Friedländer synthesis
Source: Wikipedia, the free encyclopedia.
| Friedländer synthesis | |
|---|---|
| Named after | Paul Friedländer |
| Reaction type | Ring forming reaction |
| Identifiers | |
| Organic Chemistry Portal | friedlaender-synthesis |
The Friedländer synthesis is a chemical reaction of 2-aminobenzaldehydes[1] with ketones to form quinoline derivatives.[2][3] It is named after German chemist Paul Friedländer (1857–1923).
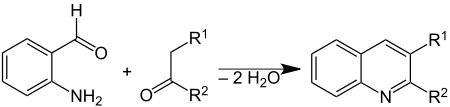
This reaction has been catalyzed by
Lewis acids.[7]
Several reviews have been published.[8][9][10]
Mechanism
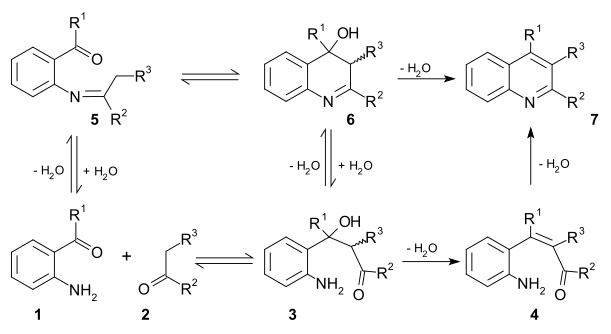
Two viable
unsaturated carbonyl compound 4 and then loses water again in imine formation to quinoline 7. In the second mechanism the first step is Schiff base formation to 5 followed by Aldol reaction to 6 and elimination to 7.[11]
The Pfitzinger reaction and the Niementowski quinoline synthesis are variations of the Friedländer reaction.
See also
- Doebner-Miller reaction
- Povarov reaction
- Skraup reaction
References
- ^ Organic Syntheses, Coll. Vol. 3, p. 56 (1955); Vol. 28, p. 11 (1948). (Article)
- .
- .
- S2CID 98625429.
- PMID 16358003.
- PMID 16358006.
- .
- .
- .
- ISBN 0471264180.
- PMID 19361199.