August 2011|accessdate=11 August 2011|deadurl=no|archiveurl=https://web.archive.org/web/20110916075249/http://www.greencarcongress.com/2011/08/lithium-20110803.html|archivedate=16 September 2011|df=dmy-all}}</ref> |
||
On June 9, 2014, the ''Financialist'' stated that demand for lithium was growing at more than 12% a year. According to Credit Suisse, this rate exceeds projected availability by 25%. The publication compared the 2014 lithium situation with oil, whereby "higher oil prices spurred investment in expensive deepwater and oil sands production techniques"; that is, the price of lithium will continue to rise until more expensive production methods that can boost total output receive the attention of investors.<ref>{{cite web|title=The Precious Mobile Metal|url=http://www.thefinancialist.com/spark/the-precious-mobile-metal/|website=The Financialist|publisher=Credit Suisse|accessdate=19 June 2014|date=9 June 2014|deadurl= |
On June 9, 2014, the ''Financialist'' stated that demand for lithium was growing at more than 12% a year. According to Credit Suisse, this rate exceeds projected availability by 25%. The publication compared the 2014 lithium situation with oil, whereby "higher oil prices spurred investment in expensive deepwater and oil sands production techniques"; that is, the price of lithium will continue to rise until more expensive production methods that can boost total output receive the attention of investors.<ref>{{cite web|title=The Precious Mobile Metal|url=http://www.thefinancialist.com/spark/the-precious-mobile-metal/|website=The Financialist|publisher=Credit Suisse|accessdate=19 June 2014|date=9 June 2014|deadurl=yes|archiveurl=https://web.archive.org/web/20160223144634/https://www.thefinancialist.com/spark/the-precious-mobile-metal/|archivedate=23 February 2016|df=dmy-all}}</ref> |
||
=== Pricing === |
=== Pricing === |
||
Revision as of 17:05, 3 January 2018
It has been suggested that Lithium as an investment be merged into this article. (Discuss) Proposed since June 2017. |
 Lithium floating in oil | |||||||||||||||||||||
| Lithium | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pronunciation | /ˈlɪθiəm/ | ||||||||||||||||||||
| Appearance | silvery-white | ||||||||||||||||||||
| Standard atomic weight Ar°(Li) | |||||||||||||||||||||
| Lithium in the periodic table | |||||||||||||||||||||
| |||||||||||||||||||||
kJ/mol | |||||||||||||||||||||
| Heat of vaporization | 136 kJ/mol | ||||||||||||||||||||
| Molar heat capacity | 24.860 J/(mol·K) | ||||||||||||||||||||
Vapor pressure
| |||||||||||||||||||||
| Atomic properties | |||||||||||||||||||||
Discovery | Johan August Arfwedson (1817) | ||||||||||||||||||||
| First isolation | William Thomas Brande (1821) | ||||||||||||||||||||
| Isotopes of lithium | |||||||||||||||||||||
| |||||||||||||||||||||
Lithium (from
The
Lithium and its compounds have several industrial applications, including heat-resistant glass and
Lithium is present in biological systems in trace amounts; its functions are uncertain. Lithium salts have proven to be useful as a mood-stabilizing drug in the treatment of bipolar disorder in humans.
Properties
Atomic and physical

Like the other
Lithium metal is soft enough to be cut with a knife. When cut, it possesses a silvery-white color that quickly changes to gray as it oxidizes to
Lithium has a very low density (0.534 g/cm3), comparable with pine wood. It is the least dense of all elements that are solids at room temperature; the next lightest solid element (potassium, at 0.862 g/cm3) is more than 60% denser. Furthermore, apart from helium and hydrogen, it is less dense than any liquid element, being only two thirds as dense as liquid nitrogen (0.808 g/cm3).[11] Lithium can float on the lightest hydrocarbon oils and is one of only three metals that can float on water, the other two being sodium and potassium.

Lithium's
Lithium has a mass specific heat capacity of 3.58 kilojoules per kilogram-kelvin, the highest of all solids.[17][18] Because of this, lithium metal is often used in coolants for heat transfer applications.[17]
Chemistry and compounds
Lithium reacts with water easily, but with noticeably less vigor than other alkali metals. The reaction forms hydrogen gas and lithium hydroxide in aqueous solution.[9] Because of its reactivity with water, lithium is usually stored in a hydrocarbon sealant, often petroleum jelly. Though the heavier alkali metals can be stored in more dense substances, such as mineral oil, lithium is not dense enough to be fully submerged in these liquids.[19] In moist air, lithium rapidly tarnishes to form a black coating of lithium hydroxide (LiOH and LiOH·H2O), lithium nitride (Li3N) and lithium carbonate (Li2CO3, the result of a secondary reaction between LiOH and CO2).[20]
When placed over a flame, lithium compounds give off a striking crimson color, but when it burns strongly the flame becomes a brilliant silver. Lithium will ignite and burn in oxygen when exposed to water or water vapors.
