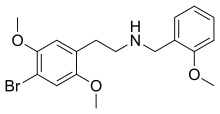
25B-NBOMe
This article needs additional citations for verification. (December 2022) |
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
25B-NBOMe (NBOMe-2C-B, Cimbi-36, Nova, BOM 2-CB) is a derivative of the
The carbon-11 labeled version of this compound ([11C]Cimbi-36) was synthesized and validated as a radioactive tracer for positron emission tomography (PET) in Copenhagen.[9][10][11][12][13][excessive citations] As a 5-HT2A receptor agonist PET radioligand, [11C]Cimbi-36 was hypothesized to provide a more functional marker of these receptors. Also, [11C]Cimbi-36 is investigated as a potential marker of serotonin release and thus could serve as an indicator of serotonin levels in vivo. [11C]Cimbi-36 is now undergoing clinical trials as a PET-ligand in humans.[14][15][16]
Toxicity and harm potential
NBOMe compounds are often associated with life-threatening toxicity and death.
NBOMe and NBOHs are regularly sold as LSD in blotter papers,[18][26] which have a bitter taste and different safety profiles.[20][17] Despite high potency, recreational doses of LSD have only produced low incidents of acute toxicity.[17] Fatalities involved in NBOMe intoxication suggest that a significant number of individuals ingested the substance which they believed was LSD,[22] and researchers report that "users familiar with LSD may have a false sense of security when ingesting NBOMe inadvertently".[20] While most fatalities are due to the physical effects of the drug, there have also been reports of death due to self-harm and suicide under the influence of the substance.[27][28][20]
Given limited documentation of NBOMe consumption, the long-term effects of the substance remain unknown.Neurotoxic and cardiotoxic actions
Many of the NBOMe compounds have high potency agonist activity at additional 5-HT receptors and prolonged activation of 5-HT2B can cause cardiac valvulopathy in high doses and chronic use.[18][23] 5-HT2B receptors have been strongly implicated in causing drug-induced valvular heart disease.[34][35][36] The high affinity of NBOMe compounds for adrenergic α1 receptor has been reported to contribute to the stimulant-type cardiovascular effects.[23]
In vitro studies, 25C-NBOMe has been shown to exhibit cytotoxicity on neuronal cell lines SH-SY5Y, PC12, and SN471, and the compound was more potent than methamphetamine at reducing the visibility of the respective cells; the neurotoxicity of the compound involves activation of MAPK/ERK cascade and inhibition of Akt/PKB signaling pathway.[19] 25C-NBOMe, including the other derivative 25D-NBOMe, reduced the visibility of cardiomyocytes H9c2 cells, and both substances downregulated expression level of p21 (CDC24/RAC)-activated kinase 1 (PAK1), an enzyme with documented cardiac protective effects.[19]
Preliminary studies on 25C-NBOMe have shown that the substance is toxic to development, heart health, and brain health in zebrafish, rats, and Artemia salina, a common organism for studying potential drug effects on humans, but more research is needed on the topic, the dosages, and if the toxicology results apply to humans. Researchers of the study also recommended further investigation of the drug's potential in damaging pregnant women and their fetus due to the substance's damaging effects to development.[37][38]Emergency treatment
Analogues and derivatives
Analogues and derivatives of 2C-B:
25-N:
- 25B-NB
- 25B-NB23DM
- 25B-NB25DM
- 25B-NB3OMe
- 25B-NB4OMe
- 25B-NBF
- 25B-NBMD
- 25B-NBOH
- 25B-NBOMe (NBOMe-2CB)
- 2C-B-FLY
- 2CBFly-NBOMe (NBOMe-2CB-Fly)
- DOB-FLY
- DOB-2-DRAGONFLY-5-BUTTERFLY
Other:
- BOB
- BOH-2C-B, β-Hydroxy-2C-B, βOH-2CB[41][42]
- BMB
- 2C-B-5-hemifly
- 2C-B-aminorex (2C-B-AR)
- 2C-B-AN
- 2C-B-BZP
- 2C-B-FLY-NB2EtO5Cl
- 2C-B-PP
- 2CB-Ind
- βk-2C-B (beta-keto 2C-B)
- N-Ethyl-2C-B
- TCB-2 (2C-BCB)
Legal status
Canada
As of October 31, 2016; 25B-NBOMe is a controlled substance (Schedule III) in Canada.[43]
Russia
Banned as a narcotic drug since May 5, 2015.[44]
Sweden
In Sweden, the Riksdag added 25B-NBOMe to schedule I ("substances, plant materials and fungi which normally do not have medical use") as narcotics in Sweden as of August 1, 2013, published by the Medical Products Agency in their regulation LVFS 2013:15 listed as 25B-NBOMe 2-(4-bromo-2,5-dimetoxifenyl)-N-(2-metoxibensyl)etanamin.[45]
United Kingdom
This substance is a
United States
In November 2013, the U.S. Drug Enforcement Administration placed 25B-NBOMe (along with 25I-NBOMe and 25C-NBOMe) in Schedule I of the Controlled Substances Act, making it illegal to manufacture, buy, possess, process, or distribute.[47]
China
As of October 2015 25B-NBOMe is a controlled substance in China.[48]
Czech Republic
25B-NBOMe is banned in the Czech Republic.[49]
Notes
- ^ The potency of N-benzylphenethylamines via buccal, sublingual, or nasal absorption is 50-100 greater (by weight) than oral route compared to the parent 2C-x compounds.[29] Researchers hypothesize the low oral metabolic stability of N-benzylphenethylamines is likely causing the low bioavailability on the oral route, although the metabolic profile of this compounds remains unpredictable; therefore researchers state that the fatalities linked to these substances may partly be explained by differences in the metabolism between individuals.[29]
References
- ^ Anvisa (July 24, 2023). "RDC Nº 804 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 804 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published July 25, 2023). Archived from the original on August 27, 2023. Retrieved August 27, 2023.
