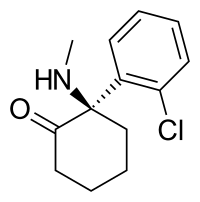
Arketamine
 | |
 | |
| Clinical data | |
|---|---|
| Other names | PCN-101; HR-071603 |
| Addiction liability | Moderate |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
JSmol) | |
| |
| |
Arketamine (developmental code names PCN-101, HR-071603), also known as (R)-ketamine or (R)-(−)-ketamine, is the (R)-(−)
Relative to esketamine, arketamine possesses 4 to 5 times lower
Novel antidepressant
Arketamine appears to be more effective as a rapid-acting antidepressant than esketamine in
In
A study conducted in mice found that ketamine's antidepressant activity is not caused by ketamine inhibiting NMDAR, but rather by sustained activation of a different glutamate receptor, the
Paradoxically, arketamine shows greater and longer-lasting rapid
Clinical development
As of November 2019, arketamine is under development for the treatment of depression under the developmental code names PCN-101 by Perception Neuroscience in the United States and HR-071603 by Jiangsu Hengrui Medicine in China.[4][5]
See also
References
- ^ ISBN 978-0-412-46630-4.
- ^ ISBN 978-1-4665-8340-5.
- ^ PMID 26707087.
- ^ PMID 31215725.
- ^ a b "Arketamine - Jiangsu Hengrui Medicine". AdisInsight. Springer Nature Switzerland AG.
- ^ ISBN 978-1-4511-4795-7.
- ^ ISBN 978-1-4614-3375-0.
- S2CID 26861697.
- S2CID 24966884.
- S2CID 22725552.
- S2CID 10307361.
- ISBN 978-1-4419-9192-8.
- ^ S2CID 140205448.
- ^ PMID 26327690.
- PMID 28418641.
- PMID 27144355.
- S2CID 19842307.
- PMID 24851126.
- PMID 24936919.
- PMID 24936922.
- PMID 25331415.