Lithium has a diagonal relationship with magnesium, an element of similar atomic and ionic radius. Chemical resemblances between the two metals include the formation of a nitride by reaction with N2, the formation of an oxide (Li
2O) and peroxide (Li
2O
2) when burnt in O2, salts with similar solubilities, and thermal instability of the carbonates and nitrides.[20][24] The metal reacts with hydrogen gas at high temperatures to produce lithium hydride (LiH).[25]
Other known
4). Lithium aluminium hydride
4) is commonly used as a reducing agent in organic synthesis.
Multiple
Isotopes
Naturally occurring lithium is composed of two stable
7Li is one of the
Lithium isotopes fractionate substantially during a wide variety of natural processes,
Nuclear weapons manufacture and other nuclear physics applications are a major source of artificial lithium fractionation, with the light isotope 6Li being retained by industry and military stockpiles to such an extent that it has caused slight but measurable change in the 6Li to 7Li ratios in natural sources, such as rivers. This has led to unusual uncertainty in the standardized
Both stable isotopes of lithium can be laser cooled and were used to produce the first quantum degenerate Bose-Fermi mixture.[37]
Occurrence
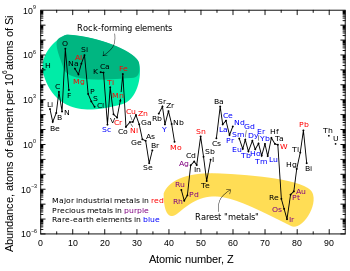
Astronomical
Though it was synthesized in the Big Bang, lithium (together with beryllium and boron), is markedly less abundant in the universe than other elements. This is a result of the comparatively low stellar temperatures necessary to destroy lithium, along with a lack of common processes to produce it.[38]
According to modern cosmological theory, lithium—in both stable isotopes (lithium-6 and lithium-7)—was one of the 3 elements synthesized in the Big Bang.[39] Though the amount of lithium generated in Big Bang nucleosynthesis is dependent upon the number of photons per baryon, for accepted values the lithium abundance can be calculated, and there is a "cosmological lithium discrepancy" in the Universe: older stars seem to have less lithium than they should, and some younger stars have much more.[40] The lack of lithium in older stars is apparently caused by the "mixing" of lithium into the interior of stars, where it is destroyed,[41] while lithium is produced in younger stars. Though it transmutes into two atoms of helium due to collision with a proton at temperatures above 2.4 million degrees Celsius (most stars easily attain this temperature in their interiors), lithium is more abundant than current computations would predict in later-generation stars.[19]
Lithium is also found in brown dwarf substellar objects and certain anomalous orange stars. Because lithium is present in cooler, less-massive brown dwarfs, but is destroyed in hotter red dwarf stars, its presence in the stars' spectra can be used in the "lithium test" to differentiate the two, as both are smaller than the Sun.[19][43][44] Certain orange stars can also contain a high concentration of lithium. Those orange stars found to have a higher than usual concentration of lithium (such as Centaurus X-4) orbit massive objects—neutron stars or black holes—whose gravity evidently pulls heavier lithium to the surface of a hydrogen-helium star, causing more lithium to be observed.[19]
Terrestrial
| Country | Production | Reserves[note 1] | Resources |
|---|---|---|---|
| 5,700 | 2,000,000 | 9,000,000 | |
| 14,300 | 1,600,000 | 2,000,000+ | |
| - | - | 100,000+ | |
| - | - | 9,000,000 | |
| 200 | 48,000 | 200,000 | |
| 480 | 180,000 | 2,000,000 | |
| 12,000 | 7,500,000 | 7,500,000+ | |
| - | - | 1,000,000 | |
| - | - | 200,000 | |
| 2,000 | 3,200,000 | 7,000,000 | |
| 200 | 60,000 | N/A | |
| - | - | 1,000,000 | |
| - | - | 1,000,000 | |
| W[note 2] | 38,000 | 6,900,000 | |
| 900 | 23,000 | 100,000+ | |
| World total | 32,500 | 14,000,000 | 46,900,000 |
Although lithium is widely distributed on Earth, it does not naturally occur in elemental form due to its high reactivity.
Estimates for the Earth's crustal content range from 20 to 70 ppm by weight.[20] In keeping with its name, lithium forms a minor part of igneous rocks, with the largest concentrations in granites. Granitic pegmatites also provide the greatest abundance of lithium-containing minerals, with spodumene and petalite being the most commercially viable sources.[20] Another significant mineral of lithium is lepidolite.[49] A newer source for lithium is hectorite clay, the only active development of which is through the Western Lithium Corporation in the United States.[50] At 20 mg lithium per kg of Earth's crust,[51] lithium is the 25th most abundant element.
