3C-BZ
This article relies largely or entirely on a single source. Relevant discussion may be found on the talk page. Please help improve this article by introducing citations to additional sources. Find sources: "3C-BZ" – news · newspapers · books · scholar · JSTOR (September 2019) |
| |
| |
| Names | |
|---|---|
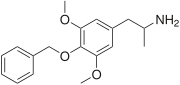
| Preferred IUPAC name
1-[4-(Benzyloxy)-3,5-dimethoxyphenyl]propan-2-amine | |
| Other names
4-Benzyloxy-3,5-methoxyamphetamine
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C18H23NO3 | |
| Molar mass | 301.386 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
3C-BZ (4-benzyloxy-3,5-dimethoxyamphetamine) is a lesser-known
Synthesis
3C-BZ was originally synthesized by Alexander Shulgin starting from 5-methoxyeugenol (4-allyl-2,6-dimethoxyphenol) through a reaction with benzyl chloride to form the benzyloxy derivative of 5-methoxyeugenol.[1] The obtained benzyl derivative was reacted with tetranitromethane to form 1-[4-(benzyloxy)-3,5-dimethoxyphenyl]-2-nitro-1-propene, from which 3C-BZ is obtained by reduction of the nitropropene with lithium aluminium hydride.
Another possible synthetic route would be the reaction of benzyl chloride with syringaldehyde to form 3,5-dimethoxy-4-benzyloxybenzaldehyde followed by condensation with nitroethane to form 1-[4-(benzyloxy)-3,5-dimethoxyphenyl]-2-nitro-1-propene. The obtained nitropropene can be reduced using lithium aluminium hydride, Red-Al, or an aluminium-mercury amalgam.
References
- ^
(NMDAR
antagonists)
| Arylcyclo‐ hexylamines |
| ||||||
|---|---|---|---|---|---|---|---|
| Adamantanes | |||||||
| Diarylethylamines | |||||||
| Morphinans |
| ||||||
| Others |
(mAChR
antagonists)
- Atropine
- Benactyzine
- Benzatropine
- Benzydamine
- Biperiden
- BRN-1484501
- Brompheniramine
- BZ
- CAR-226,086
- CAR-301,060
- CAR-302,196
- CAR-302,282
- CAR-302,368
- CAR-302,537
- CAR-302,668
- Chloropyramine
- Chlorphenamine
- Clemastine
- CS-27349
- Cyclizine
- Cyproheptadine
- Dicycloverine
- Dimenhydrinate
- Diphenhydramine
- Ditran
- Doxylamine
- EA-3167
- EA-3443
- EA-3580
- EA-3834
- Flavoxate
- Hyoscyamine
- JB-318
- JB-336
- Meclozine
- Mepyramine
- Orphenadrine
- Oxybutynin
- Pheniramine
- Phenyltoloxamine
- Procyclidine
- Promethazine
- Scopolamine
- Tolterodine
- Trihexyphenidyl
- Tripelennamine
- Triprolidine
- WIN-2299
Natural | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Synthetic |
|
enhancers
(Mixed MOA)
- Aliphatic hydrocarbons
- Aromatic hydrocarbons
- Ethers
- Haloalkanes
- 2-EMSB
- Alazocine
- Bremazocine
- Butorphan
- Butorphanol
- Cyclazocine
- Cyclorphan
- Cyprenorphine
- Diprenorphine
- Enadoline
- Herkinorin
- Heroin
- HZ-2
- Ibogaine
- Ketazocine
- Levallorphan
- Levomethorphan
- Levorphanol
- LPK-26
- Metazocine
- Morphine
- Nalbuphine
- Nalmefene
- Nalorphine
- Noribogaine
- Oxilorphan
- Pentazocine
- Phenazocine
- Proxorphan
- Racemethorphan
- Racemorphan
- Salvinorin A
- Spiradoline
- Tifluadom
- U-50488
- U-69,593
- Xorphanol
| Phenethylamines |
|
|---|---|
| Amphetamines |
|
| Phentermines |
|
| Cathinones |
|
| Phenylisobutylamines | |
| Phenylalkylpyrrolidines | |
| Catecholamines (and close relatives) |
|
| Miscellaneous |
|
| Psychedelics (5-HT2A agonists) |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Others | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
(NMDAR
antagonists)
| Arylcyclo‐ hexylamines |
| ||||||
|---|---|---|---|---|---|---|---|
| Adamantanes | |||||||
| Diarylethylamines | |||||||
| Morphinans |
| ||||||
| Others |
(mAChR
antagonists)
- Atropine
- Benactyzine
- Benzatropine
- Benzydamine
- Biperiden
- BRN-1484501
- Brompheniramine
- BZ
- CAR-226,086
- CAR-301,060
- CAR-302,196
- CAR-302,282
- CAR-302,368
- CAR-302,537
- CAR-302,668
- Chloropyramine
- Chlorphenamine
- Clemastine
- CS-27349
- Cyclizine
- Cyproheptadine
- Dicycloverine
- Dimenhydrinate
- Diphenhydramine
- Ditran
- Doxylamine
- EA-3167
- EA-3443
- EA-3580
- EA-3834
- Flavoxate
- Hyoscyamine
- JB-318
- JB-336
- Meclozine
- Mepyramine
- Orphenadrine
- Oxybutynin
- Pheniramine
- Phenyltoloxamine
- Procyclidine
- Promethazine
- Scopolamine
- Tolterodine
- Trihexyphenidyl
- Tripelennamine
- Triprolidine
- WIN-2299
Natural | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Synthetic |
|
enhancers
(Mixed MOA)
- Aliphatic hydrocarbons
- Aromatic hydrocarbons
- Ethers
- Haloalkanes
- 2-EMSB
- Alazocine
- Bremazocine
- Butorphan
- Butorphanol
- Cyclazocine
- Cyclorphan
- Cyprenorphine
- Diprenorphine
- Enadoline
- Herkinorin
- Heroin
- HZ-2
- Ibogaine
- Ketazocine
- Levallorphan
- Levomethorphan
- Levorphanol
- LPK-26
- Metazocine
- Morphine
- Nalbuphine
- Nalmefene
- Nalorphine
- Noribogaine
- Oxilorphan
- Pentazocine
- Phenazocine
- Proxorphan
- Racemethorphan
- Racemorphan
- Salvinorin A
- Spiradoline
- Tifluadom
- U-50488
- U-69,593
- Xorphanol
