Hydroboration–oxidation reaction
Hydroboration–oxidation reaction is a two-step
The general form of the reaction is as follows:

Tetrahydrofuran (THF) is the archetypal solvent used for hydroboration.
Mechanism and scope
Hydroboration step
In the first step,

Knowing that the group containing the boron will be replaced by a hydroxyl group, it can be seen that the initial hydroboration step determines the regioselectivity. Hydroboration proceeds in an
Until all hydrogens attached to boron have been transferred away, the boron group BH2 will continue adding to more alkenes. This means that one mole of hydroborane will undergo the reaction with three moles of alkene. Furthermore, it is not necessary for the hydroborane to have more than one hydrogen. For example, reagents of the type R2BH are commonly used, where R can represents the remainder of the molecule. Such modified hydroboration reagents include 9-BBN, catecholborane, and disiamylborane.
Oxidation step
In the second step of the reaction sequence, the nucleophilic

The 'H' atom in the reaction comes from B2H6, the 'O' atom comes from hydrogen peroxide (H2O2) whereas the O attached 'H' atom comes from the solvent (refer mechanism).
Alkyne hydroboration
A hydroboration reaction also takes place on alkynes. Again the mode of action is syn and secondary reaction products are aldehydes from terminal alkynes and ketones from internal alkynes. In order to prevent hydroboration across both the pi-bonds, a bulky borane like disiamyl (di-sec-iso-amyl) borane is used.[5]
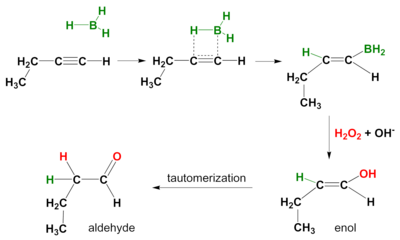
Alternative oxidations
Use of other oxidants instead of hydrogen peroxide can lead to carbonyl products rather than alcohols from alkenes.
References
- ISBN 0-19-511999-1.
- .
- .
- .
- .
- .
- .
External links
- Organic Chemistry Portal. Hydroboration (including recent literature). https://www.organic-chemistry.org/namedreactions/brown-hydroboration.shtm