According to the Handbook of Lithium and Natural Calcium, "Lithium is a comparatively rare element, although it is found in many rocks and some brines, but always in very low concentrations. There are a fairly large number of both lithium mineral and brine deposits but only comparatively few of them are of actual or potential commercial value. Many are very small, others are too low in grade."[52]
The
In June 2010,
Lithia ("lithium brine") is associated with tin mining areas in Cornwall, England and an evaluation project from 400-metre deep test boreholes is under consideration. If successful the hot brines will also provide geothermal energy to power the lithium extraction and refining process.[58]
Biological
Lithium is found in trace amount in numerous plants, plankton, and invertebrates, at concentrations of 69 to 5,760 parts per billion (ppb). In vertebrates the concentration is slightly lower, and nearly all vertebrate tissue and body fluids contain lithium ranging from 21 to 763 ppb.[47] Marine organisms tend to bioaccumulate lithium more than terrestrial organisms.[59] Whether lithium has a physiological role in any of these organisms is unknown.[47]
History

Arfwedson later showed that this same element was present in the minerals
The production and use of lithium underwent several drastic changes in history. The first major application of lithium was in high-temperature
The demand for lithium increased dramatically during the
Lithium was used to decrease the melting temperature of glass and to improve the melting behavior of
The development of lithium ion batteries increased the demand for lithium and became the dominant use in 2007.[82] With the surge of lithium demand in batteries in the 2000s, new companies have expanded brine extraction efforts to meet the rising demand.[83][84]
Production

Lithium production has greatly increased since the end of World War II. The metal is separated from other elements in igneous minerals. The metal is produced through electrolysis from a mixture of fused 55% lithium chloride and 45% potassium chloride at about 450 °C.[85]
As of 2015, most of the world's lithium production is in South America, where lithium-containing brine is extracted from underground pools and concentrated by solar evaporation. The standard extraction technique is to evaporate water from brine. Each batch takes from 18 to 24 months.[86]
In 1998, the price of lithium was about 95 USD/kg (or 43 USD/lb).[87]
Reserves
Worldwide identified reserves in 2008 were estimated by the
Deposits are found in South America throughout the Andes mountain chain. Chile is the leading producer, followed by Argentina. Both countries recover lithium from brine pools. According to USGS, Bolivia's Uyuni Desert has 5.4 million tonnes of lithium.[93][94]
In the US, lithium is recovered from brine pools in Nevada.[17] Half the world's known reserves are located in Bolivia along the central eastern slope of the Andes. In 2009, Bolivia negotiated with Japanese, French, and Korean firms to begin extraction.[93] A deposit discovered in 2013 in Wyoming's Rock Springs Uplift is estimated to contain 228,000 tons. Additional deposits in the same formation were estimated to be as much as 18 million tons.[95]
Opinions differ about potential growth. A 2008 study concluded that "realistically achievable lithium carbonate production will be sufficient for only a small fraction of future
According to a 2011 study by
On June 9, 2014, the Financialist stated that demand for lithium was growing at more than 12% a year. According to Credit Suisse, this rate exceeds projected availability by 25%. The publication compared the 2014 lithium situation with oil, whereby "higher oil prices spurred investment in expensive deepwater and oil sands production techniques"; that is, the price of lithium will continue to rise until more expensive production methods that can boost total output receive the attention of investors.[99]
Pricing
After the
Extraction
Lithium salts are extracted from water in
Lithium is present in seawater, but commercially viable methods of extraction have yet to be developed.[86]
Another potential source of lithium is the leachates of
Uses

Ceramics and glass
Lithium oxide is widely used as a
Electrical and electronics
Late in the 20th century, lithium became an important component of battery electrolytes and electrodes, because of its high
Lubricating greases
The third most common use of lithium is in greases. Lithium hydroxide is a strong
Metallurgy
Lithium (e.g. as lithium carbonate) is used as an additive to
Lithium (as lithium fluoride) is used as an additive to aluminium smelters (Hall–Héroult process), reducing melting temperature and increasing electrical resistance,[115] a use which accounts for 3% of production (2011).[45]
When used as a
Silicon nano-welding
Lithium has been found effective in assisting the perfection of silicon nano-welds in electronic components for electric batteries and other devices.[119]
Other chemical and industrial uses

Pyrotechnics
Lithium compounds are used as
Air purification
Lithium peroxide (Li2O2) in presence of moisture not only reacts with carbon dioxide to form lithium carbonate, but also releases oxygen.[122][123] The reaction is as follows:
- 2 Li2O2 + 2 CO2 → 2 Li2CO3 + O2.