- ^ "Substance Details 25B-NBOMe". Retrieved January 22, 2024.
- ^ Heim R (February 28, 2010). "Synthese und Pharmakologie potenter 5-HT2A-Rezeptoragonisten mit N-2-Methoxybenzyl-Partialstruktur. Entwicklung eines neuen Struktur-Wirkungskonzepts" (in German). diss.fu-berlin.de. Retrieved May 10, 2013.
- ^ Silva M (2009). Theoretical study of the interaction of agonists with the 5-HT2A receptor (Ph.D. thesis). Universität Regensburg.
- S2CID 3103050.
- PMID 24397362.
- ^ PMID 24780897.
- S2CID 208217863.
- .
- S2CID 12467684.
- S2CID 1474367.
- PMID 29215308.
- PMID 31367837.
- ^ "From molecule to man: The full CIMBI-36 story" (PDF). cimbi.dk. Retrieved January 10, 2014.
- ^ "Imanova announces the launch of a new imaging biomarker to investigate the serotonin system in psychiatric illness". imanova.co.uk. Archived from the original on April 9, 2015. Retrieved April 9, 2015.
- PMID 30685771.
- ^ S2CID 247764056.
- ^ PMID 30261175.
- ^ PMID 32174803.
- ^ S2CID 247888583.
- PMID 35530025.
- ^ PMID 31915427.
- ^ S2CID 10382311.
- ^ S2CID 25752763.
- ISSN 1923-4163.
- PMID 26378135.
- PMID 28893436.
- S2CID 4734566.
- ^ S2CID 254857910.
- PMID 28097528.
- PMID 27406128.
- PMID 25105138.
- S2CID 240583877.
- PMID 11104741.
- PMID 10617681.
- PMID 17202450.
- S2CID 255763701.
- PMID 33598409.
- ^ "Explore N-(2C-B)-Fentanyl | PiHKAL · info". isomerdesign.com.
- ^ "Explore N-(2C-FLY)-Fentanyl | PiHKAL · info". isomerdesign.com.
- PMID 15537358.
- ^ Beta-hydroxyphenylalkylamines and their use for treating glaucoma
- ^ "Regulations Amending the Food and Drug Regulations (Part J — 2C-phenethylamines)". Canada Gazette. Vol. 150, no. 9. May 4, 2016. Archived from the original on August 31, 2016.
- ^ "Постановление Правительства РФ от 30.06.1998 N 681 "Об утверждении перечня наркотических средств, психотропных веществ и их прекурсоров, подлежащих контролю в Российской Федерации" (с изменениями и дополнениями)". base.garant.ru.
- ^ "Föreskrifter om ändring i Läkemedelsverkets föreskrifter (LVFS 2011:10) om förteckningar över narkotika;" (PDF). lakemedelsverket.se (in Swedish). Retrieved October 4, 2013.
- ^ "The Misuse of Drugs Act 1971 (Ketamine etc.) (Amendment) Order 2014". UK Statutory Instruments 2014 No. 1106. www.legislation.gov.uk.
- ^ "2016 - Final Rule: Placement of Three Synthetic Phenethylamines Into Schedule I". www.deadiversion.usdoj.gov. Retrieved April 2, 2018.
- ^ "关于印发《非药用类麻醉药品和精神药品列管办法》的通知" (in Chinese). China Food and Drug Administration. September 27, 2015. Archived from the original on October 1, 2015. Retrieved October 1, 2015.
- ^ "Látky, o které byl doplněn seznam č. 4 psychotropních látek (příloha č. 4 k nařízení vlády č. 463/2013 Sb.)" (PDF) (in Czech). Ministerstvo zdravotnictví. Archived from the original (PDF) on March 9, 2016. Retrieved February 6, 2016.
