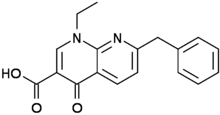
Amfonelic acid
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Amfonelic acid (AFA; WIN 25,978) is a research chemical and dopaminergic stimulant with antibiotic properties. Although limited clinical trials have been conducted, it's primarily used in scientific research.[1]
History
The stimulant properties of AFA were discovered serendipitously at
Pharmacology
In studies, it proved to be a potent and highly selective
Despite its different mechanism of action, amfonelic acid displays discriminatory substitution with 150% the stimulant potency of dextroamphetamine.[9] Amfonelic acid has been shown to be neuroprotective against methamphetamine damage to dopamine neurons.[10] It also increases the effects of the antipsychotic drugs haloperidol, trifluoperazine and spiperone.[11] Rats are shown to self-administer amfonelic acid in a dose-dependent manner.[12]
Though AFA was discovered in the course of antibiotic research, there is very little data available on the drug's antimicrobial activity. In 1988, the biologist G.C. Crumplin wrote, "[AFA] is less active against bacteria than are many other 4-quinolones, but studies in our laboratory on selected mammalian cell lines have shown it to be markedly more toxic to these cells than are the 4-quinolones that are more active antibacterial agents. Furthermore, it can be shown that sublethal doses induced marked changes in the pattern of proteins produced by the cell, thus suggesting a possible effect of 4-quinolones on gene transcription in mammalian cells."[13] When evaluated via broth microdilution the MIC of AFA for Escherichia coli is 125 μg/mL, a concentration thirty times higher than the MIC for nalidixic acid in the same E. coli strain.[1]
See also
- Nalidixic acid (WIN 18,320)
- Oxolinic acid
- Pipemidic acid
- A-77636
References
- ^ a b c d e f g Morris H (October 2015). "Sad Pink Monkey Blues". Harper's Magazine. Retrieved 2015-09-19.
- ^ US patent 3590036, "Naphthyridine-3-carboxylic Acids, Their Derivatives and Preparation Thereof"
- PMID 4393073.
- S2CID 28881012.
- S2CID 34793566.
- S2CID 24436917.
- S2CID 13008483.
- S2CID 19112365.
- S2CID 30259648.
- S2CID 23292364.
- PMID 2857083.
- S2CID 24551129.
- PMID 3279494.