Some of the aforementioned compounds, as well as
Optics
The high non-linearity of lithium niobate also makes it useful in non-linear optics applications. It is used extensively in telecommunication products such as mobile phones and optical modulators, for such components as resonant crystals. Lithium applications are used in more than 60% of mobile phones.[128]
Organic and polymer chemistry
Many other lithium compounds are used as reagents to prepare organic compounds. Some popular compounds include
Military applications
Metallic lithium and its complex hydrides, such as Li[AlH4], are used as high-energy additives to rocket propellants.[19] Lithium aluminum hydride can also be used by itself as a solid fuel.[134]

The Mark 50 torpedo stored chemical energy propulsion system (SCEPS) uses a small tank of sulfur hexafluoride gas, which is sprayed over a block of solid lithium. The reaction generates heat, creating steam to propel the torpedo in a closed Rankine cycle.[135]
Lithium hydride containing lithium-6 is used in thermonuclear weapons, where it serves as fuel for the fusion stage of the bomb.[136]
Nuclear
Lithium-6 is valued as a source material for

In conceptualized (hypothetical) nuclear fusion power plants, lithium will be used to produce tritium in magnetically confined reactors using deuterium and tritium as the fuel. Naturally occurring tritium is extremely rare, and must be synthetically produced by surrounding the reacting plasma with a 'blanket' containing lithium where neutrons from the deuterium-tritium reaction in the plasma will fission the lithium to produce more tritium:
- 6Li + n → 4He + 3H.
Lithium is also used as a source for
In 2013, the US Government Accountability Office said a shortage of lithium-7 critical to the operation of 65 out of 100 American nuclear reactors “places their ability to continue to provide electricity at some risk”. The problem stems from the decline of US nuclear infrastructure. The equipment needed to separate lithium-6 from lithium-7 is mostly a cold war leftover. The US shut down most of this machinery in 1963, when it had a huge surplus of separated lithium, mostly consumed during the twentieth century. The report said it would take five years and $10 million to $12 million to reestablish the ability to separate lithium-6 from lithium-7.[143]
Reactors that use lithium-7 heat water under high pressure and transfer heat through heat exchangers that are prone to corrosion. The reactors use lithium to counteract the corrosive effects of boric acid, which is added to the water to absorb excess neutrons.[143]
Medicine
Lithium is useful in the treatment of
Lithium has also been researched as a possible treatment for cluster headaches.[146]
Biological role
Primary food sources of lithium are grains and vegetables, and, in some areas, drinking water also contains significant amounts.[147] Human intake varies depending on location and diet.
Lithium was first detected in human organs and fetal tissues in the late 19th century. In humans there are no defined lithium deficiency diseases, but low lithium intakes from water supplies were associated with increased rates of suicides, homicides and the arrest rates for drug use and other crimes. The biochemical mechanisms of action of lithium appear to be multifactorial and are intercorrelated with the functions of several enzymes, hormones and vitamins, as well as with growth and transforming factors. Evidence now appears to be sufficient to accept lithium as essential; a provisional RDA of 1,000 µg/day is suggested for a 70 kg adult.[147][148]
Precautions
| NFPA 704 fire diamond | |
|---|---|
Fire diamond hazard sign for lithium metal[149] |
Lithium is
Regulation
Some jurisdictions limit the sale of lithium batteries, which are the most readily available source of lithium for ordinary consumers. Lithium can be used to reduce pseudoephedrine and ephedrine to methamphetamine in the Birch reduction method, which employs solutions of alkali metals dissolved in anhydrous ammonia.[151][152]
Carriage and shipment of some kinds of lithium batteries may be prohibited aboard certain types of transportation (particularly aircraft) because of the ability of most types of lithium batteries to fully discharge very rapidly when short-circuited, leading to overheating and possible explosion in a process called thermal runaway. Most consumer lithium batteries have built-in thermal overload protection to prevent this type of incident, or are otherwise designed to limit short-circuit currents. Internal shorts from manufacturing defect or physical damage can lead to spontaneous thermal runaway.[153][154]
See also
- Dilithium
- List of countries by lithium production
- Lithium as an investment
- Lithium compounds
- Lithium (medication)
- Lithium soap
- Lithium-ion battery
- Lithium–air battery
Notes
- ^ a b Apendixes Archived 6 November 2011 at the Wayback Machine. By USGS definitions, the reserve base "may encompass those parts of the resources that have a reasonable potential for becoming economically available within planning horizons beyond those that assume proven technology and current economics. The reserve base includes those resources that are currently economic (reserves), marginally economic (marginal reserves), and some of those that are currently subeconomic (subeconomic resources)."
- ^ withheld to avoid disclosing company proprietary data
- ^ Beryllium and fluorine occur only as one isotope, 9Be and 19F respectively. These two, together with 7Li, as well as 2H, 11B, 15N, 209Bi, and the stable isotopes of C, and O, are the only nuclides with low enough thermal neutron capture cross sections aside from actinides to serve as major constituents of a molten salt breeder reactor fuel.
References
- ^ "Standard Atomic Weights: Lithium". CIAAW. 2009.
- ISSN 1365-3075.
- ^ ISBN 978-1-62708-155-9.
- PMID 29114648.
- ISBN 0-8493-0464-4.
- .
- ^ doi:10.1086/375492. Archived from the original (PDF) on 7 November 2015.
{{cite journal}}: Invalid|ref=harv(help); Unknown parameter|deadurl=ignored (|url-status=suggested) (help) Graphed at File:SolarSystemAbundances.jpg - ^ Nuclear Weapon Design. Federation of American Scientists (1998-10-21). fas.org
- ^ ISBN 0-313-33438-2.
