Claisen condensation
Appearance
Source: Wikipedia, the free encyclopedia.
Chemical reaction
Not to be confused with Claisen rearrangement or Claisen–Schmidt condensation.
| Claisen condensation | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Named after | Rainer Ludwig Claisen | ||||||||||||
| Reaction type | Condensation reaction | ||||||||||||
| Reaction | |||||||||||||
| |||||||||||||
| Conditions | |||||||||||||
| Typical solvents | R'-OH
| ||||||||||||
| Identifiers | |||||||||||||
| Organic Chemistry Portal | claisen-condensation | ||||||||||||
| RSC ontology ID | RXNO:0000043 | ||||||||||||
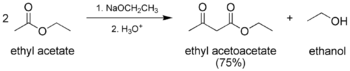
The Claisen acetoacetic esters.[5]
Requirements
At least one of the
α-proton and be able to undergo deprotonation to form the enolate anion). There are a number of different combinations of enolizable and nonenolizable carbonyl compounds that form a few different types
of Claisen.
The base used must not interfere with the reaction by undergoing
electrophilic
ester.
The alkoxy portion of the ester must be a relatively good
methoxide
and ethoxide, respectively, are commonly used.
Types
- The classic Claisen condensation, a self-condensation between two molecules of a compound containing an enolizable ester.
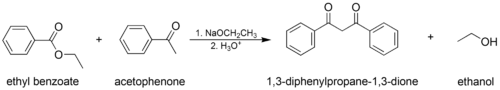
- The mixed (or "crossed") Claisen condensation, where one enolizable ester or ketone and one nonenolizable ester are used.
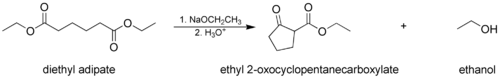
- The Dieckmann condensation, where a molecule with two ester groups reacts intramolecularly, forming a cyclic β-keto ester. In this case, the ring formed must not be strained, usually a 5- or 6-membered chain or ring.
- Retro-Claisen condensation is the reverse of the title reaction, i.e., the base-induced cleavage of 2-ketoesters
Mechanism

In the first step of the mechanism, an α-proton is removed by a strong base, resulting in the formation of an enolate anion, which is made relatively stable by the
α-hydrogen
because of the driving force effect of deprotonation of the β-keto ester in the last step.

|
| animation |
See also
- Aldol condensation
- Stobbe condensation
- Fatty acid synthesis
- Polyketide synthase
- Dieckmann condensation
References
- ISBN 0-07-111562-5.
- .
- .
- ISBN 0471264180.
- ISBN 3527306730.
External links
- "Claisen Condensation". Organic Chemistry Portal.
| Authority control databases: National |
|---|