- ISBN 0-8493-0486-5.
- ^ "Nitrogen, N2, Physical properties, safety, MSDS, enthalpy, material compatibility, gas liquid equilibrium, density, viscosity, inflammability, transport properties". Encyclopedia.airliquide.com. Archived from the original on 21 July 2011. Retrieved 29 September 2010.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ "Coefficients of Linear Expansion". Engineering Toolbox. Archived from the original on 30 November 2012.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - PMID 17495921.
- PMID 12386338.
- .
- .
- ^ ISBN 0-8493-0481-4.[page needed]
- ^ SPECIFIC HEAT OF SOLIDS. bradley.edu
- ^ ISBN 0-19-850341-5.
- ^ .
- .
- ISBN 0-313-33438-2. Archived from the original on 4 August 2016.)
{{cite book}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help - ^ Institute, American Geological; Union, American Geophysical; Society, Geochemical (1 January 1994). "Geochemistry international". 31 (1–4): 115. Archived from the original on 4 June 2016.
{{cite journal}}: Cite journal requires|journal=(help); Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ISBN 978-0-08-022057-4.
- ^ Beckford, Floyd. "University of Lyon course online (powerpoint) slideshow". Archived from the original on 4 November 2005. Retrieved 27 July 2008.
definitions:Slides 8–10 (Chapter 14)
{{cite web}}: Unknown parameter|dead-url=ignored (|url-status=suggested) (help) - ISBN 0-471-54930-4. Archived from the original on 31 July 2016.)
{{cite book}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help); Unknown parameter|last-author-amp=ignored (|name-list-style=suggested) (help - ^ Bretislav Friedrich (8 April 2013). "APS Physics". 6: 42. Archived from the original on 20 December 2016.
{{cite journal}}: Cite journal requires|journal=(help); Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ "Isotopes of Lithium". Berkeley National Laboratory, The Isotopes Project. Archived from the original on 13 May 2008. Retrieved 21 April 2008.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ File:Binding energy curve - common isotopes.svg shows binding energies of stable nuclides graphically; the source of the data-set is given in the figure background.
- ^ Sonzogni, Alejandro. "Interactive Chart of Nuclides". National Nuclear Data Center: Brookhaven National Laboratory. Archived from the original on 23 July 2007. Retrieved 6 June 2008.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - doi:10.1086/503538.
- doi:10.1016/j.gca.2005.08.016. Archived from the original (PDF) on 18 July 2010.)
{{cite journal}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help - Bibcode:2000A&A...358L..49D.
- .
- ISBN 1-4200-6009-0.
- ^ .
- PMID 11283362. Archived from the original on 15 August 2017.)
{{cite journal}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help - ^ "Element Abundances" (PDF). Archived from the original (PDF) on 1 September 2006. Retrieved 17 November 2009.
- . A86-14507 04–90.
- ^ Woo, Marcus (21 February 2017). "The Cosmic Explosions That Made the Universe". earth. BBC. Archived from the original on 21 February 2017. Retrieved 21 February 2017.
A mysterious cosmic factory is producing lithium. Scientists are now getting closer at finding out where it comes from
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ Cain, Fraser (16 August 2006). "Why Old Stars Seem to Lack Lithium". Archived from the original on 4 June 2016.
{{cite news}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ "First Detection of Lithium from an Exploding Star". Archived from the original on 29 July 2015. Retrieved 29 July 2015.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ Cain, Fraser. "Brown Dwarf". Universe Today. Archived from the original on 25 February 2011. Retrieved 17 November 2009.
- ^ Reid, Neill (10 March 2002). "L Dwarf Classification". Archived from the original on 21 May 2013. Retrieved 6 March 2013.
{{cite web}}: Unknown parameter|dead-url=ignored (|url-status=suggested) (help) - ^ a b c d e Lithium Statistics and Information, U.S. Geological Survey, 2017, archived from the original on 3 March 2016
{{citation}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ "Lithium Occurrence". Institute of Ocean Energy, Saga University, Japan. Archived from the original on 2 May 2009. Retrieved 13 March 2009.
- ^ a b c d "Some Facts about Lithium". ENC Labs. Archived from the original on 10 July 2011. Retrieved 15 October 2010.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - .
- ISBN 0199236178.
- ^ Moores, S. (June 2007). "Between a rock and a salt lake". Industrial Minerals. 477: 58.
- ^ Taylor, S. R.; McLennan, S. M.; The continental crust: Its composition and evolution, Blackwell Sci. Publ., Oxford, 330 pp. (1985). Cited in Abundances of the elements (data page)
- ^ Garrett, Donald (2004) Handbook of Lithium and Natural Calcium, Academic Press, cited in The Trouble with Lithium 2 Archived 14 July 2011 at the Wayback Machine, Meridian International Research (2008)
- ^ Clarke, G.M. and Harben, P.W., "Lithium Availability Wall Map". Published June 2009. Referenced at International Lithium Alliance Archived 20 October 2012 at archive.today
- ^ a b "The Trouble with Lithium 2" (PDF). Meridian International Research. 2008. Archived from the original (PDF) on 14 July 2011. Retrieved 29 September 2010.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ISBN 978-80-7075-904-2. Archived from the original (PDF) on 6 January 2017.)
{{cite book}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help - ^ Risen, James (13 June 2010). "U.S. Identifies Vast Riches of Minerals in Afghanistan". The New York Times. Archived from the original on 17 June 2010. Retrieved 13 June 2010.
{{cite news}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ Page, Jeremy; Evans, Michael (15 June 2010). "Taleban zones mineral riches may rival Saudi Arabia says Pentagon". The Times. London. Archived from the original on 14 May 2011.
{{cite news}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ Morris, Steven (20 January 2017). "Mining firm hopes to extract lithium from Cornwall's hot springs". The Guardian. p. 31.
{{cite news}}:|access-date=requires|url=(help) - PMID 6440674.
- D'Andraba (1800). "Des caractères et des propriétés de plusieurs nouveaux minérauxde Suède et de Norwège, avec quelques observations chimiques faites sur ces substances". Journal de chimie et de physique. 51: 239. Archived from the original on 13 July 2015.)
{{cite journal}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help - ^ "Petalite Mineral Information". Mindat.org. Archived from the original on 16 February 2009. Retrieved 10 August 2009.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ a b c d e f g "Lithium:Historical information". Archived from the original on 16 October 2009. Retrieved 10 August 2009.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ISBN 0-7661-3872-0. Retrieved 10 August 2009.
- ^ Berzelius (1817). "Ein neues mineralisches Alkali und ein neues Metall" [A new mineral alkali and a new metal]. Journal für Chemie und Physik. 21: 44–48. Archived from the original on 3 December 2016.
{{cite journal}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) From p. 45: "Herr August Arfwedson, ein junger sehr verdienstvoller Chemiker, der seit einem Jahre in meinem Laboratorie arbeitet, fand bei einer Analyse des Petalits von Uto's Eisengrube, einen alkalischen Bestandtheil, … Wir haben es Lithion genannt, um dadurch auf seine erste Entdeckung im Mineralreich anzuspielen, da die beiden anderen erst in der organischen Natur entdeckt wurden. Sein Radical wird dann Lithium genannt werden." (Mr. August Arfwedson, a young, very meritorious chemist, who has worked in my laboratory for a year, found during an analysis of petalite from Uto's iron mine, an alkaline component … We've named it lithion, in order to allude thereby to its first discovery in the mineral realm, since the two others were first discovered in organic nature. Its radical will then be named "lithium".) - ^ "Johan August Arfwedson". Periodic Table Live!. Archived from the original on 7 October 2010. Retrieved 10 August 2009.
- ^ "Johan Arfwedson". Archived from the original on 5 June 2008. Retrieved 10 August 2009.
- ^ a b c van der Krogt, Peter. "Lithium". Elementymology & Elements Multidict. Archived from the original on 16 June 2011. Retrieved 5 October 2010.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ Clark, Jim (2005). "Compounds of the Group 1 Elements". Archived from the original on 11 March 2009. Retrieved 10 August 2009.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ See:
- Arwedson, Aug. (1818) "Undersökning af några vid Utö Jernmalmsbrott förekommende Fossilier, och af ett deri funnet eget Eldfast Alkali" Archived 25 November 2017 at the Wayback Machine, Afhandlingar i Fysik, Kemi och Mineralogi, 6 : 145–172. (in Swedish)
- Arwedson, Aug. (1818) "Untersuchung einiger bei der Eisen-Grube von Utö vorkommenden Fossilien und von einem darin gefundenen neuen feuerfesten Alkali" (Investigation of some minerals occurring at the iron mines of Utö and of a new refractory alkali found therein), Journal für Chemie und Physik, 22 (1) : 93–117. (in German)
- doi:10.1002/andp.18180590702. Archived from the original on 9 November 2015.)
p. 238 Es löste sich in diesem ein Salz auf, das an der Luft zerfloss, und nach Art der Strontiansalze den Alkohol mit einer purpurrothen Flamme brennen machte. (There dissolved in this [solvent; namely, absolute alcohol] a salt that deliquesced in air, and in the manner of strontium salts, caused the alcohol to burn with a purple-red flame.)
{{cite journal}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help - ^ ISBN 978-3-527-30666-4.
- ^ Brande, William Thomas (1821) A Manual of Chemistry, 2nd ed. London, England: John Murray, vol. 2, pp. 57-58. Archived 22 November 2015 at the Wayback Machine
- ^ Various authors (1818). "The Quarterly journal of science and the arts" (PDF). The Quarterly Journal of Science and the Arts. 5. Royal Institution of Great Britain: 338. Retrieved 5 October 2010.
- ^ "Timeline science and engineering". DiracDelta Science & Engineering Encyclopedia. Archived from the original on 5 December 2008. Retrieved 18 September 2008.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ Brande, William Thomas; MacNeven, William James (1821). A manual of chemistry. Long. p. 191. Retrieved 8 October 2010.
- .
- ^ Green, Thomas (11 June 2006). "Analysis of the Element Lithium". echeat. Archived from the original on 21 April 2012.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ISBN 9780080472904. Archived from the original on 3 December 2016.)
{{cite book}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help - ^ a b Ober, Joyce A. (1994). "Commodity Report 1994: Lithium" (PDF). United States Geological Survey. Archived from the original (PDF) on 9 June 2010. Retrieved 3 November 2010.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - .
- .
- ^ Ober, Joyce A. (1994). "Minerals Yearbook 2007 : Lithium" (PDF). United States Geological Survey. Archived from the original (PDF) on 17 July 2010. Retrieved 3 November 2010.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ISBN 978-0-87335-233-8.
- ISBN 978-0-8247-2478-8. Archived from the original on 28 May 2013. Retrieved 29 September 2010.)
{{cite book}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help - ISBN 978-0-08-037941-8.
- ^ a b Martin, Richard (8 June 2015). "Quest to Mine Seawater for Lithium Advances". MIT Technology Review. Retrieved 10 February 2016.
- ^ Ober, Joyce A. "Lithium" (PDF). United States Geological Survey. pp. 77–78. Archived from the original (PDF) on 11 July 2007. Retrieved 19 August 2007.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - PMID 20489722.
- ^ Lithium: The New California Gold Rush Archived 29 July 2017 at the Wayback Machine, Forbes magazine. 2011-10-19
- .
- doi:10.1016/j.apenergy.2013.04.005. Archived from the original on 11 October 2017. Retrieved 11 October 2017.)
{{cite journal}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help - )
- ^ a b Romero, Simon (2 February 2009). "In Bolivia, a Tight Grip on the Next Big Resource". The New York Times. Archived from the original on 1 July 2017.
{{cite news}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ "USGS Mineral Commodities Summaries 2009" (PDF). USGS. Archived from the original (PDF) on 14 June 2010.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ Money Game Contributors (26 April 2013). "New Wyoming Lithium Deposit". Business Insider. Archived from the original on 3 May 2013. Retrieved 1 May 2013.
{{cite web}}:|author=has generic name (help); Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - .
- ^ Gaines, LL. Nelson, P. (2010). "Lithium-Ion Batteries: Examining Material Demand and Recycling Issues". Argonne National Laboratory. Archived from the original on 3 August 2016. Retrieved 11 June 2016.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help)CS1 maint: multiple names: authors list (link) - ^ "University of Michigan and Ford researchers see plentiful lithium resources for electric vehicles". Green Car Congress. 3 August 2011. Archived from the original on 16 September 2011. Retrieved 11 August 2011.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ "The Precious Mobile Metal". The Financialist. Credit Suisse. 9 June 2014. Archived from the original on 23 February 2016. Retrieved 19 June 2014.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ "SQM Announces New Lithium Prices – SANTIAGO, Chile, September 30 /PRNewswire-FirstCall/". PR Newswire. 30 September 2009. Archived from the original on 30 May 2013. Retrieved 1 May 2013.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ Riseborough, Jesse. "IPad Boom Strains Lithium Supplies After Prices Triple". Bloomberg BusinessWeek. Archived from the original on 22 June 2012. Retrieved 1 May 2013.
{{cite web}}: Unknown parameter|dead-url=ignored (|url-status=suggested) (help) - ^ Parker, Ann. Mining Geothermal Resources Archived 17 September 2012 at the Wayback Machine. Lawrence Livermore National Laboratory
- ^ Patel, P. (2011-11-16) Startup to Capture Lithium from Geothermal Plants. technologyreview.com
- ^ Wald, M. (2011-09-28) Start-Up in California Plans to Capture Lithium, and Market Share Archived 8 April 2017 at the Wayback Machine. The New York Times
- ^ a b "Lithium" (PDF). 2016. Archived from the original (PDF) on 30 November 2016. Retrieved 29 November 2016 – via US Geological Survey (USGS).
{{cite news}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ Worldwide demand by sector Archived 7 September 2014 at the Wayback Machine
- ^ Clark, Jim (2005). "Some Compounds of the Group 1 Elements". chemguide.co.uk. Archived from the original on 27 June 2013. Retrieved 8 August 2013.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ "Disposable Batteries - Choosing between Alkaline and Lithium Disposable Batteries". Batteryreview.org. Archived from the original on 6 January 2014. Retrieved 10 October 2013.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ "Battery Anodes > Batteries & Fuel Cells > Research > The Energy Materials Center at Cornell". Emc2.cornell.edu. Archived from the original on 22 December 2013. Retrieved 10 October 2013.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ISBN 0-8031-2096-6. Archived from the original on 23 July 2016.)
{{cite book}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help); Unknown parameter|last-author-amp=ignored (|name-list-style=suggested) (help - ISBN 0-8031-2097-4. Archived from the original on 31 July 2016.)
{{cite book}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help - ^ The Theory and Practice of Mold Fluxes Used in Continuous Casting: A Compilation of Papers on Continuous Casting Fluxes Given at the 61st and 62nd Steelmaking Conference, Iron and Steel Society
- .
- ^ "Testing 1-2-3: Eliminating Veining Defects", Modern Casting, July 2014, archived from the original on 2 April 2015
{{citation}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ Haupin, W. (1987), Mamantov, Gleb; Marassi, Roberto (eds.), "Chemical and Physical Properties of the Hall-Héroult Electrolyte", Molten Salt Chemistry: An Introduction and Selected Applications, Springer, p. 449
- ISBN 9780080472904. Archived from the original on 3 December 2016.)
{{cite book}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help - ISBN 9780124016798.
- ISBN 978-0-87170-496-2. Archived from the original on 28 May 2013. Retrieved 16 May 2011.)
{{cite book}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help - PMID 22339576. Archived from the original (PDF) on 10 August 2017.)
{{cite journal}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help - .
- ISBN 0-12-352651-5, p. 1089
- doi:10.1007/b107253.)
{{cite book}}: Unknown parameter|last-author-amp=ignored (|name-list-style=suggested) (help - ^ "Application of lithium chemicals for air regeneration of manned spacecraft". Lithium Corporation of America & Aerospace Medical Research Laboratories. 1965. Archived from the original on 7 October 2012.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - .
- ISBN 0-470-40229-6. Archived from the original on 23 June 2016.)
{{cite book}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help - ISBN 981-238-180-5. Archived from the original on 6 June 2016.)
{{cite book}}:|journal=ignored (help); Unknown parameter|deadurl=ignored (|url-status=suggested) (help - .
- ^ "You've got the power: the evolution of batteries and the future of fuel cells" (PDF). Toshiba. Archived from the original (PDF) on 17 July 2011. Retrieved 17 May 2009.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ "Organometallics". IHS Chemicals. February 2012.
- .
- .
- ISBN 0-12-031118-6.
- ISBN 0-7637-0665-5. Archived from the original on 18 June 2016.)
{{cite book}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help - ^ LiAl-hydride
- doi:10.2514/3.62644.)
{{cite journal}}: Unknown parameter|last-author-amp=ignored (|name-list-style=suggested) (help - ^ Emsley, John (2011). Nature's Building Blocks.
- ISBN 0-262-63204-7. Archived from the original on 13 June 2016.)
{{cite book}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help); Unknown parameter|last-author-amp=ignored (|name-list-style=suggested) (help - ISBN 0-309-05226-2. Archived from the original on 13 June 2016.)
{{cite book}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help - ISBN 0-415-07674-9. Archived from the original on 9 June 2016.)
{{cite book}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help - .
- ISBN 81-7648-743-0. Archived from the original on 29 June 2016.)
{{cite book}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help - ^ "'Splitting the Atom': Cockcroft and Walton, 1932: 9. Rays or Particles?" Archived 2 September 2012 at the Wayback Machine Department of Physics, University of Cambridge
- ^ a b Wald, Matthew L. (8 October 2013). "Report Says a Shortage of Nuclear Ingredient Looms". The New York Times. Archived from the original on 1 July 2017.
{{cite news}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ a b Kean, Sam (2011). The Disappearing Spoon.
- PMID 18982835.
- .
- ^ PMID 11838882.
- ^ Marshall, Timothy M. (2015). "Lithium as a Nutrient" (PDF). Journal of American Physicians and Surgeons. 20 (4): 104–9. Archived from the original (PDF) on 15 February 2017.
{{cite journal}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ Technical data for Lithium Archived 23 March 2015 at the Wayback Machine. periodictable.com
- ISBN 978-0-8493-2523-6.
- ^ "Illinois Attorney General – Basic Understanding Of Meth". Illinoisattorneygeneral.gov. Archived from the original on 10 September 2010. Retrieved 6 October 2010.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ Harmon, Aaron R. (2006). "Methamphetamine remediation research act of 2005: Just what the doctor ordered for cleaning up methfields—or sugar pill placebo?" (PDF). North Carolina Journal of Law & Technology. 7. Archived from the original (PDF) on 1 December 2008. Retrieved 5 October 2010.
{{cite journal}}: Unknown parameter|dead-url=ignored (|url-status=suggested) (help) - ISBN 978-0-306-44758-7.)
{{cite book}}: Unknown parameter|last-author-amp=ignored (|name-list-style=suggested) (help - ^ "TSA: Safe Travel with Batteries and Devices". Tsa.gov. 1 January 2008. Archived from the original on 4 January 2012.
External links
- Lithium at The Periodic Table of Videos(University of Nottingham)
- International Lithium Alliance
- USGS: Lithium Statistics and Information
- Lithium Supply & Markets 2009 IM Conference 2009 Sustainable lithium supplies through 2020 in the face of sustainable market growth
- University of Southampton, Mountbatten Centre for International Studies, Nuclear History Working Paper No5.



